Advanced Ruthenium Photosensitizers for Sustainable Industrial Wastewater Purification
Advanced Ruthenium Photosensitizers for Sustainable Industrial Wastewater Purification
The escalating crisis of industrial water pollution, particularly from textile dye effluents, demands innovative chemical solutions that transcend traditional remediation limitations. Patent CN115197127A introduces a breakthrough in this domain through the development of a novel carboxyl-substituted ruthenium-containing complex, specifically formulated as [RuII(tpy-ph(COO-)2)2](PF6)2. This advanced coordination compound functions as a highly efficient photosensitizer, designed to drive the photocatalytic degradation of persistent organic pollutants such as Malachite Green. By leveraging the unique photophysical properties of Ruthenium(II) polypyridyl systems, this technology offers a pathway to mineralize toxic dyes into harmless inorganic small molecules like carbon dioxide and water. For R&D directors and sustainability officers, this represents a critical shift from waste transfer methods to true waste elimination strategies, aligning industrial operations with increasingly stringent environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional wastewater treatment technologies, including adsorption, ion exchange, and membrane filtration, have long served as the backbone of industrial effluent management. However, these physical separation techniques suffer from a fundamental thermodynamic limitation: they merely transfer pollutants from the liquid phase to a solid phase without destroying them. This process generates significant volumes of secondary solid waste, such as spent activated carbon or saturated resin, which often contain concentrated toxins and require costly, energy-intensive disposal procedures like incineration or secure landfilling. Furthermore, membrane fouling and the high operational costs associated with regenerating ion-exchange resins create substantial economic burdens for manufacturing facilities. The inability to completely degrade carcinogenic and mutagenic dye structures means that the environmental risk is deferred rather than resolved, posing long-term liability issues for chemical producers and textile manufacturers alike.
The Novel Approach
In stark contrast, the ruthenium complex disclosed in CN115197127A employs a photocatalytic mechanism that fundamentally alters the fate of organic pollutants. Instead of sequestering dye molecules, the photosensitizer absorbs visible light energy to generate reactive species that oxidatively cleave the chromophoric structures of the dyes. This advanced oxidation process ensures the thorough decomposition of complex organic molecules into benign end products, effectively eliminating the generation of secondary solid waste streams. The introduction of carboxyl groups on the terpyridine ligand framework enhances the complex's stability and interaction within aqueous media, optimizing the efficiency of the degradation process. For procurement and supply chain leaders, this technological pivot translates to a drastic simplification of waste management logistics, removing the need for hazardous solid waste transport and disposal contracts while simultaneously reducing the overall time consumption associated with treatment cycles.
Mechanistic Insights into Ru(II)-Polypyridyl Photocatalysis
The efficacy of this system relies on the precise engineering of the coordination sphere around the central metal ion. The synthesis begins with the construction of a specialized ligand, 4'-(3,5-dicarboxyphenyl)-2,2':6',2''-terpyridine, which provides a rigid, tridentate binding environment for the Ruthenium(II) center. Upon irradiation with visible light, the complex undergoes a metal-to-ligand charge transfer (MLCT) excitation, promoting an electron from the metal d-orbitals to the pi-antibonding orbitals of the ligand. This excited state possesses a sufficiently long lifetime to facilitate electron transfer processes with dissolved oxygen or substrate molecules, generating reactive oxygen species such as superoxide radicals and hydroxyl radicals. These highly oxidative species are responsible for the non-selective attack on the conjugated double bonds of dye molecules, leading to their rapid decolorization and mineralization. The presence of electron-withdrawing carboxyl groups on the peripheral phenyl rings fine-tunes the redox potentials of the complex, ensuring thermodynamic feasibility for the degradation of recalcitrant pollutants.
