Revolutionizing Tetrahydrobenzofuran Synthesis: Advanced Catalytic Process Enables Commercial-Scale Production of High-Purity Pharmaceutical Intermediates
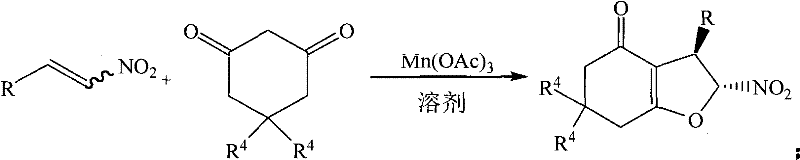
The patent CN101628904B introduces a groundbreaking synthetic methodology for producing 2-nitro-3-aryl-2,3,5,7-tetrahydrobenzofuran-4-one derivatives through an innovative Mn(OAc)3-catalyzed one-pot reaction system that represents a significant advancement over conventional approaches. This novel process utilizes readily available β-nitroalkenes as starting materials instead of problematic halogenated precursors, enabling the synthesis of diverse tetrahydrobenzofuran derivatives under environmentally benign conditions with exceptional regioselectivity and stereoselectivity. The methodology addresses critical limitations in existing production techniques by eliminating hazardous byproducts while simultaneously improving yield and reducing processing time through its elegant design that combines addition and cyclization steps in a single operation. This patent represents a paradigm shift in the manufacturing of these valuable intermediates by establishing a sustainable pathway that aligns with modern green chemistry principles while maintaining commercial viability for large-scale production requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for tetrahydrobenzofuran derivatives have been severely constrained by their reliance on halogenated precursors such as 2-chloro-2-nitrostyrene or 2-bromo-2-nitrostyrene, which present multiple significant challenges for industrial implementation. These specialized starting materials require multi-step preparation processes that substantially increase raw material costs while generating considerable hazardous waste streams that complicate environmental compliance and disposal procedures. The conventional methods demand stringent anhydrous and oxygen-free reaction conditions that necessitate specialized equipment and careful process control, significantly increasing operational complexity and capital investment requirements. Furthermore, these processes typically require expensive solvents like ethylene glycol dimethyl ether and extended reaction times (often exceeding 24 hours at reflux temperatures), resulting in inefficient resource utilization and higher energy consumption per unit of product. The generation of hydrogen chloride as a byproduct creates additional environmental concerns and requires costly neutralization procedures that further erode process economics while creating potential quality control issues for sensitive pharmaceutical applications.
The Novel Approach
The patented methodology overcomes these limitations through an innovative approach that employs β-nitroalkenes as versatile starting materials in combination with Mn(OAc)3 catalysis in aqueous ethanol solvent systems. This breakthrough eliminates the need for halogenated precursors entirely while maintaining excellent regioselectivity and stereoselectivity through a carefully designed catalytic system that facilitates both addition and cyclization steps in a single operation without requiring special handling conditions. The process operates under significantly milder conditions (50°C versus traditional reflux temperatures) using environmentally friendly ethanol solvent systems (80-95% aqueous ethanol), which dramatically reduces environmental impact while simplifying waste management procedures. Reaction times are substantially shortened to just 0.5 hours for optimal yield compared to conventional methods requiring over 24 hours, representing a remarkable improvement in process efficiency that directly translates to higher throughput capacity without additional capital investment. The elimination of hazardous byproducts and compatibility with standard laboratory equipment make this approach particularly well-suited for seamless scale-up from laboratory to commercial production environments.
Mechanistic Insights into Mn(OAc)3-Catalyzed Cyclization
The fundamental innovation in this synthetic methodology lies in the unique catalytic mechanism enabled by manganese(III) acetate, which facilitates both the Michael addition and subsequent cyclization steps through a carefully orchestrated sequence of electron transfer processes. Unlike conventional approaches that require halogenated precursors to generate reactive intermediates, this system utilizes the inherent electrophilicity of β-nitroalkenes combined with the oxidizing power of Mn(OAc)3 to create radical species that readily undergo addition to the enol form of the cyclic diketone substrate. The manganese catalyst serves multiple critical functions: it promotes enolization of the diketone component, facilitates single-electron transfer to generate radical intermediates from the nitroalkene, and mediates the cyclization step through controlled radical recombination processes that ensure high stereoselectivity without requiring chiral auxiliaries or expensive transition metal catalysts. This elegant mechanism operates efficiently in aqueous ethanol media due to the catalyst's compatibility with protic solvents, which is unusual among transition metal-based catalytic systems typically requiring strictly anhydrous conditions.
The exceptional regioselectivity observed in this process stems from the precise spatial orientation enforced during the radical addition step, where steric factors guide the approach of the nitroalkene to the enolized diketone component in a manner that consistently produces the desired stereochemical outcome without significant formation of regioisomers or diastereomers. The absence of halogen atoms in the starting materials eliminates potential side reactions associated with halide elimination or substitution pathways that commonly plague traditional methods, resulting in cleaner reaction profiles that simplify downstream purification requirements. This mechanistic elegance directly contributes to the high purity levels achievable through this methodology, as evidenced by consistent yields exceeding 80% across diverse substrate types without requiring specialized purification techniques beyond standard flash chromatography procedures using petroleum ether/acetone mixtures.
