Advanced Acid-Catalyzed Esterification for Commercial Scale Production of High-Purity Acetate Fragrances
The global demand for high-purity acetate fragrances, such as linalyl acetate and geranyl acetate, continues to surge within the fine chemicals and perfumery sectors, driving the need for more sustainable and efficient synthetic methodologies. Patent CN112142592B, published in early 2023, presents a transformative approach to alcohol esterification that addresses long-standing challenges in the industry, specifically regarding waste generation and substrate sensitivity. This technology utilizes enol acetates as acetylating agents under acidic catalysis, offering a distinct advantage over traditional acetic anhydride methods by producing neutral, volatile by-products that do not induce substrate rearrangement. For R&D directors and procurement specialists seeking a reliable acetate fragrance supplier, this innovation represents a significant leap forward in process chemistry, enabling the production of delicate terpene esters with exceptional selectivity and minimal environmental footprint. The method effectively bridges the gap between laboratory-scale precision and industrial-scale robustness, making it an ideal candidate for commercial scale-up of complex polymer additives and fragrance intermediates alike.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of acetate esters typically relies on the reaction of alcohols with acetic anhydride, catalyzed either by strong bases like DMAP or pyridine, or by protonic acids. While base-catalyzed methods often achieve high conversion rates, they suffer from a critical drawback: the generation of stoichiometric amounts of waste salts, such as sodium acetate or pyridinium salts, which require complex and costly disposal procedures. Furthermore, acid-catalyzed routes using acetic anhydride produce acetic acid as a by-product, which can accumulate in the reaction medium and promote undesirable side reactions like dehydration, isomerization, and rearrangement, particularly in sensitive allylic alcohols like linalool and geraniol. These side reactions not only lower the overall yield but also complicate the purification process, leading to increased production costs and reduced product quality. The accumulation of acidic by-products necessitates neutralization steps that further contribute to waste streams, creating a significant burden for cost reduction in flavor and fragrance manufacturing initiatives.
The Novel Approach
In stark contrast, the methodology described in CN112142592B employs enol acetates, such as vinyl acetate or isopropenyl acetate, as the acetyl donor in the presence of a specialized dual acid catalyst system. This innovative route fundamentally alters the reaction thermodynamics and kinetics by generating small, neutral molecules like acetaldehyde or acetone as by-products instead of corrosive acids or salts. These volatile by-products can be continuously removed from the reaction system via reactive distillation, effectively shifting the equilibrium towards the desired ester product without the need for excess reagents. This approach not only eliminates the formation of waste salts but also creates a mild reaction environment that preserves the structural integrity of sensitive terpene backbones. By upgrading from a batch neutralization process to a continuous reactive distillation setup, manufacturers can achieve superior process stability and control, directly addressing the pain points of reducing lead time for high-purity acetate intermediates while enhancing overall operational safety and environmental compliance.
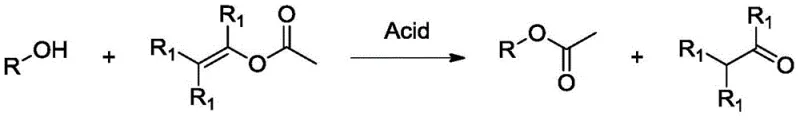
Mechanistic Insights into Acid-Catalyzed Enol Acetate Transesterification
The core of this technological breakthrough lies in the sophisticated interplay between the enol acetate reagent and the dual catalyst system, which typically comprises a protic acid (such as a dialkyl phosphate) and a Lewis acid (such as zinc chloride or lithium bromide). The protic acid functions primarily to activate the carbonyl group of the enol acetate through hydrogen bonding, facilitating the nucleophilic attack by the alcohol substrate. Simultaneously, the Lewis acid assists in the stabilization of the transition state and promotes the proton transfer steps required for the regeneration of the active catalytic species. This synergistic catalytic effect allows the reaction to proceed efficiently at moderate temperatures ranging from 60°C to 100°C, significantly lowering the energy input compared to traditional high-temperature batch processes. The mechanistic pathway avoids the formation of highly acidic intermediates that would otherwise trigger the Wagner-Meerwein rearrangements common in terpene chemistry, thereby ensuring that the final product retains the desired olfactory profile and chemical purity essential for high-purity OLED material and fragrance applications.
Furthermore, the integration of reactive distillation into the reaction design plays a pivotal role in the mechanistic success of this process. As the transesterification proceeds, the low-boiling by-products (acetaldehyde or acetone) are continuously vaporized and removed from the liquid phase, preventing them from participating in any reverse reactions or side interactions with the substrate. This continuous removal drives the reaction to near-complete conversion, often exceeding 99% for substrates like linalool and geraniol, as evidenced by the experimental data in the patent. The ability to operate under solvent-free conditions further simplifies the downstream processing, as there is no need to recover or dispose of large volumes of organic solvents. For supply chain managers, this mechanistic elegance translates into a streamlined workflow where the catalyst, having a much higher boiling point than the products, remains in the reactor pot and can be recycled multiple times without significant loss of activity, ensuring a consistent and reliable supply chain for critical fragrance ingredients.
