Advanced Manganese-Promoted Synthesis of 2-Nitro-3-Aryl-2,3,5,7-Tetrahydrobenzofuran-4-One Derivatives for Scalable Pharma Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for complex heterocyclic scaffolds. Patent CN101628904B introduces a significant breakthrough in the synthesis of 2-nitro-3-aryl-2,3,5,7-tetrahydrobenzofuran-4-one derivatives, which serve as critical intermediates for bioactive molecules. This technology leverages a manganese(III) acetate-promoted radical cyclization strategy that fundamentally shifts the paradigm from harsh, halogen-dependent chemistry to a more sustainable and efficient process. By utilizing readily available beta-nitroalkenes and 1,3-cyclic diketones, this method achieves high regioselectivity and stereoselectivity under mild conditions. For R&D directors and procurement managers alike, this represents a tangible opportunity to optimize supply chains for high-purity pharmaceutical intermediates while adhering to stricter green chemistry standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of dihydrofuran rings fused to cyclohexanone systems relied heavily on nucleophilic substitution strategies involving halogenated precursors. As illustrated in the prior art by Daniel Dauzonne, the traditional synthesis required the use of 2-chloro-2-nitrostyrene reacting with 5,5-dimethyl-1,3-cyclohexanedione in the presence of potassium fluoride.  This legacy approach suffers from severe logistical and economic drawbacks. The requirement for anhydrous ethylene glycol dimethyl ether (DME) as a solvent necessitates rigorous drying protocols and increases raw material costs significantly. Furthermore, the reaction conditions are exceptionally harsh, requiring reflux for 24 hours under strictly anhydrous and oxygen-free inert gas protection. From a supply chain perspective, the reliance on specialized 2-halo-2-nitrostyrene substrates creates a bottleneck, as these materials are difficult to prepare, involve multiple synthetic steps, and generate hazardous hydrogen chloride byproducts that complicate waste management and increase environmental compliance costs.
This legacy approach suffers from severe logistical and economic drawbacks. The requirement for anhydrous ethylene glycol dimethyl ether (DME) as a solvent necessitates rigorous drying protocols and increases raw material costs significantly. Furthermore, the reaction conditions are exceptionally harsh, requiring reflux for 24 hours under strictly anhydrous and oxygen-free inert gas protection. From a supply chain perspective, the reliance on specialized 2-halo-2-nitrostyrene substrates creates a bottleneck, as these materials are difficult to prepare, involve multiple synthetic steps, and generate hazardous hydrogen chloride byproducts that complicate waste management and increase environmental compliance costs.
The Novel Approach
In stark contrast, the methodology disclosed in CN101628904B utilizes a manganese(III) acetate promoter to facilitate a direct oxidative radical cyclization between beta-nitroalkenes and 1,3-cyclic diketones. 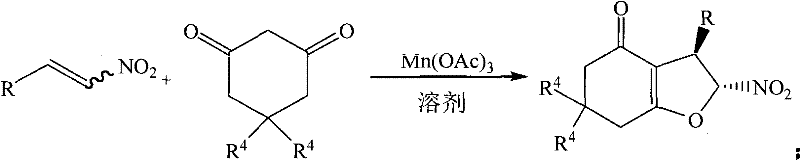 This innovative route operates efficiently in 95% aqueous ethanol, a solvent that is not only inexpensive and widely available but also significantly reduces the environmental footprint compared to anhydrous ethers. The reaction proceeds rapidly at mild temperatures ranging from 30°C to 60°C, typically completing within 0.5 to 2 hours, which drastically improves throughput capacity. By eliminating the need for pre-halogenated substrates, this process enhances atom economy, ensuring that nearly all atoms from the starting materials are incorporated into the final product. This shift not only simplifies the operational workflow but also aligns perfectly with modern demands for cost reduction in API manufacturing and sustainable chemical production.
This innovative route operates efficiently in 95% aqueous ethanol, a solvent that is not only inexpensive and widely available but also significantly reduces the environmental footprint compared to anhydrous ethers. The reaction proceeds rapidly at mild temperatures ranging from 30°C to 60°C, typically completing within 0.5 to 2 hours, which drastically improves throughput capacity. By eliminating the need for pre-halogenated substrates, this process enhances atom economy, ensuring that nearly all atoms from the starting materials are incorporated into the final product. This shift not only simplifies the operational workflow but also aligns perfectly with modern demands for cost reduction in API manufacturing and sustainable chemical production.
Mechanistic Insights into Mn(OAc)3-Promoted Radical Cyclization
The core of this technological advancement lies in the unique role of manganese(III) acetate as a single-electron oxidant. Unlike traditional base-mediated nucleophilic substitutions, this mechanism likely involves the generation of a radical species from the 1,3-dicarbonyl compound. The Mn(III) species abstracts an electron to form a carbon-centered radical at the active methylene position of the cyclic diketone. This nucleophilic radical then adds selectively to the electron-deficient double bond of the beta-nitroalkene. The presence of the nitro group is crucial here, as it activates the alkene towards radical addition while stabilizing the resulting intermediate radical through resonance. Subsequent intramolecular cyclization onto the carbonyl oxygen or further oxidation leads to the formation of the dihydrofuran ring. This radical pathway bypasses the need for leaving groups like chlorine or bromine, thereby avoiding the formation of corrosive acid byproducts and allowing for a much broader scope of substrates.
