Advanced Manufacturing of Aripiprazole Intermediates via Novel Beckmann Rearrangement Technology
Introduction to Novel Aripiprazole Synthesis Technology
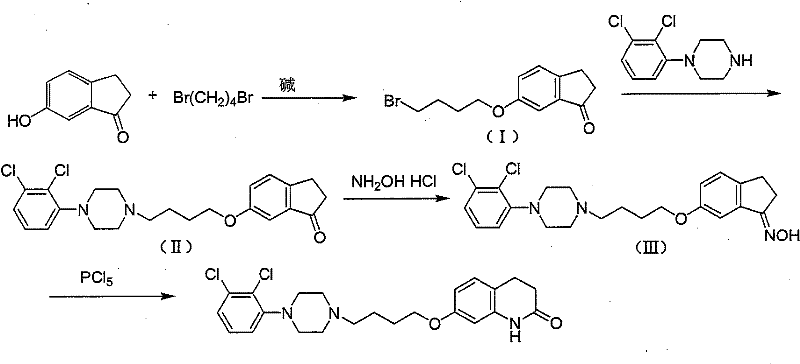
The pharmaceutical landscape for atypical antipsychotics continues to evolve, driven by the need for more efficient and environmentally sustainable manufacturing processes. Patent CN101323590B introduces a groundbreaking synthetic methodology for Aripiprazole, a critical active pharmaceutical ingredient used globally for treating schizophrenia and bipolar disorders. This technology fundamentally shifts the synthetic strategy away from traditional, harsh Friedel-Crafts alkylation towards a sophisticated sequence culminating in a Beckmann rearrangement. By utilizing 6-hydroxy-1-indanone as the foundational building block, this route achieves superior reaction kinetics and operational simplicity. The process is characterized by moderate reaction conditions, significantly reduced toxicity profiles, and high overall yields, making it exceptionally suitable for large-scale industrial production. For global supply chain stakeholders, this represents a pivotal advancement in securing reliable sources of high-quality psychiatric medication intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Aripiprazole has relied heavily on Friedel-Crafts alkylation strategies, such as those disclosed in earlier patents like CN1304373C. These conventional approaches typically involve the direct alkylation of a quinolone derivative, a process fraught with significant chemical engineering challenges. The primary drawback is the necessity for substantial quantities of aluminum chloride, a potent Lewis acid that generates massive amounts of hazardous waste upon quenching. Furthermore, these reactions often require toxic solvents such as chloroform, posing severe health risks to operators and complicating environmental compliance. The reaction conditions are frequently violent and difficult to control, leading to extended reaction times and cumbersome post-treatment procedures. These factors collectively inflate production costs and create bottlenecks in the supply chain, limiting the ability of manufacturers to respond agilely to market demand fluctuations.
The Novel Approach
In stark contrast, the methodology described in CN101323590B employs a convergent strategy that builds the molecular complexity gradually under mild conditions. The process initiates with the etherification of 6-hydroxy-1-indanone, followed by a catalyzed nucleophilic substitution with 1-(2,3-dichlorophenyl)piperazine. This is subsequently converted to an oxime intermediate, which then undergoes a Beckmann rearrangement to form the target lactam ring. This route eliminates the need for corrosive aluminum chloride and toxic chloroform entirely. Instead, it utilizes safer, recyclable solvents like ethanol and acetonitrile. The reaction speeds are notably fast, and the workup involves straightforward crystallization techniques rather than complex extractions or neutralizations. This paradigm shift not only enhances operator safety but also drastically simplifies the purification workflow, resulting in a more robust and economically viable manufacturing process for high-purity pharmaceutical intermediates.
Mechanistic Insights into Beckmann Rearrangement and Coupling
The core innovation of this synthetic pathway lies in the strategic application of the Beckmann rearrangement to construct the seven-membered lactam ring of the quinolone moiety. Unlike direct ring closure methods that struggle with regioselectivity, the oxime precursor allows for a controlled migration of the alkyl group under the influence of phosphorus pentachloride. The mechanism involves the activation of the oxime hydroxyl group by the phosphorus reagent, creating a good leaving group that facilitates the anti-periplanar migration of the carbon substituent. This intramolecular rearrangement occurs efficiently at temperatures between 0°C and 30°C, preserving the integrity of the sensitive dichlorophenyl-piperazine side chain. The mild thermal profile prevents thermal degradation and the formation of polymeric byproducts, which are common issues in high-temperature cyclizations. This precise control over the reaction trajectory ensures a clean conversion to the target scaffold with minimal impurity generation.
Furthermore, the preceding coupling steps are optimized for maximum efficiency through the use of phase transfer catalysts or halide salts like sodium iodide. In the second step, the displacement of the terminal bromide by the piperazine nitrogen is accelerated by the in situ generation of the more reactive iodide species. This catalytic effect lowers the activation energy of the nucleophilic substitution, allowing the reaction to proceed to completion rapidly in acetonitrile. The subsequent oximation step utilizes hydroxylamine hydrochloride buffered with sodium acetate in aqueous methanol, ensuring complete conversion of the ketone without affecting the ether linkage. The cumulative effect of these mechanistic optimizations is a highly linear and predictable synthesis where each intermediate can be isolated and purified to stringent specifications, providing multiple quality control checkpoints before the final API is generated.