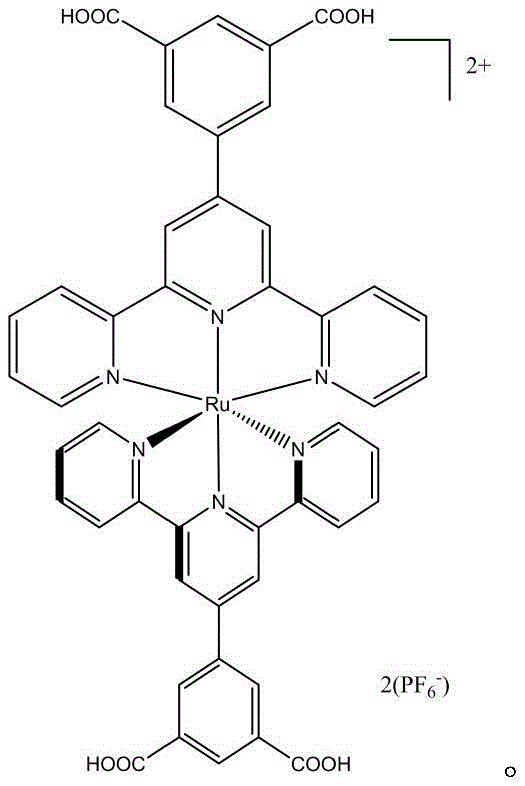
Beyond the primary photocatalytic cycle, the structural integrity of the ligand plays a pivotal role in impurity control and catalyst longevity. The steric bulk provided by the terpyridine framework protects the metal center from nucleophilic attack by solvent molecules or anions, which is a common deactivation pathway for simpler ruthenium complexes. During the synthesis, careful control of reaction conditions, such as the oxidation of methyl groups to carboxylic acids using concentrated nitric acid at elevated temperatures, ensures high regioselectivity and minimizes the formation of isomeric byproducts. The subsequent esterification and complexation steps are optimized to maximize yield while maintaining high purity, which is critical for consistent photocatalytic performance. For quality assurance teams, understanding these mechanistic nuances allows for the establishment of rigorous specification limits regarding ligand purity and metal content, ensuring that every batch of photosensitizer delivers predictable and reproducible degradation kinetics in real-world wastewater matrices.
How to Synthesize [RuII(tpy-ph(COO-)2)2](PF6)2 Efficiently
The preparation of this high-performance photosensitizer involves a convergent synthetic strategy that balances yield optimization with operational simplicity. The process initiates with an aldol condensation to build the carbon skeleton, followed by cyclization to form the heterocyclic core. Subsequent functional group transformations, including oxidation and esterification, prepare the ligand for metal coordination. The final assembly involves reacting the ligand precursor with ruthenium trichloride under inert atmosphere, followed by anion exchange to stabilize the cationic complex. This streamlined approach minimizes the number of isolation steps and utilizes commercially available starting materials, making it highly attractive for scale-up operations.
- Condense 3,5-dimethylbenzaldehyde with 2-acetylpyridine under basic conditions to form the chalcone intermediate.
- Cyclize the intermediate with additional acetylpyridine and ammonium hydroxide to generate the terpyridine ligand scaffold.
- Oxidize methyl groups to carboxylic acids using concentrated nitric acid, followed by esterification and final ruthenium coordination.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-based photocatalytic technology offers compelling economic and logistical advantages over legacy treatment methods. The primary value driver is the elimination of secondary waste disposal costs. By mineralizing dyes rather than capturing them, facilities can bypass the expensive logistics of handling, transporting, and treating hazardous solid sludge. This shift not only reduces direct expenditure on waste management vendors but also mitigates regulatory risks associated with hazardous waste storage and reporting. Furthermore, the synthesis route utilizes commodity chemicals such as 3,5-dimethylbenzaldehyde and acetylpyridine, which are readily available in the global market, ensuring a stable and resilient supply chain for the photosensitizer itself. The robustness of the synthetic protocol, which relies on standard unit operations like reflux and filtration, facilitates easy technology transfer to existing manufacturing infrastructure without requiring capital-intensive new equipment.
