Advanced Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazoles, which serve as critical scaffolds in numerous high-value active pharmaceutical ingredients (APIs). As illustrated in the structural diversity of modern medicines, compounds such as Maraviroc, Sitagliptin, and Deferasirox rely heavily on the unique physicochemical properties imparted by the triazole ring. 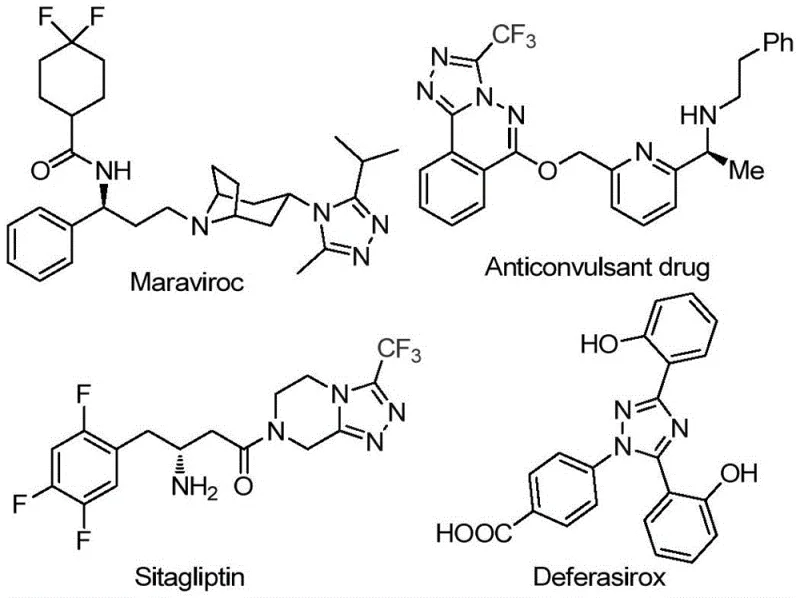 . Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds that addresses long-standing challenges in introducing both trifluoromethyl and acyl groups simultaneously. This innovation represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, offering a pathway that bypasses the limitations of traditional metal-catalyzed couplings. By leveraging a tandem iodine-promoted oxidation and cyclization strategy, this technology enables the efficient construction of complex heterocyclic cores essential for next-generation therapeutics.
. Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds that addresses long-standing challenges in introducing both trifluoromethyl and acyl groups simultaneously. This innovation represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, offering a pathway that bypasses the limitations of traditional metal-catalyzed couplings. By leveraging a tandem iodine-promoted oxidation and cyclization strategy, this technology enables the efficient construction of complex heterocyclic cores essential for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, especially those bearing electron-withdrawing trifluoromethyl groups alongside acyl functionalities, has been fraught with synthetic inefficiencies. Conventional methodologies often rely on precious transition metal catalysts, which not only inflate the raw material costs but also introduce severe complications regarding residual metal limits in final drug substances. Furthermore, many existing protocols demand rigorous anhydrous and oxygen-free conditions, necessitating specialized equipment and inert atmosphere handling that drastically increases operational complexity and safety risks. The substrate scope in traditional methods is frequently narrow, failing to tolerate diverse functional groups on the aromatic rings, which limits the ability of medicinal chemists to explore structure-activity relationships (SAR) effectively. Additionally, multi-step sequences required to install the trifluoromethyl moiety separately often result in poor overall yields and generate substantial chemical waste, undermining the sustainability goals of modern green chemistry initiatives in API manufacturing.
The Novel Approach
In stark contrast, the methodology described in patent CN113105402B introduces a streamlined, metal-free protocol that utilizes inexpensive elemental iodine and dimethyl sulfoxide (DMSO) to drive the reaction forward. This novel approach capitalizes on the dual role of iodine as both an oxidant and a promoter, facilitating a Kornblum oxidation of aryl ethyl ketones to generate reactive aryl diketone intermediates in situ. These intermediates then undergo a seamless condensation and cyclization with trifluoroethylimide hydrazides to forge the target triazole ring in a single pot. 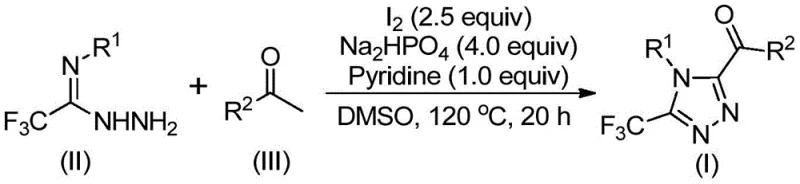 . This telescoped process eliminates the need for isolating unstable intermediates, thereby reducing solvent consumption and processing time. The reaction proceeds under ambient atmospheric conditions without the need for gloveboxes or Schlenk lines, making it exceptionally accessible for both laboratory optimization and industrial scale-up. The broad compatibility with various substituents on both the hydrazide and ketone components ensures high versatility for generating diverse chemical libraries.