How to Synthesize Linalyl Acetate Efficiently
The synthesis of high-value acetate esters like linalyl acetate using this patented method involves a carefully controlled sequence of mixing, heating, and distillation steps designed to maximize yield and catalyst longevity. The process begins with the charging of the alcohol substrate and the dual catalyst system into a reactor equipped with a fractionation column, followed by the controlled addition of the enol acetate reagent while maintaining specific temperature and pressure parameters to facilitate the continuous removal of by-products. Detailed standard operating procedures regarding exact molar ratios, reflux ratios, and specific temperature ramps are critical for reproducing the high selectivity reported in the patent literature. For a comprehensive guide on implementing this technology in a pilot or production plant, please refer to the standardized synthesis steps outlined below.
- Charge the reactor with alcohol substrate, protic acid catalyst (e.g., dialkyl phosphate), and Lewis acid catalyst (e.g., zinc chloride) under solvent-free conditions.
- Heat the mixture to 60-100°C and continuously feed enol acetate (e.g., vinyl acetate) while maintaining reactive distillation to remove volatile by-products like acetaldehyde.
- Upon completion, separate the product via vacuum distillation and recover the high-boiling acid catalyst from the bottom residue for reuse in subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this enol acetate-based esterification technology offers profound advantages for procurement and supply chain stakeholders, primarily driven by the elimination of waste treatment costs and the enhancement of process continuity. Traditional methods that rely on acetic anhydride and base catalysts generate substantial quantities of saline waste, which incurs significant disposal fees and regulatory compliance burdens. By switching to a method that produces neutral, volatile by-products, manufacturers can drastically simplify their waste management protocols, leading to substantial cost savings in environmental compliance and effluent treatment. Additionally, the solvent-free nature of the reaction reduces the volume of hazardous materials handled on-site, lowering insurance premiums and storage requirements, which collectively contribute to a more lean and agile manufacturing operation capable of responding quickly to market demands.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming from both raw material efficiency and operational simplifications. Since the catalyst system can be recycled and reused for multiple batches without significant degradation in performance, the recurring cost of catalyst procurement is minimized, directly impacting the bottom line. Moreover, the high selectivity of the reaction reduces the formation of difficult-to-separate impurities, which lowers the energy and time costs associated with final product purification and distillation. The avoidance of stoichiometric base usage also removes the cost associated with purchasing and handling large quantities of neutralizing agents, creating a more cost-effective production model that enhances competitiveness in the global fragrance market.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of this synthetic route, which relies on readily available and stable raw materials like vinyl acetate and simple phosphate esters. The continuous nature of the reactive distillation process allows for a steady output of product, reducing the risks associated with batch-to-batch variability and equipment downtime. This consistency ensures that downstream customers receive a uniform product quality, fostering stronger long-term partnerships and reducing the likelihood of supply disruptions. Furthermore, the mild reaction conditions reduce wear and tear on reactor vessels and ancillary equipment, extending the lifespan of capital assets and ensuring uninterrupted production schedules even during periods of high demand.
- Scalability and Environmental Compliance: Scaling this technology from pilot to commercial production is facilitated by the inherent simplicity of the unit operations involved, primarily requiring standard distillation columns and heated reactors. The process aligns perfectly with modern green chemistry principles by minimizing atom economy waste and eliminating the generation of persistent organic pollutants or heavy metal contaminants. This environmental stewardship not only future-proofs the manufacturing facility against tightening regulations but also enhances the brand value of the end products, appealing to eco-conscious consumers and corporate buyers who prioritize sustainability in their sourcing strategies. The ability to operate under reduced pressure also adds a layer of safety, mitigating the risks of thermal runaway and ensuring a safe working environment for personnel.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel esterification technology, derived directly from the detailed specifications and experimental results provided in the patent documentation. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines or for procurement officers assessing the quality standards of potential suppliers. These insights clarify how the specific choice of catalysts and reagents influences the final product profile and operational efficiency.
Q: Why is enol acetate preferred over acetic anhydride for sensitive terpene alcohols?
A: Enol acetates generate neutral, low-boiling by-products (acetaldehyde or acetone) instead of acetic acid. This prevents acid-catalyzed rearrangement and dehydration side reactions common in sensitive substrates like linalool, ensuring higher selectivity and purity without needing base neutralization.
Q: How does the reactive distillation process improve production efficiency?
A: By continuously removing the volatile ketone or aldehyde by-products from the reaction equilibrium, reactive distillation drives the esterification to completion. This eliminates the need for excess reagents and allows for a continuous operation mode, significantly enhancing space-time yield compared to batch processes.
Q: Can the acid catalyst system be recycled in this manufacturing process?
A: Yes, the dual catalyst system comprising phosphoric acid derivatives and Lewis acids has a high boiling point and remains in the reactor residue after product distillation. Patent data indicates the catalyst retains activity for over five cycles, drastically reducing catalyst consumption costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linalyl Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global fragrance and fine chemical industries. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the enol acetate esterification method are translated into reliable, high-volume supply chains. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of acetate fragrance we deliver meets the highest international standards for olfactory quality and chemical composition. Our capability to handle complex synthetic routes allows us to offer customized solutions that optimize both cost and performance for our partners.
We invite you to collaborate with us to explore how this cutting-edge esterification technology can enhance your product portfolio and streamline your supply chain operations. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate the tangible benefits of partnering with a leader in sustainable chemical manufacturing. Let us help you engineer a more efficient and profitable future for your fragrance formulations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
