Scalable Metal-Free Synthesis of Trifluoromethyl-Triazoles for Advanced Pharmaceutical Intermediate Manufacturing
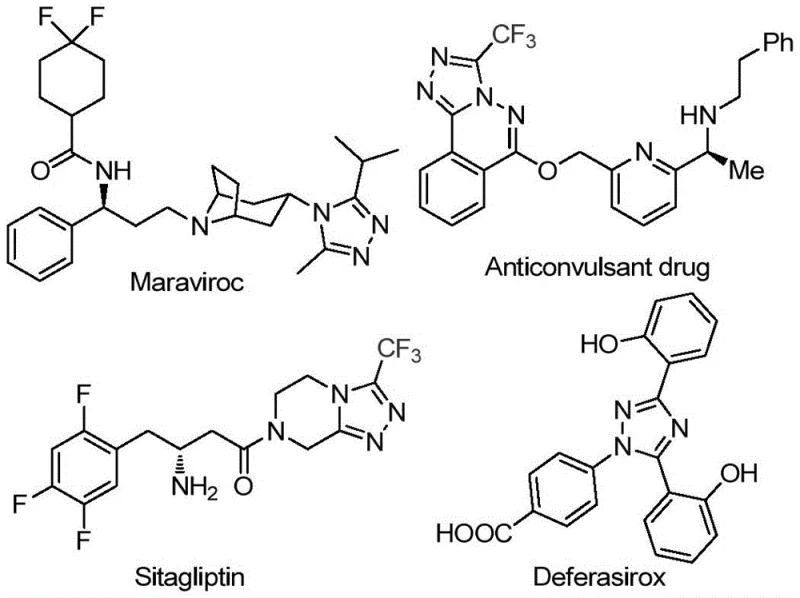
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly those incorporating trifluoromethyl groups which enhance metabolic stability and lipophilicity. Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, a privileged scaffold found in numerous bioactive molecules. As illustrated in the structural diversity of approved drugs, this heterocyclic core is integral to the efficacy of agents such as Maraviroc, Sitagliptin, and Deferasirox, highlighting its critical role in modern medicinal chemistry. The disclosed technology offers a streamlined, metal-free approach that addresses long-standing challenges in impurity control and process safety, positioning it as a vital asset for any reliable pharmaceutical intermediate supplier aiming to optimize their portfolio.
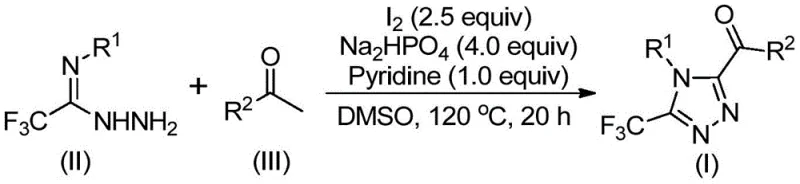
Traditional synthetic routes for accessing polysubstituted 1,2,4-triazoles often rely heavily on transition metal catalysis or harsh reaction conditions that complicate downstream processing. Conventional methods frequently necessitate the use of copper or palladium catalysts, which not only inflate raw material costs but also introduce significant regulatory burdens regarding residual heavy metal limits in final Active Pharmaceutical Ingredients (APIs). Furthermore, many existing protocols require stringent anhydrous and oxygen-free environments, demanding specialized equipment and increasing operational complexity. These limitations often result in lower overall yields and generate substantial hazardous waste, creating bottlenecks for procurement managers focused on cost reduction in API manufacturing and supply chain heads concerned with environmental compliance.
In stark contrast, the novel approach detailed in the patent leverages a simple yet highly effective iodine-promoted tandem reaction system. By utilizing elemental iodine and dimethyl sulfoxide (DMSO), the method facilitates an in situ Kornblum oxidation of aryl ethyl ketones followed by a condensation-cyclization sequence with trifluoroethylimide hydrazide. This strategy eliminates the need for toxic heavy metals entirely, thereby simplifying the purification process and significantly reducing the environmental footprint. The reaction operates efficiently under atmospheric conditions without the need for inert gas protection, making it exceptionally user-friendly and scalable. This technological shift represents a paradigm change towards greener, more economical synthesis of complex heterocycles.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and cyclocondensation steps driven by the unique reactivity of the iodine-DMSO system. Initially, the aryl ethyl ketone undergoes an iodination and subsequent Kornblum oxidation to generate an reactive alpha-dicarbonyl intermediate or its equivalent species within the reaction mixture. This activated species then engages in a nucleophilic attack by the hydrazide moiety of the trifluoroethylimide hydrazide, forming a hydrazone intermediate. The presence of sodium dihydrogen phosphate and pyridine acts as a crucial buffer and base system, facilitating the dehydration and subsequent intramolecular cyclization that constructs the triazole ring. This cascade occurs seamlessly in a single pot, minimizing intermediate isolation steps and maximizing atom economy.