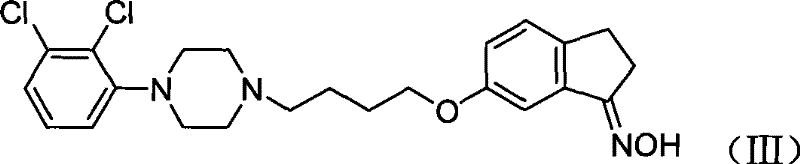
How to Synthesize 6-[4'-[4''-(2'',3''-dichlorophenyl)-1''-piperazidine]-butoxy]-1-indeneoxime Efficiently
The preparation of the key oxime intermediate is a critical juncture in this synthesis, as it sets the stage for the final ring-closing rearrangement. The process begins with the alkylation of 6-hydroxy-1-indanone using 1,4-dibromobutane in the presence of a base such as potassium carbonate in ethanol. Following isolation, this bromo-intermediate is reacted with 1-(2,3-dichlorophenyl)piperazine in acetonitrile with a catalytic amount of sodium iodide to yield the ketone precursor. Finally, the ketone is treated with hydroxylamine hydrochloride and sodium acetate trihydrate in a methanol-water mixture under reflux. Detailed standardized synthetic steps including specific stoichiometry, temperature profiles, and workup procedures are provided in the guide below.
- React 6-hydroxy-1-indanone with 1,4-dibromobutane in the presence of base to form the bromo-butoxy intermediate.
- Couple the bromo-intermediate with 1-(2,3-dichlorophenyl)piperazine using a catalyst like NaI to form the ketone precursor.
- Convert the ketone to the oxime intermediate (Formula III) using hydroxylamine hydrochloride and sodium acetate.
- Perform Beckmann rearrangement on the oxime using phosphorus pentachloride to cyclize and form the final Aripiprazole lactam structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers profound strategic benefits beyond mere chemical elegance. The elimination of hazardous reagents and solvents translates directly into reduced operational expenditures related to waste disposal and regulatory compliance. By avoiding the use of chloroform and massive quantities of aluminum salts, manufacturers can significantly lower their environmental footprint and mitigate the risks associated with handling corrosive materials. This aligns perfectly with modern green chemistry initiatives and reduces the liability exposure for production facilities. Moreover, the use of common, commodity-grade solvents like ethanol and acetonitrile ensures that raw material sourcing remains stable and cost-effective, insulating the supply chain from volatility in specialty chemical markets.
- Cost Reduction in Manufacturing: The streamlined nature of this process eliminates several costly unit operations found in traditional methods. The absence of complex neutralization steps required for aluminum chloride waste removes a significant burden on utility consumption and labor hours. Additionally, the high yields reported in the patent examples suggest a more efficient utilization of starting materials, reducing the cost of goods sold per kilogram of finished intermediate. The ability to recycle solvents further enhances the economic viability of the process, driving down the overall manufacturing cost without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 6-hydroxy-1-indanone and 1,4-dibromobutane ensures a robust supply base. These commodities are produced by multiple global suppliers, reducing the risk of single-source dependency. The mild reaction conditions also imply that the process can be scaled up in standard glass-lined or stainless steel reactors without requiring specialized high-pressure or high-temperature equipment. This flexibility allows for rapid capacity expansion to meet surging market demand, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The simplicity of the workup procedures, primarily involving filtration and crystallization, makes this process highly amenable to continuous manufacturing or large-batch production. The reduced generation of hazardous waste simplifies the permitting process for new production lines and facilitates easier adherence to increasingly strict environmental regulations. This scalability ensures that the technology can support commercial volumes ranging from pilot plant scales to multi-ton annual production, providing a future-proof solution for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Beckmann rearrangement-based synthesis. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical aspects of adopting this technology for commercial production.
Q: What are the primary advantages of this Beckmann rearrangement route over traditional Friedel-Crafts methods?
A: This method avoids the use of large quantities of aluminum chloride and toxic chloroform solvents required in traditional Friedel-Crafts alkylation. It operates under milder conditions with faster reaction times and simplified workup procedures, significantly reducing environmental impact and processing complexity.
Q: Is the intermediate 6-[4'-[4''-(2'',3''-dichlorophenyl)-1''-piperazidine]-butoxy]-1-indeneoxime stable for storage?
A: Yes, the key intermediate (Formula III) is a stable solid with a melting point of 173-174°C. Its stability allows for flexible production scheduling and quality control testing before the final rearrangement step.
Q: How does this process impact the purity profile of the final API?
A: By utilizing specific crystallization steps and avoiding harsh Lewis acid catalysts that often generate complex impurity profiles, this route yields high-purity Aripiprazole. The use of ethanol and acetonitrile facilitates easier solvent removal and recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aripiprazole Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly analyzed the potential of the Beckmann rearrangement route described in CN101323590B and is fully prepared to implement this technology at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this innovative route can optimize your supply chain. By leveraging our expertise, you can achieve significant process improvements and secure a stable source of high-quality materials. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process and drive your project forward with confidence.