- Cost Reduction in Manufacturing: The economic benefits extend beyond waste disposal savings to include significant reductions in operational expenditures. Traditional methods often require frequent replacement of consumables like activated carbon or membranes, which represent a recurring cost burden. In contrast, the photocatalytic system utilizes a reusable or long-lived catalyst that drives the reaction using ambient or artificial light energy. The synthesis process itself is designed for high atom economy, particularly in the later stages where the complex is formed, minimizing raw material wastage. Additionally, the avoidance of high-pressure or cryogenic conditions in the synthesis reduces energy consumption during production. These factors collectively contribute to a lower total cost of ownership for the wastewater treatment process, enhancing the overall profitability of the manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain continuity is critical for uninterrupted industrial operations, and this technology supports that goal through原料 accessibility. The precursors required for the ligand synthesis are bulk petrochemical derivatives with established global supply networks, reducing the risk of shortages that often plague specialty reagents. The synthetic route avoids the use of exotic or controlled substances that might trigger complex import/export licensing requirements. Moreover, the final product is a stable solid salt that can be stored and transported under ambient conditions without special handling precautions, simplifying logistics and inventory management. This stability ensures that facilities can maintain adequate stock levels to buffer against any temporary disruptions in supply, thereby safeguarding production schedules and compliance timelines.
- Scalability and Environmental Compliance: From a scalability perspective, the synthesis is amenable to batch processing in standard glass-lined or stainless steel reactors, allowing for seamless transition from pilot scale to commercial tonnage production. The process generates minimal hazardous byproducts, and the solvent systems used, such as ethanol and water, are environmentally benign and easily recoverable. This aligns perfectly with green chemistry principles and helps facilities meet increasingly strict environmental discharge regulations. The ability to treat wastewater on-site with high efficiency reduces the volume of effluent requiring external processing, further lowering the environmental footprint. For organizations aiming to achieve sustainability certifications or carbon neutrality goals, implementing this photocatalytic technology serves as a tangible demonstration of commitment to environmental stewardship and circular economy practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium complex technology. These insights are derived directly from the patent specifications and are intended to clarify the operational parameters and benefits for potential adopters. Understanding these details is essential for conducting accurate feasibility studies and integrating the technology into existing process flows. Clear communication on these points ensures alignment between technical capabilities and business expectations.
Q: How does this ruthenium complex improve upon traditional wastewater treatment methods?
A: Unlike adsorption or membrane methods that generate secondary solid waste requiring disposal, this photosensitizer facilitates complete mineralization of organic dyes into harmless CO2 and H2O, eliminating secondary pollution risks.
Q: What are the key structural features enabling its photocatalytic activity?
A: The complex features a central Ruthenium(II) ion coordinated by carboxyl-substituted terpyridine ligands. The carboxyl groups enhance solubility and interaction with aqueous environments, while the Ru center provides excellent light-harvesting capabilities in the visible spectrum.
Q: Is the synthesis route scalable for industrial production?
A: Yes, the synthesis utilizes common starting materials like 3,5-dimethylbenzaldehyde and acetylpyridine. The steps involve standard unit operations such as reflux, filtration, and recrystallization, which are readily adaptable to large-scale reactor systems.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ruthenium Complex Supplier
As the demand for advanced environmental remediation technologies grows, partnering with an experienced chemical manufacturer is essential for successful implementation. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the supply of high-purity photosensitizers meets the rigorous demands of industrial applications. Our stringent purity specifications and rigorous QC labs guarantee that every batch of [RuII(tpy-ph(COO-)2)2](PF6)2 delivers consistent photocatalytic performance, minimizing variability in wastewater treatment outcomes. We understand the critical nature of supply continuity for environmental compliance and are committed to providing reliable support throughout the product lifecycle.
We invite technical procurement teams to contact us for a Customized Cost-Saving Analysis tailored to your specific wastewater composition and treatment capacity. Our experts are ready to provide specific COA data and route feasibility assessments to help you evaluate the integration of this photocatalytic solution into your operations. By collaborating with us, you gain access to not just a chemical product, but a comprehensive partnership focused on optimizing your environmental performance and reducing operational costs through cutting-edge chemistry.