From a quality control perspective, the absence of transition metals is a decisive advantage for maintaining a clean impurity profile. Heavy metal catalysts often lead to difficult-to-remove organometallic byproducts that can persist through multiple purification stages, posing risks to patient safety. By employing elemental iodine, which is volatile and easily removed during workup, the process ensures that the final high-purity pharmaceutical intermediate meets stringent regulatory specifications with minimal effort. Additionally, the broad functional group tolerance observed in the substrate scope allows for the incorporation of various electron-donating and electron-withdrawing groups on the aromatic rings, enabling the rapid generation of diverse analog libraries for structure-activity relationship (SAR) studies without compromising reaction efficiency.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory discovery and pilot plant operations. The procedure involves a straightforward two-stage heating regimen in DMSO, where reagents are added sequentially to control the exothermic nature of the oxidation step. Detailed standard operating procedures for this synthesis, including precise stoichiometric ratios and temperature ramps, are outlined below to ensure reproducible results across different batches. Adhering to these optimized parameters guarantees high conversion rates and minimizes the formation of side products, ensuring a robust supply of critical intermediates.
- Mix aryl ethyl ketone and elemental iodine in DMSO, heating to 90-110°C for 4-6 hours to initiate oxidation.
- Add trifluoroethylimide hydrazide, sodium dihydrogen phosphate, pyridine, and additional iodine, then heat to 110-130°C for 12-20 hours.
- Perform post-treatment via filtration and silica gel column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this iodine-mediated synthesis offers tangible strategic benefits that extend beyond mere chemical novelty. The reliance on commodity chemicals such as acetophenones, elemental iodine, and DMSO drastically reduces raw material volatility compared to processes dependent on scarce noble metals. This stability in sourcing translates directly into more predictable budgeting and reduced risk of supply disruptions, which is critical for maintaining continuous manufacturing lines in a competitive global market. Furthermore, the simplified workup procedure reduces the consumption of silica gel and solvents during purification, contributing to significant operational expenditure savings.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or copper removes a major cost driver from the bill of materials. Additionally, the avoidance of specialized anhydrous conditions lowers energy consumption and equipment maintenance costs associated with inert atmosphere systems. The use of cheap and readily available starting materials ensures that the overall production cost remains low, allowing for substantial cost savings in the manufacturing of complex heterocyclic intermediates without sacrificing quality or yield.
- Enhanced Supply Chain Reliability: Since the key reagents like aryl ethyl ketones and elemental iodine are bulk commodities available from multiple global suppliers, the risk of single-source dependency is minimized. This diversification of the supply base enhances resilience against market fluctuations and geopolitical instabilities. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation, ensuring consistent supply continuity for downstream API production.
- Scalability and Environmental Compliance: The method is explicitly designed for scalability, having been demonstrated to expand easily from milligram to gram scales with consistent performance. The absence of heavy metals simplifies waste treatment protocols, as the effluent does not require complex heavy metal scavenging processes. This aligns perfectly with increasingly strict environmental regulations, reducing the administrative burden and cost associated with hazardous waste disposal while promoting a greener manufacturing image.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing production workflows.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metallic promoter, completely avoiding the use of toxic and expensive heavy metal catalysts like palladium or copper.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction proceeds in dimethyl sulfoxide (DMSO) solvent, typically requiring a two-stage heating process: initial oxidation at 90-110°C followed by cyclization at 110-130°C under atmospheric conditions without strict anhydrous requirements.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method is easily expandable to the gram level and beyond, utilizing cheap and readily available starting materials like aryl ethyl ketones, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that efficient synthetic methodologies play in accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3,4,5-trisubstituted 1,2,4-triazole intermediate meets the highest international standards, providing our partners with the confidence needed to advance their clinical programs.
We invite you to collaborate with us to leverage this advanced iodine-catalyzed technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and efficiency for your organization.