From an impurity control perspective, this mechanism offers superior selectivity. The patent data indicates excellent regioselectivity, where the cyclization occurs specifically to form the 2,3-dihydrofuran ring fused to the cyclohexanone without significant formation of isomeric byproducts. The mild reaction temperature of 50°C minimizes thermal degradation of sensitive functional groups, which is often a concern in high-temperature reflux conditions. Additionally, the use of ethanol as a protic solvent may assist in proton transfer steps during the cyclization, further driving the reaction to completion. For quality assurance teams, this means a cleaner crude reaction profile, reducing the burden on downstream purification processes like column chromatography and ensuring higher overall purity specifications for the final pharmaceutical intermediate.
How to Synthesize 2-Nitro-3-Aryl-2,3,5,7-Tetrahydrobenzofuran-4-One Efficiently
Implementing this synthesis requires careful attention to stoichiometry and solvent composition to maximize yield. The patent specifies that a molar ratio of beta-nitrostyrene to 1,3-cyclic diketone greater than 1:2 is effective, with a 1:2 ratio often providing optimal results. The promoter, manganese(III) acetate, is typically used in a 2-fold molar excess relative to the nitroalkene to ensure complete conversion. The choice of solvent is flexible, with 95% ethanol proving to be the most effective medium, balancing solubility and reaction rate. Detailed standardized operating procedures regarding mixing orders, heating rates, and workup protocols are essential for reproducibility.
- Dissolve beta-nitrostyrene derivative, manganese acetate promoter, and 1,3-cyclic diketone in 95% ethanol solvent.
- Heat the reaction mixture in a water bath at 30-60°C, preferably 50°C, and monitor progress via TLC.
- Upon completion (approx. 0.5-2 hours), purify the crude product using flash column chromatography with petroleum ether and acetone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this manganese-promoted protocol offers substantial strategic benefits beyond mere chemical efficiency. The elimination of specialized halogenated starting materials removes a significant vulnerability from the supply chain, as beta-nitrostyrenes are commodity chemicals available from numerous global suppliers. This diversification of raw material sources enhances supply security and mitigates the risk of price volatility associated with niche intermediates. Furthermore, the switch from expensive, anhydrous specialty solvents to common 95% ethanol drastically reduces raw material expenditure and simplifies solvent recovery and recycling operations. The reduction in reaction time from 24 hours to under 2 hours effectively increases reactor occupancy turnover, allowing existing manufacturing infrastructure to produce significantly higher volumes without capital investment in new equipment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of costly and hazardous halogenation steps. By utilizing direct radical cyclization, manufacturers avoid the expenses associated with synthesizing and handling 2-chloro- or 2-bromo-nitrostyrenes. Additionally, the use of aqueous ethanol eliminates the need for rigorous drying of solvents and reaction vessels, saving both energy and time. The simplified post-processing, which avoids the neutralization of large amounts of hydrogen chloride, further lowers waste disposal costs. These cumulative factors contribute to a leaner manufacturing cost structure, enabling more competitive pricing for high-purity pharmaceutical intermediates in the global market.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of robust, commercially available feedstocks. Beta-nitrostyrenes and dimedone (5,5-dimethyl-1,3-cyclohexanedione) are produced at scale globally, ensuring consistent availability. The mild reaction conditions reduce the risk of batch failures due to thermal runaway or sensitivity to trace moisture, which are common issues with anhydrous alkali metal reactions. This stability translates to predictable lead times and consistent delivery schedules for downstream clients. Moreover, the scalability of the process from gram to kilogram scales has been demonstrated in the patent examples, providing confidence for commercial scale-up of complex pharmaceutical intermediates without unforeseen technical hurdles.
- Scalability and Environmental Compliance: Environmental regulations are becoming increasingly stringent, and this process is well-positioned to meet them. The replacement of chlorinated or ether solvents with ethanol significantly reduces volatile organic compound (VOC) emissions and toxicity hazards. The absence of halogenated byproducts simplifies effluent treatment, reducing the load on wastewater processing facilities. The high atom economy of the reaction means less chemical waste is generated per unit of product. These factors not only ensure compliance with current environmental standards but also future-proof the manufacturing process against tighter regulations, making it a sustainable choice for long-term production of agrochemical and pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement teams assessing vendor capabilities. The answers are derived directly from the experimental data and claims within patent CN101628904B, ensuring accuracy and relevance for industrial application.
Q: What are the primary advantages of this Mn(OAc)3 method over traditional halogenated routes?
A: This method eliminates the need for difficult-to-prepare 2-halo-2-nitrostyrenes, uses cheaper and greener ethanol solvents instead of anhydrous ethers, and significantly reduces reaction time from 24 hours to under 2 hours while maintaining high yields.
Q: What is the substrate scope for this cyclization reaction?
A: The process is highly versatile, accommodating various beta-nitroalkenes including beta-nitrostyrenes with electron-donating or withdrawing groups, as well as heterocyclic variants like nitrofuran, nitrothiophene, and nitropyrrole derivatives.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of aqueous ethanol as a solvent, mild temperatures (30-60°C), and readily available starting materials makes this protocol highly amenable to scale-up for industrial pharmaceutical intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitro-3-Aryl-Tetrahydrobenzofuran-4-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our technical team has extensively analyzed the manganese-promoted cyclization technology described in CN101628904B and possesses the expertise to implement it seamlessly. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-nitro-3-aryl-2,3,5,7-tetrahydrobenzofuran-4-one derivatives meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced chemistry for your projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data for our catalog intermediates or to discuss route feasibility assessments for your custom synthesis needs. Let us help you optimize your supply chain with reliable, high-quality chemical solutions.
