Advanced Synthesis of Potent Kinase Inhibitors for Commercial Pharmaceutical Production
Advanced Synthesis of Potent Kinase Inhibitors for Commercial Pharmaceutical Production
The pharmaceutical landscape is continuously evolving with the demand for highly selective kinase inhibitors to treat complex hematological malignancies and autoimmune disorders. Patent CN103073508A introduces a sophisticated class of (aminophenylamino)pyrimidinyl benzamide compounds that function as potent inhibitors of Bruton's tyrosine kinase (Btk). This technology represents a significant advancement in the field of targeted therapy, offering a robust chemical framework for developing next-generation therapeutics. The disclosed molecular architecture allows for extensive structural modulation, enabling researchers to fine-tune pharmacokinetic properties while maintaining high binding affinity. For procurement and supply chain leaders, understanding the underlying chemistry of these pharmaceutical intermediates is crucial for securing reliable sources and ensuring continuity in drug development pipelines.
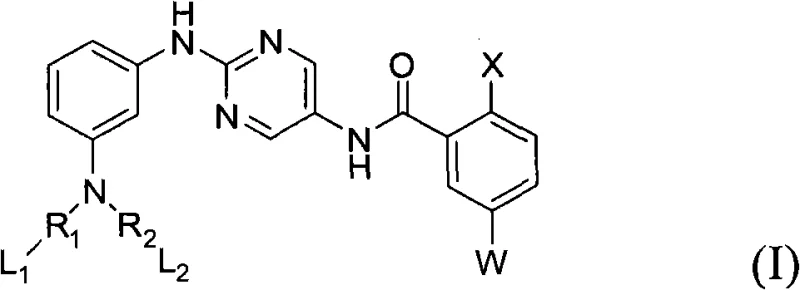
The core structure defined in the patent features a central pyrimidine ring linked to substituted phenyl groups via amino and amide bonds. This specific arrangement is critical for interacting with the ATP-binding pocket of the Btk enzyme. The versatility of the substituents, denoted as W, X, R1, and R2 in the general formula, provides a vast chemical space for optimization. From a manufacturing perspective, the ability to introduce diverse functional groups such as trifluoromethyl, methyl, or various cyclic amines allows for the creation of a library of analogs. This modularity is essential for overcoming resistance mechanisms often encountered in long-term cancer treatments. As a reliable pharmaceutical intermediates supplier, leveraging such flexible synthetic platforms ensures that clients can access a wide range of candidates for their preclinical and clinical programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for kinase inhibitors often suffer from low overall yields, harsh reaction conditions, and the use of expensive transition metal catalysts that are difficult to remove. Many legacy routes rely on multiple protection and deprotection steps that add significant time and cost to the manufacturing process. Furthermore, conventional methods may struggle with regioselectivity, leading to complex mixtures of isomers that require extensive and wasteful purification efforts. The presence of residual heavy metals is a particular concern for high-purity pharmaceutical intermediates, as regulatory bodies impose strict limits on elemental impurities. These challenges frequently result in extended lead times and increased production costs, creating bottlenecks in the supply chain for critical oncology drugs.
The Novel Approach
The synthetic strategy outlined in CN103073508A addresses these inefficiencies through a streamlined, convergent approach. By utilizing readily available starting materials such as m-phenylenediamine and substituted benzoic acids, the process minimizes the reliance on exotic reagents. The route employs standard organic transformations like nucleophilic aromatic substitution and amide coupling, which are well-understood and easily controlled on a large scale. A key advantage is the use of catalytic hydrogenation for nitro reduction, which is cleaner and more atom-economical than stoichiometric metal reductions. This novel methodology not only improves the overall yield but also simplifies the workup procedures, thereby reducing solvent consumption and waste generation. Such improvements are vital for achieving cost reduction in pharmaceutical intermediates manufacturing while adhering to green chemistry principles.
Mechanistic Insights into Amide Coupling and Nucleophilic Substitution
The construction of the target molecule relies heavily on two fundamental reactions: nucleophilic aromatic substitution (SnAr) and amide bond formation. The initial coupling of the protected diamine with 2-chloro-5-nitropyrimidine proceeds via an SnAr mechanism, where the electron-deficient pyrimidine ring facilitates the displacement of the chloride leaving group by the amine nucleophile. This step is critical for establishing the core heterocyclic linkage. Subsequent transformation of the nitro group to an amine via palladium-catalyzed hydrogenation activates the scaffold for further functionalization. The efficiency of this reduction step, reported with quantitative yields in the patent examples, underscores the robustness of the chosen catalytic system.
