Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Molybdenum-Copper Co-Catalysis
Scalable Production of 3-Trifluoromethyl-1,2,4-Triazoles via Molybdenum-Copper Co-Catalysis
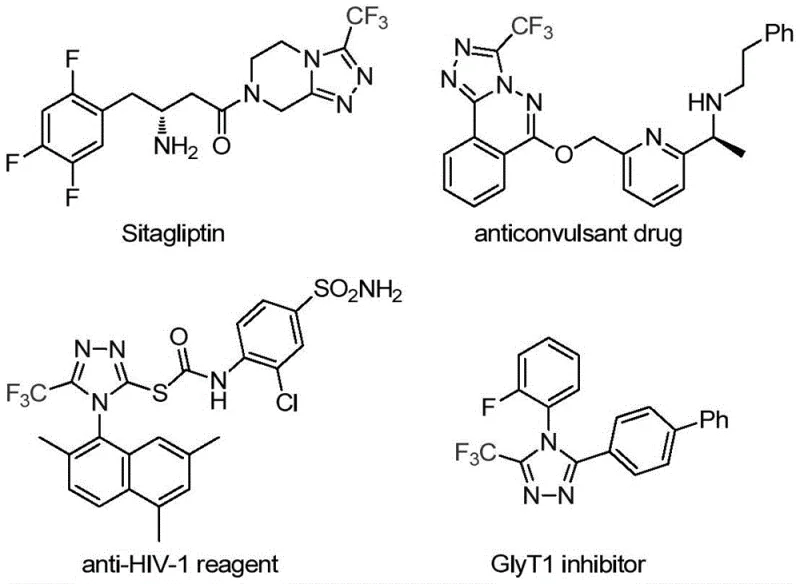
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds remains a cornerstone of modern medicinal chemistry, profoundly influencing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As highlighted in recent intellectual property disclosures, specifically patent CN113307778A, a significant technological breakthrough has been achieved in the efficient construction of 3-trifluoromethyl substituted 1,2,4-triazole compounds. This novel synthetic methodology addresses long-standing challenges in heterocyclic chemistry by utilizing a dual-metal catalytic system that operates under remarkably mild conditions. The relevance of this chemical motif is underscored by its presence in blockbuster pharmaceuticals such as Sitagliptin and various anticonvulsant agents, where the triazole ring serves as a critical pharmacophore. For R&D directors and process chemists, mastering this transformation opens new avenues for rapid library synthesis and lead optimization without the burden of harsh reaction parameters typically associated with fluorinated heterocycles.
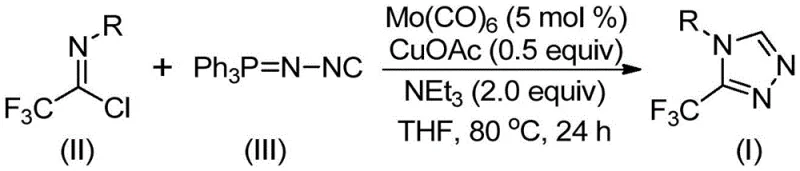
Historically, the construction of fully substituted 1,2,4-triazoles, particularly those bearing trifluoromethyl groups at the 3-position, has relied on cumbersome and often hazardous protocols. Conventional literature methods frequently involve the cyclization of trifluoroacetyl hydrazine with amidine compounds or the hydrazinolysis of trifluoromethyl substituted 1,2,4-oxazolinone. These traditional routes are plagued by significant limitations, including the requirement for strongly acidic or basic conditions, poor atom economy, and the generation of difficult-to-remove impurities. Furthermore, alternative copper-catalyzed multi-component reactions utilizing diazonium salts pose severe safety risks due to the explosive nature of diazo intermediates, making them unsuitable for large-scale commercial manufacturing. The reliance on such unstable precursors not only increases operational costs regarding safety infrastructure but also introduces unacceptable variability in batch-to-batch consistency, a critical failure point for supply chain reliability in the pharmaceutical sector.
In stark contrast, the novel approach detailed in the patent data leverages a sophisticated yet operationally simple co-catalytic system involving molybdenum hexacarbonyl and cuprous acetate. This method bypasses the need for dangerous diazonium salts entirely, instead utilizing stable and commercially accessible trifluoroethylimidoyl chloride and functionalized isonitrile (NIITP) as the primary building blocks. The reaction proceeds through a streamlined [3+2] cycloaddition pathway that tolerates a wide array of functional groups, including electron-donating and electron-withdrawing substituents on the aromatic ring. By shifting the paradigm from hazardous multi-step sequences to a direct, one-pot cyclization, this technology drastically simplifies the synthetic route. The ability to access diverse 3,4-disubstituted 1,2,4-triazoles with high efficiency represents a substantial leap forward, offering process chemists a robust tool for generating high-purity intermediates essential for downstream API synthesis.
Mechanistic Insights into Mo/Cu Co-Catalyzed Cycloaddition
The mechanistic elegance of this transformation lies in the synergistic interaction between the molybdenum and copper centers, which orchestrates the assembly of the triazole ring with high precision. The reaction initiates with the activation of the functionalized isonitrile by molybdenum hexacarbonyl, forming a reactive metal-isocyanide complex that enhances the nucleophilicity of the carbon center. Subsequently, the cuprous acetate promoter facilitates the [3+2] cycloaddition of this activated species onto the trifluoroethylimidoyl chloride. This step is critical, as it constructs the five-membered heterocyclic core while maintaining the integrity of the sensitive trifluoromethyl group. The final stage of the catalytic cycle involves the elimination of triphenylphosphine oxide, driven by the presence of water or moisture within the system, to yield the thermodynamically stable 3-trifluoromethyl-1,2,4-triazole product. Understanding this mechanism is vital for R&D teams aiming to further optimize reaction kinetics or adapt the protocol for continuous flow processing.