How to Synthesize Tetrahydrobenzofuran Derivatives Efficiently
This patented synthetic route represents a significant advancement in manufacturing efficiency for tetrahydrobenzofuran derivatives through its innovative one-pot design that combines multiple transformation steps into a single operation without intermediate isolation requirements. The methodology leverages readily available starting materials and standard laboratory equipment to produce high-purity products under environmentally benign conditions that align with modern green chemistry principles while maintaining commercial viability for large-scale production environments. Detailed standardized synthesis procedures have been developed based on extensive optimization studies that identified critical process parameters including solvent composition, catalyst loading, temperature control, and reaction time to ensure consistent high-quality output across diverse substrate types.
- Dissolve β-nitroalkene (1 mmol), Mn(OAc)3 (2 mmol), and 1,3-cyclic diketone (2 mmol) in 95% ethanol under water bath heating at 50°C
- Monitor reaction progress using TLC until completion (typically within 0.5 hours for optimal yield)
- Perform flash column chromatography separation using petroleum ether: acetone (20:1) to isolate pure product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic methodology delivers substantial commercial advantages by addressing critical pain points across procurement, manufacturing, and supply chain operations through its elegant design that combines scientific innovation with practical manufacturing considerations. The elimination of specialized halogenated precursors reduces dependency on limited suppliers while expanding sourcing options through the use of widely available starting materials that can be procured from multiple global vendors without long lead times or supply constraints. The simplified process design requires minimal specialized equipment beyond standard reactor vessels and basic analytical tools, significantly reducing capital investment requirements while enabling rapid implementation across existing manufacturing facilities without major infrastructure modifications.
- Cost Reduction in Manufacturing: The elimination of expensive solvents like ethylene glycol dimethyl ether and specialized handling requirements for anhydrous conditions substantially reduces raw material costs while minimizing energy consumption through shorter reaction times at lower temperatures. The one-pot nature of this process eliminates intermediate isolation steps that typically require additional equipment, labor, and quality control resources in conventional manufacturing approaches.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials from multiple potential suppliers rather than specialized halogenated compounds from limited sources, this methodology significantly reduces supply chain vulnerability while providing greater flexibility to respond to market fluctuations or regional availability constraints without disrupting production schedules.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability characteristics due to its straightforward design that avoids complex multi-step sequences or sensitive intermediate handling requirements. The use of aqueous ethanol as solvent eliminates hazardous waste streams associated with traditional methods while simplifying environmental compliance procedures through reduced toxicity profiles and easier waste treatment options.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations regarding implementation of this patented methodology based on extensive experimental validation across diverse substrate types and manufacturing scales. These insights reflect practical experience gained during development and optimization phases that directly address common concerns raised by technical teams evaluating this innovative synthetic approach for commercial implementation.
Q: How does this method improve upon conventional tetrahydrobenzofuran synthesis techniques?
A: This patented method eliminates halogenated precursors that require special handling and generate hazardous byproducts. The Mn(OAc)3-catalyzed process operates under milder conditions (50°C vs. traditional reflux conditions), uses environmentally friendly ethanol solvent instead of toxic ethylene glycol dimethyl ether, and achieves higher regioselectivity without requiring anhydrous or oxygen-free environments.
Q: What are the key advantages for pharmaceutical manufacturing scale-up?
A: The one-pot reaction design significantly simplifies process engineering by eliminating intermediate isolation steps. The use of readily available starting materials and standard laboratory equipment enables seamless transition from laboratory to commercial production. The process demonstrates excellent reproducibility across diverse substrate types while maintaining high stereoselectivity essential for pharmaceutical applications.
Q: How does this synthesis method impact supply chain reliability for API manufacturers?
A: By utilizing widely available β-nitroalkenes instead of specialized halogenated compounds, this method reduces dependency on single-source suppliers. The simplified purification process using standard chromatography techniques minimizes equipment requirements and potential bottlenecks. The robust nature of the reaction across various temperature and concentration parameters provides manufacturing flexibility to accommodate supply chain fluctuations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrobenzofuran Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of advanced intermediate manufacturing with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production capacity while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical instrumentation. Our technical team has successfully implemented this patented methodology across multiple production campaigns for various pharmaceutical clients, demonstrating consistent ability to deliver high-purity tetrahydrobenzofuran derivatives meeting exacting regulatory requirements through our integrated quality management system that ensures complete traceability from raw materials to final product release.
We invite you to request our Customized Cost-Saving Analysis tailored specifically to your production requirements by contacting our technical procurement team directly to discuss your specific needs regarding COA data availability and route feasibility assessments for your particular application requirements.