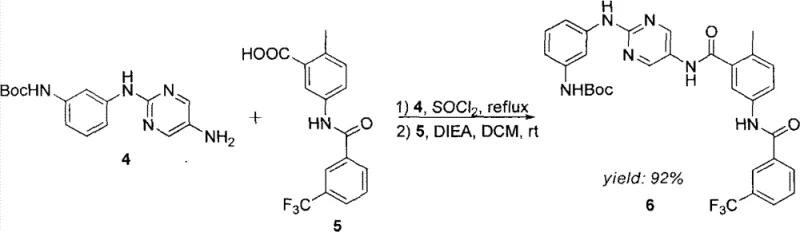
The formation of the benzamide linkage is another pivotal moment in the synthesis. As illustrated in the reaction schemes, the carboxylic acid component is typically activated, often via conversion to an acid chloride or using coupling reagents, before reacting with the aniline derivative. This acylation step must be carefully controlled to prevent over-reaction or hydrolysis. The patent demonstrates that using bases like diisopropylethylamine (DIEA) in dichloromethane provides an optimal environment for this transformation. The final installation of the acryloyl group via acylation of the terminal amine completes the pharmacophore. Understanding these mechanistic details allows process chemists to identify critical control points, ensuring consistent quality and minimizing the formation of genotoxic impurities often associated with alkylating agents like acryloyl chloride.
How to Synthesize Potent Btk Inhibitor Intermediates Efficiently
The synthesis of these complex molecules requires precise control over reaction parameters to maximize yield and purity. The patented process offers a clear roadmap, beginning with the selective mono-protection of diamines to direct regioselectivity. Following the assembly of the pyrimidine core, the sequence involves reduction, coupling, deprotection, and final functionalization. Each step has been optimized to use commercially available reagents and standard laboratory equipment, making the transfer to pilot and production scales straightforward. For detailed operational parameters, stoichiometry, and specific workup instructions, refer to the standardized guide below which encapsulates the critical process knowledge.
- Initial protection of m-phenylenediamine followed by nucleophilic substitution with 2-chloro-5-nitropyrimidine to form the core heterocyclic scaffold.
- Catalytic hydrogenation of the nitro group to an amine, followed by activation of benzoic acid derivatives and subsequent amide coupling.
- Final deprotection of the Boc group and acrylation using acryloyl chloride to yield the active kinase inhibitor compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the economic implications of this synthetic route are profound. The use of commodity chemicals and the avoidance of precious metal catalysts in the final steps significantly lower the raw material costs. Moreover, the high yields reported in the patent examples, particularly the quantitative reduction step and the high-yielding coupling reactions, translate directly into better material throughput. This efficiency reduces the amount of starting material required per kilogram of final product, effectively lowering the cost of goods sold (COGS). Additionally, the simplified purification protocols reduce the consumption of chromatography media and solvents, further contributing to substantial cost savings in the manufacturing budget.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive transition metal cross-coupling reactions often found in similar kinase inhibitor syntheses. By relying on nucleophilic substitution and standard amide coupling, the process avoids the costs associated with palladium or copper catalysts and the subsequent rigorous purification needed to remove metal residues. This simplification leads to a more economical process that is less sensitive to fluctuations in the price of precious metals.
- Enhanced Supply Chain Reliability: The starting materials, such as substituted benzoic acids and phenylenediamines, are widely available from multiple global suppliers. This diversity in the supply base mitigates the risk of shortages that can occur with specialized or proprietary reagents. Furthermore, the stability of the intermediates allows for strategic stockpiling, ensuring that production schedules can be maintained even during periods of raw material volatility. This reliability is crucial for maintaining the commercial scale-up of complex pharmaceutical intermediates.
- Scalability and Environmental Compliance: The reactions are conducted in common organic solvents like dichloromethane, tetrahydrofuran, and methanol, which have established recovery and recycling infrastructure in most chemical plants. The absence of hazardous reagents in the final steps simplifies waste treatment and disposal. The process is inherently safer and more environmentally friendly, aligning with modern regulatory standards for sustainable manufacturing. This ease of scale-up ensures that reducing lead time for high-purity pharmaceutical intermediates is achievable without compromising on safety or environmental compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these kinase inhibitor compounds. The answers are derived from the technical specifications and experimental data provided in the patent documentation, offering clarity on the feasibility and scope of the technology.
Q: What is the primary therapeutic target of these pyrimidinyl benzamide compounds?
A: These compounds are designed to inhibit Bruton's tyrosine kinase (Btk), a critical enzyme in B-cell receptor signaling, making them effective for treating B-cell malignancies and autoimmune diseases.
Q: How does the synthetic route ensure high purity for pharmaceutical use?
A: The process utilizes robust purification techniques such as silica gel column chromatography and recrystallization at multiple stages, ensuring the removal of impurities and isomers to meet stringent API intermediate standards.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the reaction conditions utilize common solvents like DCM, THF, and methanol, and avoid exotic catalysts, facilitating straightforward scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Btk Inhibitor Intermediate Supplier
The development of effective Btk inhibitors requires a partner who understands both the intricate chemistry and the rigorous demands of the pharmaceutical industry. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from gram-scale research to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for clinical trial materials. We are committed to delivering high-quality intermediates that accelerate your drug discovery timelines.
We invite you to discuss your specific requirements with our technical team. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can benefit your bottom line. Contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your project needs. Let us be your strategic partner in bringing these life-saving therapies to market.
