Revolutionizing Quinoline Synthesis: Enzymatic Catalysis for High-Purity Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for sustainable and efficient manufacturing processes. Patent CN102586356B introduces a groundbreaking methodology for the synthesis of quinoline heterocycle derivatives, specifically leveraging enzyme catalysis to overcome the limitations of traditional thermal and chemical catalysis. This innovation focuses on the condensation of dibromo-o-aminobenzaldehyde (Formula IV) with various cyclic ketones (Formulas V, VI, and VII) to produce structurally diverse quinoline scaffolds essential for pharmaceutical applications. By replacing corrosive acid or base catalysts with biocompatible enzymes such as lipases and proteases, this technology achieves remarkable reaction efficiency under ambient conditions. The strategic shift towards biocatalysis not only aligns with green chemistry principles but also offers a robust pathway for producing high-purity intermediates required in complex drug synthesis. For R&D directors and process chemists, this patent represents a critical opportunity to modernize synthetic routes, ensuring that the production of nitrogen-containing heterocycles meets the rigorous standards of modern regulatory environments while minimizing environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline derivatives has relied heavily on classical named reactions such as the Skraup, Combes, and traditional Friedlander syntheses, which invariably demand harsh reaction conditions to drive the cyclization forward. These conventional protocols typically necessitate the use of strong mineral acids like sulfuric acid or potent Lewis acids, often coupled with elevated temperatures that can exceed 100°C to achieve acceptable conversion rates. Such aggressive environments frequently lead to the degradation of sensitive functional groups, resulting in complex impurity profiles that are difficult and costly to remove during downstream purification. Furthermore, the reliance on stoichiometric amounts of acidic or basic reagents generates substantial quantities of inorganic salt waste during the neutralization phase, creating a significant burden on wastewater treatment facilities and increasing the overall carbon footprint of the manufacturing process. The operational hazards associated with handling corrosive reagents at high temperatures also pose safety risks to personnel and require specialized equipment capable of withstanding extreme chemical stress, thereby inflating capital expenditure for production facilities.
The Novel Approach
In stark contrast to these legacy methods, the enzymatic approach detailed in the patent utilizes biological catalysts to facilitate the Friedlander-type condensation under exceptionally mild and controlled conditions. By employing enzymes such as pig pancreas lipase or porcine pepsin, the reaction can proceed efficiently at room temperature, typically around 25°C, eliminating the need for energy-intensive heating cycles. This biological catalysis mechanism allows for high chemoselectivity, preserving the integrity of the dibromo-substituted aromatic ring and preventing unwanted side reactions that commonly plague thermal methods. The process utilizes common organic solvents like ethanol, which are not only cost-effective but also easier to recover and recycle compared to high-boiling polar aprotic solvents often used in traditional synthesis. The result is a streamlined workflow where the reaction mixture can be worked up simply by filtration and extraction, bypassing the cumbersome neutralization and salt-removal steps entirely. This paradigm shift enables the production of quinoline derivatives with superior purity profiles while drastically reducing the environmental load associated with chemical manufacturing.
Mechanistic Insights into Enzyme-Catalyzed Cyclization
The core of this technological advancement lies in the unique ability of hydrolytic enzymes, traditionally known for ester bond cleavage, to promote carbon-carbon and carbon-nitrogen bond formation in non-aqueous or low-water media. In this specific transformation, the enzyme active site likely acts as a micro-environment that stabilizes the transition state of the condensation between the amino group of the aldehyde and the carbonyl group of the cyclic ketone. The enzyme facilitates the dehydration step crucial for aromatization of the quinoline ring without the need for external dehydrating agents or strong acids. This biocatalytic pathway effectively lowers the activation energy barrier for the cyclization, allowing the reaction to reach completion within a practical timeframe of 6 to 18 hours at ambient temperature. The specificity of the enzyme ensures that the reaction proceeds with high regioselectivity, particularly important when dealing with substituted substrates where multiple reactive sites might exist. Understanding this mechanism is vital for process optimization, as it highlights the importance of maintaining the structural integrity of the enzyme through careful control of solvent polarity and temperature.