. This telescoped process eliminates the need for isolating unstable intermediates, thereby reducing solvent consumption and processing time. The reaction proceeds under ambient atmospheric conditions without the need for gloveboxes or Schlenk lines, making it exceptionally accessible for both laboratory optimization and industrial scale-up. The broad compatibility with various substituents on both the hydrazide and ketone components ensures high versatility for generating diverse chemical libraries.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated example of cascade reactivity driven by the unique redox properties of the iodine-DMSO system. Initially, the aryl ethyl ketone undergoes alpha-iodination followed by Kornblum oxidation, where DMSO acts as the oxygen source to convert the methyl ketone into an alpha-dicarbonyl species. This highly electrophilic diketone intermediate is then trapped by the nucleophilic nitrogen of the trifluoroethylimide hydrazide, forming a hydrazone linkage through dehydration. Subsequently, the remaining iodine species, in conjunction with the base (sodium dihydrogen phosphate and pyridine), promotes an intramolecular oxidative cyclization. This step involves the formation of the N-N bond and the aromatization of the triazole ring, locking the trifluoromethyl group at the 3-position and the acyl group at the 5-position. The precise control of reaction temperature, typically ramping from 90-110°C for oxidation to 110-130°C for cyclization, is critical to maximizing conversion while minimizing side reactions such as over-oxidation or polymerization.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals removes the risk of metal-ligand complex formation, which can be notoriously difficult to purge during downstream processing. The use of simple inorganic bases and organic solvents ensures that the impurity profile is dominated by organic byproducts that are generally easier to separate via standard chromatographic techniques or crystallization. Furthermore, the mild nature of the iodine promotion prevents the degradation of sensitive functional groups such as halogens or ethers, which might otherwise undergo dehalogenation or cleavage under harsher catalytic conditions. This high level of chemoselectivity ensures that the final product maintains high purity, a prerequisite for meeting the stringent quality standards required for high-purity pharmaceutical intermediates intended for clinical use.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazoles Efficiently
The practical execution of this synthesis is designed for operational simplicity, allowing chemists to produce valuable triazole derivatives with minimal setup. The process begins by combining the aryl ethyl ketone and a stoichiometric amount of iodine in DMSO, heating the mixture to initiate the oxidation phase. Once the diketone intermediate is generated, the trifluoroethylimide hydrazide, along with additional iodine, phosphate buffer, and pyridine, is introduced directly to the reaction vessel. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and thermal profiles optimized for maximum yield. This one-pot strategy significantly reduces the labor intensity associated with multi-step syntheses, as it avoids the need for intermediate isolation, drying, and re-dissolution. The robustness of the protocol allows for easy adaptation to different substrate combinations, as demonstrated by the successful synthesis of various derivatives including those with methoxy, chloro, and heteroaryl substitutions shown in the product scope. 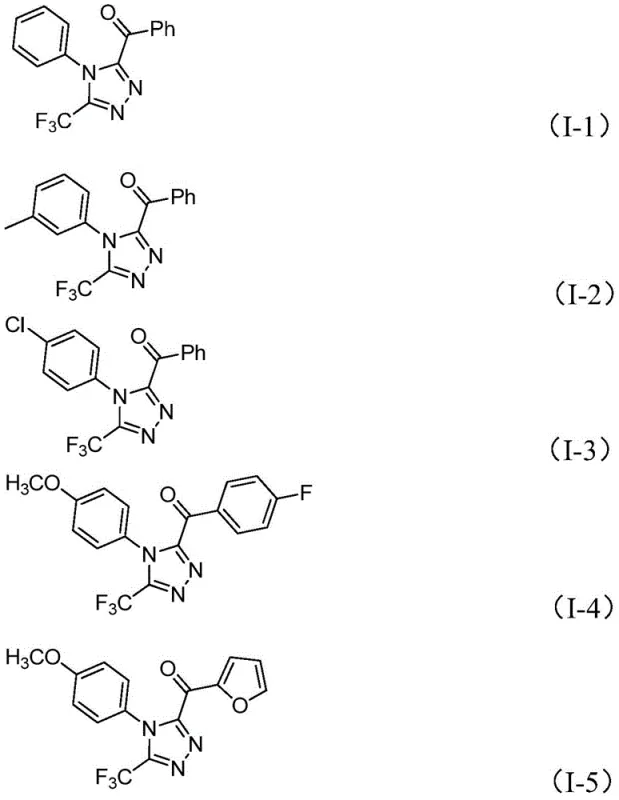 .
