Sustainable Manufacturing of 3-Trifluoromethyl-1,2,4-Triazoles via Glucose Cascade Cyclization
Sustainable Manufacturing of 3-Trifluoromethyl-1,2,4-Triazoles via Glucose Cascade Cyclization
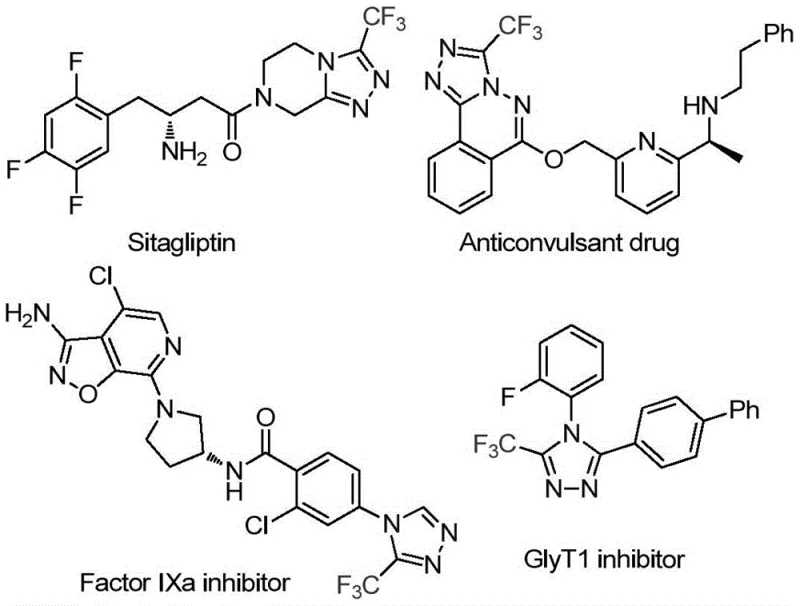
The pharmaceutical industry is constantly seeking more sustainable and cost-effective routes for constructing nitrogen-containing heterocycles, particularly the 1,2,4-triazole scaffold which serves as a critical pharmacophore in numerous blockbuster drugs. Patent CN113880781A introduces a groundbreaking methodology for synthesizing 3-trifluoromethyl-substituted 1,2,4-triazole compounds by utilizing glucose as a renewable carbon source. This innovation represents a significant shift from traditional petrochemical-dependent pathways, leveraging the abundance of biomass to generate high-value chemical intermediates. The trifluoromethyl group is renowned for enhancing the metabolic stability and lipophilicity of drug candidates, making this scaffold indispensable in modern medicinal chemistry programs targeting various therapeutic areas.
The strategic importance of this technology lies in its ability to streamline the supply chain for complex heterocyclic building blocks. By replacing expensive, multi-step aldehyde precursors with inexpensive glucose, the process fundamentally alters the economic landscape of triazole manufacturing. This approach not only aligns with green chemistry principles by utilizing renewable feedstocks but also simplifies the synthetic sequence through a cascade reaction mechanism. For R&D directors and procurement managers alike, this patent offers a compelling value proposition: high-efficiency synthesis with reduced dependency on volatile petrochemical markets, ensuring greater long-term supply security for critical API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 3-trifluoromethyl-1,2,4-triazoles often rely on the condensation of pre-functionalized aldehydes or nitriles with hydrazine derivatives, followed by cyclization and oxidation steps. These conventional methods frequently suffer from several critical drawbacks that hinder large-scale production efficiency. Firstly, the requisite aldehyde starting materials are often costly, unstable, or require hazardous reagents for their own preparation, adding significant upstream costs and safety risks to the overall process. Secondly, many existing protocols demand stringent reaction conditions, such as strictly anhydrous environments or the use of toxic heavy metal catalysts, which complicate waste treatment and increase the environmental footprint of the manufacturing facility.
Furthermore, the purification of intermediates in traditional multi-step sequences can be labor-intensive, leading to lower overall yields and higher solvent consumption. The reliance on petrochemical-derived precursors also exposes the supply chain to price volatility and geopolitical instability. For a reliable pharmaceutical intermediate supplier, these factors create bottlenecks in scaling up production to meet the demands of global clinical trials and commercial launches. The accumulation of impurities from harsh reaction conditions often necessitates complex chromatographic separations, further driving up the cost of goods sold (COGS) and extending lead times for delivery to downstream API manufacturers.
The Novel Approach
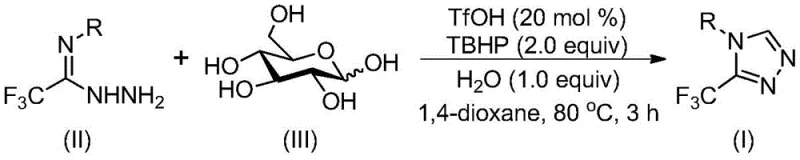
In stark contrast, the methodology disclosed in CN113880781A utilizes a biomimetic cascade strategy that transforms glucose directly into the target triazole skeleton. This novel approach bypasses the need for isolated aldehyde intermediates by generating them in situ through the acid-catalyzed cleavage of glucose. The reaction employs trifluoromethanesulfonic acid (TfOH) as a potent yet catalytic promoter and tert-butyl hydroperoxide (TBHP) as a clean oxidant, facilitating a seamless transition from the sugar backbone to the aromatic heterocycle. This telescoped process significantly reduces the number of unit operations required, thereby minimizing solvent usage and waste generation while maximizing atom economy.