From an impurity control perspective, this catalytic system offers distinct advantages over traditional acid-mediated cyclizations. The mild thermal conditions (70-90°C) and neutral to slightly basic environment provided by triethylamine minimize the risk of hydrolysis or decomposition of the trifluoromethyl moiety, which is often prone to defluorination under harsh acidic conditions. Moreover, the use of molecular sieves in the reaction mixture helps to sequester water generated during the process, driving the equilibrium towards product formation and preventing side reactions associated with hydrolysis of the imidoyl chloride starting material. The resulting crude reaction mixtures are generally cleaner, facilitating simpler purification workflows such as standard silica gel column chromatography. This high level of chemoselectivity ensures that the final API intermediates meet stringent purity specifications required by regulatory bodies, reducing the burden on analytical quality control laboratories.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The implementation of this synthesis protocol is designed for ease of execution in standard laboratory and pilot plant settings. The procedure involves charging a reaction vessel with the catalytic system comprising molybdenum hexacarbonyl, cuprous acetate, and triethylamine in an aprotic solvent like THF. Following the addition of the substrates, the mixture is heated to promote the cycloaddition. The robustness of the method allows for a broad substrate scope, accommodating various aryl and phenethyl groups without significant loss in yield. For detailed operational parameters and specific stoichiometric ratios validated across multiple examples, please refer to the standardized synthesis guide below.
- Combine molybdenum hexacarbonyl (5 mol %), cuprous acetate (0.5 equiv), triethylamine (2.0 equiv), and molecular sieves in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride and the functionalized isonitrile (Ph3P=N-NC) to the reaction mixture under inert atmosphere.
- Heat the reaction mixture to 70-90°C for 18-30 hours, then filter and purify via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this Mo/Cu co-catalyzed methodology presents a compelling value proposition centered on cost efficiency and supply security. The shift away from exotic or hazardous reagents towards commodity chemicals significantly de-risks the sourcing strategy. Trifluoroethylimidoyl chloride and the requisite isonitriles are readily available from global chemical suppliers, ensuring a stable and competitive supply base. This accessibility eliminates the bottlenecks often associated with custom-synthesized precursors, allowing for more accurate demand forecasting and inventory management. Furthermore, the simplified reaction workflow reduces the overall manufacturing cycle time, enabling faster response to market demands for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive noble metal catalysts with abundant and inexpensive transition metals like copper and molybdenum. Unlike palladium or rhodium-based systems that require rigorous and costly metal scavenging steps to meet residual metal limits in APIs, this copper/molybdenum system utilizes catalysts that are easier to remove and significantly cheaper to procure. Additionally, the high reaction efficiency and yields reported (often exceeding 90% for optimized substrates) translate directly into improved material throughput and reduced waste disposal costs. The elimination of cryogenic conditions or high-pressure equipment further lowers the capital expenditure (CAPEX) and operational expenditure (OPEX) required for production facilities.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust starting materials that possess long shelf lives and do not require specialized cold-chain logistics. The stability of the trifluoroethylimidoyl chloride precursor ensures that raw material inventories can be maintained without significant degradation, mitigating the risk of production stoppages due to spoiled reagents. Moreover, the scalability of the reaction, demonstrated from milligram to gram scales in the patent data, suggests a smooth path to kilogram and ton-scale production. This scalability ensures that suppliers can reliably meet the volume requirements of multinational pharmaceutical clients without the need for complex process re-engineering.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, this method aligns well with green chemistry principles. The avoidance of explosive diazonium salts removes a major safety hazard from the manufacturing floor, reducing insurance premiums and safety compliance overheads. The use of THF as a solvent, while requiring recovery systems, is a standard industry practice with well-established recycling protocols. The high atom economy of the cycloaddition reaction minimizes the generation of organic waste streams, simplifying wastewater treatment processes. These factors collectively contribute to a more sustainable manufacturing profile, which is increasingly a prerequisite for vendor qualification by top-tier pharmaceutical companies committed to reducing their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the practical application and scope of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating this route for their specific projects. Understanding these nuances is essential for effective technology transfer and process validation.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The patent specifies reacting at 70-90°C for 18-30 hours in THF solvent, using Mo(CO)6 and CuOAc as co-catalysts with triethylamine as the base.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the method demonstrates broad substrate tolerance, successfully synthesizing derivatives with methyl, methoxy, fluoro, chloro, nitro, and naphthyl substituents with yields ranging from moderate to excellent.
Q: Is this process suitable for large-scale manufacturing?
A: The patent explicitly states the method can be expanded to gram-level reactions and utilizes cheap, commercially available starting materials, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole delivered meets the exacting standards required for GMP API synthesis. Our commitment to technical excellence ensures that we can handle the complexities of fluorinated chemistry with precision and safety.
We invite you to collaborate with us to leverage this advanced synthetic technology for your pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact us directly to obtain specific COA data for our catalog compounds and to discuss route feasibility assessments for custom derivatives. Let us help you secure a reliable supply of these valuable building blocks while optimizing your overall production costs.