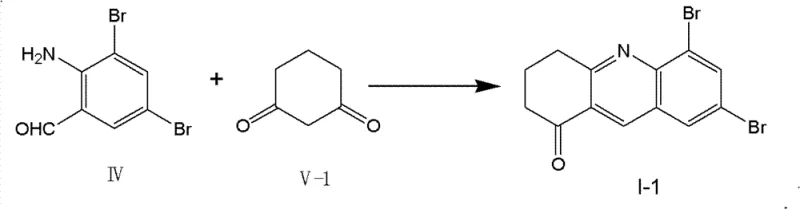
Furthermore, the impurity control mechanism inherent in this enzymatic process is a significant advantage for pharmaceutical intermediate manufacturing. Traditional acid-catalyzed routes often suffer from polymerization of the aldehyde starting material or over-reaction of the ketone, leading to tarry byproducts that complicate isolation. The mild pH environment maintained by the enzymatic system prevents acid-catalyzed decomposition of the sensitive o-aminobenzaldehyde moiety, which is prone to oxidation and self-condensation under harsh conditions. Consequently, the crude reaction mixture obtained from the enzymatic process is significantly cleaner, as evidenced by the high isolated yields reported in the patent embodiments, ranging from 60% to 95% depending on the specific ketone substrate. This reduction in side-product formation translates directly to reduced solvent consumption during chromatographic purification and higher overall throughput for the manufacturing line. For quality control teams, this means a more consistent product specification with fewer unknown impurities, facilitating faster regulatory approval for downstream drug candidates.
How to Synthesize Quinoline Derivatives Efficiently
The practical implementation of this enzymatic synthesis route is designed to be straightforward and adaptable to standard laboratory and pilot-scale equipment without requiring specialized high-pressure reactors. The protocol begins with the precise weighing of dibromo-o-aminobenzaldehyde and the chosen cyclic ketone substrate, maintaining a molar ratio of approximately 1:2 to ensure complete consumption of the limiting aldehyde reagent. These solids are suspended in a green solvent such as ethanol, which serves both as the reaction medium and a compatible environment for the enzyme's activity. The biocatalyst, typically added as a powder, is introduced at a loading of 10 to 15 mg per mmol of substrate, initiating the cascade upon gentle agitation. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are outlined in the guide below to ensure reproducibility and scalability for industrial partners.
- Prepare the reaction mixture by combining dibromo-o-aminobenzaldehyde (Formula IV) and the selected cyclic ketone compound (Formula V, VI, or VII) in an organic solvent such as ethanol.
- Add the biocatalyst, preferably pig pancreas lipase, at a loading of 10 to 15 mg per mmol of aldehyde substrate to initiate the condensation.
- Maintain the reaction at room temperature (approximately 25°C) with shaking at 150 rpm for 6 to 18 hours until TLC monitoring indicates completion.
- Upon completion, add water and ethyl acetate to the mixture, stir, filter, separate the organic phase, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this enzymatic technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of hazardous corrosive reagents fundamentally alters the cost structure of the manufacturing process by removing the need for expensive corrosion-resistant reactor linings and specialized safety infrastructure. Additionally, the use of commercially available enzymes, which are produced at scale for various industries, ensures a stable and reliable supply of the catalyst without the volatility associated with specialty chemical reagents. The simplified workup procedure, which avoids aqueous acid-base extractions, significantly reduces the volume of wastewater generated, leading to substantial cost savings in waste disposal and environmental compliance fees. These factors combine to create a more resilient supply chain that is less susceptible to regulatory crackdowns on chemical emissions and more capable of meeting the stringent sustainability criteria increasingly demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The transition to enzyme catalysis removes the dependency on stoichiometric amounts of strong acids or bases, which not only reduces raw material costs but also eliminates the downstream costs associated with neutralizing and disposing of large volumes of saline waste. By operating at room temperature, the process drastically cuts energy consumption related to heating and cooling cycles, contributing to a lower overall utility bill for the production facility. The high selectivity of the enzyme minimizes the loss of valuable starting materials to side reactions, thereby improving the atom economy and reducing the cost per kilogram of the final active pharmaceutical ingredient intermediate. Furthermore, the simplified purification process requires less silica gel and solvent for chromatography, directly lowering the consumable expenses for the manufacturing team.