The operational simplicity of this new route is a major advantage for industrial adoption. It proceeds under mild thermal conditions (70–90 °C) and tolerates the presence of water, eliminating the need for expensive drying agents or inert atmosphere gloveboxes. This robustness allows for the use of standard stainless steel reactors commonly found in multipurpose chemical plants, facilitating rapid technology transfer from the laboratory to pilot and commercial scales. By leveraging glucose, a commodity chemical with a stable global supply, this method ensures cost reduction in pharmaceutical intermediate manufacturing while providing a versatile platform for introducing diverse functional groups onto the triazole ring through simple substrate modification.
Mechanistic Insights into TfOH-Catalyzed Cascade Cyclization
The mechanistic pathway of this transformation is a sophisticated example of acid-promoted biomass valorization. Initially, the strong acid catalyst, trifluoromethanesulfonic acid, activates the glucose molecule, promoting its fragmentation to generate reactive aldehyde species. These transient aldehydes immediately undergo condensation with the trifluoroethylimide hydrazide to form a hydrazone intermediate. This in situ generation of the electrophile prevents side reactions associated with free aldehydes and drives the equilibrium forward. Subsequently, an intramolecular nucleophilic addition occurs, closing the five-membered triazole ring. The final aromatization step is achieved through the oxidative action of TBHP, which removes the necessary hydrogen atoms to establish the fully conjugated 1,2,4-triazole system.
From an impurity control perspective, this mechanism offers distinct advantages. The rapid consumption of the reactive aldehyde intermediate minimizes the formation of aldol condensation byproducts that typically plague sugar-based chemistry. Furthermore, the use of TBHP as a terminal oxidant results in tert-butanol as the primary byproduct, which is relatively benign and easy to remove during aqueous workup. The high chemoselectivity of the TfOH catalyst ensures that sensitive functional groups on the aromatic ring, such as halogens or sulfides, remain intact during the vigorous cyclization process. This selectivity is crucial for maintaining the integrity of complex drug-like molecules and reduces the burden on downstream purification teams to separate structurally similar impurities.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing high-purity triazole derivatives suitable for drug discovery and development. The procedure is designed to be operationally simple, requiring standard laboratory equipment and commercially available reagents. By optimizing the molar ratios of glucose, hydrazide, and oxidant, chemists can achieve excellent conversion rates across a wide range of substrates. The following guide summarizes the critical parameters for successful implementation of this technology in a research or production setting.
- Combine trifluoroethylimide hydrazide, glucose, trifluoromethanesulfonic acid (catalyst), tert-butyl hydroperoxide (oxidant), and water in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature between 70°C and 90°C and maintain stirring for 2 to 4 hours to allow the cascade cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final 3-trifluoromethyl-substituted 1,2,4-triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this glucose-based synthesis route offers transformative benefits regarding cost structure and supply reliability. The substitution of expensive, custom-synthesized aldehydes with bulk-grade glucose represents a fundamental shift in the bill of materials, drastically lowering the raw material input costs. Since glucose is produced on a massive global scale for the food and fermentation industries, its price is stable and decoupled from the fluctuations of the petrochemical market. This stability allows for more accurate long-term budgeting and reduces the financial risk associated with raw material price spikes.
- Cost Reduction in Manufacturing: The elimination of pre-formed aldehyde precursors removes several synthetic steps from the supply chain, directly translating to lower manufacturing costs. Additionally, the catalytic nature of the acid and the use of inexpensive aqueous TBHP reduce reagent expenses significantly. The simplified workup procedure, which avoids complex extractions or heavy metal scavenging, further decreases processing time and utility consumption, leading to substantial overall cost savings in the production of these valuable heterocycles.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the use of glucose, a commodity chemical with multiple global suppliers and established logistics networks. This diversification of supply sources mitigates the risk of single-source bottlenecks that often occur with specialized fine chemical intermediates. The robustness of the reaction conditions also means that production can be easily transferred between different manufacturing sites without the need for highly specialized infrastructure, ensuring continuous supply even during regional disruptions.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne scales. The absence of toxic heavy metals and the use of water-tolerant conditions simplify wastewater treatment and regulatory compliance. This environmental friendliness aligns with the increasing pressure from regulators and end-clients for greener manufacturing processes, potentially accelerating regulatory approvals and enhancing the marketability of the final API.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this glucose-derived triazole synthesis. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for technical teams evaluating this route for potential licensing or contract manufacturing opportunities.
Q: What are the key advantages of using glucose in this triazole synthesis?
A: Using glucose eliminates the need for pre-synthesized aldehyde starting materials, significantly reducing raw material costs. As a widely available biomass feedstock, glucose enhances supply chain stability and sustainability compared to petrochemical-derived precursors.
Q: Does this method require strict anhydrous or anaerobic conditions?
A: No, one of the distinct operational advantages of this protocol is that it does not require rigorous anhydrous or oxygen-free environments. The reaction tolerates the presence of water, which simplifies the operational setup and reduces equipment costs for industrial scale-up.
Q: What is the substrate scope for the R group in the triazole product?
A: The method demonstrates broad compatibility with various substituents on the aryl ring, including alkyl, alkoxy, alkylthio, halogens, and trifluoromethyl groups at ortho, meta, or para positions, allowing for the synthesis of diverse pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic methodologies like the glucose-based cascade cyclization described in CN113880781A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are efficiently translated into reliable commercial supplies. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including the safe management of peroxide oxidants and the rigorous purification needed to meet stringent purity specifications for pharmaceutical applications.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective technology for their pipeline projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data for our triazole intermediates and to discuss route feasibility assessments for your target molecules, ensuring a seamless integration of this sustainable chemistry into your supply chain.