.
- Oxidation Phase: React aryl ethyl ketone with elemental iodine in DMSO at 90-110°C for 4-6 hours to generate the aryl diketone intermediate via Kornblum oxidation.
- Cyclization Phase: Add trifluoroethylimide hydrazide, additional iodine, sodium dihydrogen phosphate, and pyridine to the mixture.
- Completion: Heat the reaction to 110-130°C for 12-20 hours to effect intramolecular cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iodine-promoted synthesis route offers transformative economic and logistical benefits. The primary driver of cost efficiency lies in the replacement of expensive noble metal catalysts with commodity-grade elemental iodine, which is abundant and inexpensive. This shift not only lowers the direct material cost but also eliminates the downstream expenses associated with metal scavenging resins and extensive purification protocols required to meet regulatory limits for heavy metals in APIs. The simplified workflow reduces the overall cycle time per batch, enhancing throughput capacity without requiring additional capital investment in specialized reactor infrastructure. Furthermore, the use of readily available starting materials such as substituted acetophenones ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized reagents that often suffer from long lead times.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts results in substantial cost savings by removing the need for expensive palladium or copper complexes and their associated ligands. Additionally, the simplified workup procedure, which often requires only filtration and standard column chromatography, reduces solvent usage and waste disposal costs significantly. The high atom economy of the tandem reaction ensures that a larger proportion of the starting mass is converted into the desired product, minimizing raw material waste and improving the overall process mass intensity (PMI). These factors collectively contribute to a more lean and cost-effective manufacturing process that enhances profit margins for high-volume API production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like iodine, DMSO, and common aryl ketones ensures that the supply chain is not vulnerable to the bottlenecks often seen with specialized catalytic systems. Since the reaction does not require stringent anhydrous or oxygen-free conditions, it can be performed in standard glass-lined or stainless steel reactors available in most contract manufacturing organizations (CMOs). This flexibility allows for rapid technology transfer between sites and reduces the dependency on single-source suppliers for critical reagents. The robustness of the process against minor variations in reaction conditions further guarantees consistent batch-to-batch quality, ensuring uninterrupted supply for downstream drug formulation.
- Scalability and Environmental Compliance: The protocol is inherently scalable, having been demonstrated to proceed efficiently from milligram to gram scales with consistent yields, indicating strong potential for ton-scale production. The absence of toxic heavy metals aligns perfectly with increasingly strict environmental regulations and corporate sustainability goals, simplifying the permitting process for new manufacturing lines. The use of DMSO, a high-boiling polar aprotic solvent, facilitates high-temperature reactions without the need for pressurized vessels, enhancing operational safety. Moreover, the reduced generation of hazardous metal-containing waste streams simplifies effluent treatment and lowers the environmental footprint of the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this triazole synthesis platform. These insights are derived directly from the experimental data and scope analysis provided in the patent documentation, offering clarity on substrate compatibility and process parameters. Understanding these details is crucial for R&D teams planning to integrate this methodology into their existing pipelines for heterocycle construction. The answers reflect the practical realities of scaling this chemistry while maintaining high purity and yield standards.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented process utilizes elemental iodine as a non-metal promoter, completely eliminating the need for costly and toxic heavy metal catalysts, which simplifies downstream purification and reduces environmental impact.
Q: What is the substrate scope for the R1 and R2 groups in this triazole synthesis?
A: The method demonstrates broad functional group tolerance, successfully accommodating substituted phenyl groups with methyl, methoxy, chloro, or trifluoromethyl substituents, as well as heteroaryl groups like furan.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the protocol is designed for scalability, operating under standard atmospheric conditions without stringent anhydrous or oxygen-free requirements, making it highly viable for commercial kilogram-to-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in accelerating drug discovery and development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We are committed to delivering high-purity 3,4,5-trisubstituted 1,2,4-triazoles that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in metal-free synthesis strategies allows us to offer cost-effective solutions that do not compromise on quality or regulatory compliance.
We invite you to collaborate with us to leverage this innovative iodine-promoted technology for your next API program. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce time-to-market for your therapeutic candidates.
