Advanced Ru-PNN Catalysis for Commercial Scale-up of Complex Pharmaceutical Intermediates
Advanced Ru-PNN Catalysis for Commercial Scale-up of Complex Pharmaceutical Intermediates
The landscape of fine chemical manufacturing is continuously evolving, driven by the urgent need for more efficient and sustainable synthetic routes. Patent CN114080379A introduces a groundbreaking methodology for the hydrogenation of esters to their corresponding alcohols using molecular hydrogen in the presence of novel ruthenium complexes. This technology leverages tridentate PNN ligands that offer distinct advantages over conventional catalysts, addressing critical pain points in the production of high-value intermediates. By enabling reactions under moderate temperatures and pressures, this innovation provides a robust platform for generating essential building blocks used in pharmaceuticals and agrochemicals. The strategic implementation of these catalysts allows manufacturers to achieve superior conversion rates while maintaining exceptional selectivity profiles.
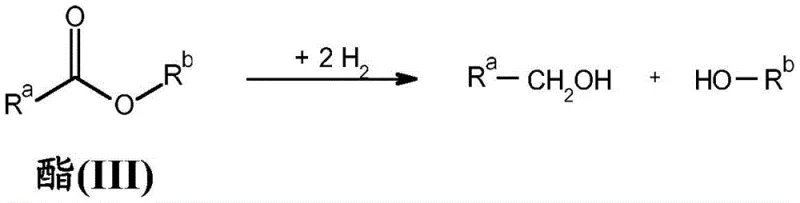
The transformation of esters into alcohols is a fundamental operation in organic synthesis, yet traditional methods often suffer from significant limitations regarding safety and efficiency. Conventional approaches frequently rely on stoichiometric reducing agents like lithium aluminum hydride, which generate substantial amounts of hazardous waste and require rigorous quenching procedures. Alternatively, heterogeneous hydrogenation may demand extreme conditions that compromise functional group tolerance, leading to complex purification challenges. The novel approach detailed in the patent utilizes homogeneous catalysis with specialized ruthenium complexes to overcome these barriers effectively. This shift represents a paradigm change towards greener chemistry practices that align with modern regulatory and environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of esters has been plagued by the use of catalysts with high molecular weights and complex ligand architectures that hinder operational efficiency. Prior art technologies, such as those employing triphos ligands, often necessitate high phosphorus-to-ruthenium molar ratios, which increases the cost of raw materials and complicates the removal of metal residues from the final product. Furthermore, many existing ligands are susceptible to oxidation, requiring stringent storage conditions and inert atmospheres that add logistical burdens to the supply chain. The synthesis of these traditional ligands is also notoriously laborious, involving multi-step sequences that reduce overall yield and increase the carbon footprint of the manufacturing process.
The Novel Approach
In contrast, the technology disclosed in CN114080379A utilizes tridentate PNN ligands that are structurally optimized for both performance and practicality. These ligands feature a lower molecular weight compared to their predecessors, which enhances the atom economy of the catalytic system and reduces the mass of waste generated per unit of product. The synthesis of these advanced ligands can be achieved through simple condensation reactions, often allowing for one-pot procedures that eliminate the need for intermediate isolation. This streamlined preparation not only lowers production costs but also accelerates the timeline from laboratory development to full-scale commercial implementation.
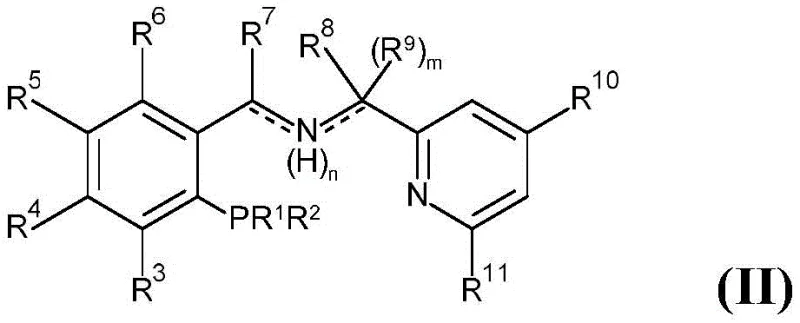
Mechanistic Insights into Ru-PNN Catalyzed Hydrogenation
The efficacy of this hydrogenation process is rooted in the unique electronic and steric properties of the tridentate PNN ligand coordinated to the ruthenium center. The ligand occupies three coordination sites, creating a stable environment that facilitates the activation of molecular hydrogen and the subsequent transfer to the ester substrate. This specific geometry prevents the decomposition of the active catalyst species, ensuring longevity and consistent turnover numbers throughout the reaction cycle. The presence of nitrogen and phosphorus donor atoms allows for fine-tuning of the electronic density at the metal center, which is crucial for activating the carbonyl bond of the ester without affecting other sensitive functional groups present in complex molecules.
Impurity control is another critical aspect where this mechanistic design excels, particularly for applications requiring high-purity pharmaceutical intermediates. The high selectivity of the Ru-PNN complex minimizes side reactions such as over-reduction or transesterification, which are common pitfalls in less sophisticated catalytic systems. By maintaining a well-defined coordination sphere, the catalyst directs the reaction pathway exclusively towards the formation of the desired alcohol product. This precision reduces the burden on downstream purification units, such as distillation columns or crystallization tanks, thereby lowering energy consumption and improving the overall yield of the manufacturing process.