- Enhanced Supply Chain Reliability: Sourcing high-purity quinoline intermediates is often bottlenecked by the limited number of suppliers capable of managing the hazards of traditional synthesis; this enzymatic route broadens the potential supplier base by lowering the technical barrier to entry. The stability of the enzyme catalysts allows for easier storage and transportation compared to moisture-sensitive Lewis acids or volatile liquid acids, reducing the risk of supply disruptions due to shipping restrictions. The robustness of the reaction conditions means that production can be maintained consistently even with minor fluctuations in ambient plant conditions, ensuring on-time delivery for critical project milestones. This reliability is crucial for pharmaceutical companies managing tight development timelines where delays in intermediate supply can stall entire drug discovery programs.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the absence of exothermic spikes typically seen in acid-catalyzed condensations, allowing for safer operation in larger reactors without complex cooling jackets. The green nature of the process, utilizing ethanol and biodegradable enzymes, aligns perfectly with modern environmental regulations, future-proofing the manufacturing site against tightening emission standards. The reduction in hazardous waste generation simplifies the permitting process for new production lines, accelerating the time-to-market for new products. Moreover, the compatibility of this method with continuous flow chemistry setups offers a clear pathway for further intensification and efficiency gains as demand grows.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this enzymatic synthesis technology, derived directly from the experimental data and specifications within the patent documentation. These insights are intended to clarify the feasibility of adopting this route for commercial production and to assist technical teams in evaluating its fit within their existing process portfolios. Understanding these details is essential for making informed decisions about process transfer and scale-up strategies.
Q: What are the optimal reaction conditions for this enzymatic synthesis?
A: The patent specifies that the reaction proceeds optimally at room temperature (15°C to 50°C, preferably 25°C) in solvents like ethanol or DMSO. The molar ratio of aldehyde to ketone is ideally 1:2, with an enzyme loading of 10-15 mg/mmol.
Q: Which enzymes are suitable for catalyzing this Friedlander-type cyclization?
A: The technology supports a range of commercially available enzymes including pig pancreas lipase, porcine pepsin, bovine albumin, and trypsin. Pig pancreas lipase is identified as the most effective catalyst, achieving conversion efficiencies up to 100% in specific embodiments.
Q: How does this method compare to traditional acid-base catalyzed routes?
A: Unlike traditional methods requiring harsh acids or bases and high temperatures, this enzymatic approach operates under mild, neutral conditions. This eliminates the need for extensive neutralization steps, reduces three-waste discharge, and simplifies the purification process while maintaining high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of enzymatic catalysis in the production of complex heterocyclic intermediates and are fully equipped to leverage this technology for our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of quinoline derivative meets the exacting standards required for pharmaceutical applications. Our commitment to green chemistry aligns with the principles of this patent, allowing us to offer a sustainable supply solution that reduces the environmental footprint of your supply chain while maintaining cost competitiveness.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project requirements. By partnering with our technical procurement team, you can access specific COA data and comprehensive route feasibility assessments that demonstrate the tangible benefits of switching to this enzymatic platform. Whether you require custom synthesis of novel analogs or reliable tonnage supply of established intermediates, our infrastructure is designed to support your growth with flexibility and precision. Contact us today to discuss how we can optimize your quinoline supply chain through innovative catalytic technologies.