How to Synthesize 1,4-Benzenedimethanol Efficiently
The practical application of this technology is exemplified by the hydrogenation of dimethyl terephthalate to 1,4-benzenedimethanol, a key intermediate in polymer and resin production. The protocol involves charging the reactor with the ester substrate, a ruthenium precursor, and the PNN ligand under a protective gas atmosphere to prevent premature oxidation. A base such as sodium methoxide is added to activate the catalytic species, followed by the introduction of molecular hydrogen at controlled pressures. The reaction mixture is then heated to the optimal temperature range, allowing the transformation to proceed to completion with minimal formation of byproducts.
- Prepare the catalytic system by reacting a Ru precursor complex with the tridentate PNN ligand in situ within the reaction vessel.
- Introduce the ester substrate and a suitable base such as sodium methoxide into the reactor under an inert protective gas atmosphere.
- Pressurize the system with molecular hydrogen and maintain temperatures between 50 to 200 degrees Celsius until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers tangible benefits that extend beyond mere technical performance. The simplified synthesis of the ligands means that the catalyst itself can be sourced more reliably, reducing the risk of supply disruptions caused by complex upstream manufacturing bottlenecks. Additionally, the robustness of the catalyst system allows for the use of standard industrial equipment, eliminating the need for costly capital investments in specialized high-pressure or cryogenic infrastructure. This compatibility with existing assets significantly lowers the barrier to entry for manufacturers looking to upgrade their production capabilities.
- Cost Reduction in Manufacturing: The lower molecular weight of the PNN ligands directly contributes to substantial cost savings by reducing the amount of precious metal required per batch. Since the ligands are easier to synthesize from readily available starting materials, the overall cost of goods sold for the catalyst system is significantly reduced compared to legacy technologies. Furthermore, the elimination of expensive activating solvents and the ability to recycle the catalyst in certain configurations enhance the economic viability of the process. These factors combine to create a more competitive cost structure for the final alcohol products.
- Enhanced Supply Chain Reliability: The stability of the novel ligands against oxidation ensures that inventory can be stored for extended periods without degradation, providing greater flexibility in procurement planning. This resilience reduces the dependency on just-in-time delivery models that are vulnerable to logistical delays. Moreover, the use of common solvents like toluene or alcohols simplifies the sourcing of raw materials, as these commodities are widely available in the global chemical market. This accessibility strengthens the supply chain against regional shortages or price volatility.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to stoichiometric reduction methods, aligning with increasingly strict environmental regulations. The ability to operate without specialized activating solvents simplifies waste treatment protocols and reduces the environmental footprint of the facility. Scalability is further supported by the compatibility of the reaction conditions with continuous flow processing, which offers potential for even greater efficiency gains. This forward-looking design ensures long-term compliance and operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation technology. They are derived from the specific beneficial effects and background technical problems outlined in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production lines. The answers provide clarity on performance metrics and operational requirements.
Q: What are the primary advantages of the new PNN ligands over traditional triphos ligands?
A: The novel tridentate PNN ligands described in patent CN114080379A offer significantly lower molecular weights compared to traditional triphos ligands, which reduces the overall mass of the catalyst complex. This structural efficiency translates to improved atom economy and simplifies the downstream disposal process, as there is less phosphorus-containing waste to manage. Furthermore, these ligands demonstrate superior chemical stability against oxidation, allowing for easier storage and handling without stringent protective measures.
Q: Can this hydrogenation process be scaled for industrial production of API intermediates?
A: Yes, the process is specifically designed for scalability, utilizing standard reaction apparatus such as stirred tank reactors or autoclaves suitable for gas-liquid reactions. The ability to form the active ruthenium complex in situ eliminates the need for isolating sensitive ligand intermediates, thereby streamlining the workflow for large-scale manufacturing. Operating conditions ranging from 0.1 to 20 MPa and temperatures up to 200 degrees Celsius are compatible with existing industrial infrastructure, facilitating the commercial scale-up of complex organic syntheses.
Q: How does this technology impact the purity profile of the resulting alcohols?
A: The homogeneous catalytic nature of the Ru-PNN complex ensures high selectivity towards the desired alcohol product, minimizing the formation of over-reduced byproducts or incomplete reduction intermediates. The robust coordination of the tridentate ligand to the ruthenium center stabilizes the active species, leading to consistent performance across various ester substrates including aromatic and aliphatic types. This high level of control is critical for producing high-purity pharmaceutical intermediates where strict impurity specifications must be met.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Benzenedimethanol Supplier
The technological potential of Ru-PNN catalyzed ester hydrogenation is immense, offering a pathway to produce high-value alcohols with unprecedented efficiency. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this advanced chemistry is seamless. Our facility is equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry. We are committed to delivering consistency and quality in every shipment.
We invite you to discuss how this innovative process can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us partner with you to drive efficiency and innovation in your chemical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
