Advanced Iodine-Promoted Cyclization for High-Purity 3-Trifluoromethyl-1,2,4-Triazoles
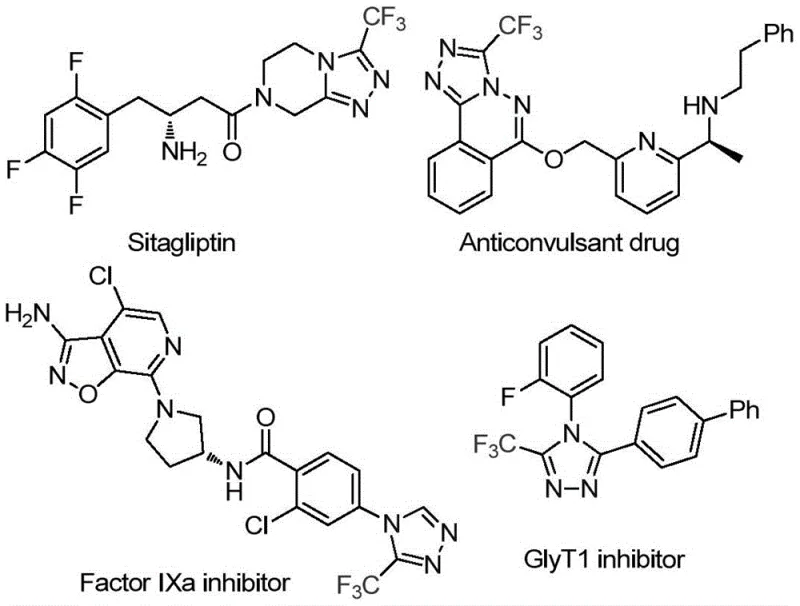
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable methods to construct fluorinated heterocycles, which serve as critical pharmacophores in modern drug design. Patent CN114920707B, published in April 2023, introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds that addresses many of the historical bottlenecks in this chemical space. This technology leverages the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source, enabling a highly efficient tandem cyclization promoted by molecular iodine. The significance of this development cannot be overstated for R&D teams targeting bioactive molecules; as illustrated in the structural diversity of known inhibitors like Sitagliptin and Factor IXa inhibitors, the 1,2,4-triazole core is a privileged scaffold. By providing a route that operates under air atmosphere with readily available starting materials, this patent offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to streamline their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of trifluoromethyl-substituted 1,2,4-triazoles has been plagued by operational complexities that hinder large-scale adoption. Conventional routes often rely on harsh reaction conditions, requiring strictly anhydrous and anaerobic environments to prevent side reactions or decomposition of sensitive intermediates. Furthermore, many existing methodologies necessitate the use of specialized, expensive C1 building blocks or transition metal catalysts that introduce heavy metal contamination risks, necessitating costly purification steps to meet stringent regulatory standards for high-purity pharmaceutical intermediates. The multi-step nature of these legacy processes often results in lower overall yields and generates significant chemical waste, creating both economic and environmental burdens for manufacturing facilities. Additionally, the sensitivity of reagents often limits the substrate scope, making it difficult to introduce diverse functional groups without protecting group strategies, thereby extending development timelines.
The Novel Approach
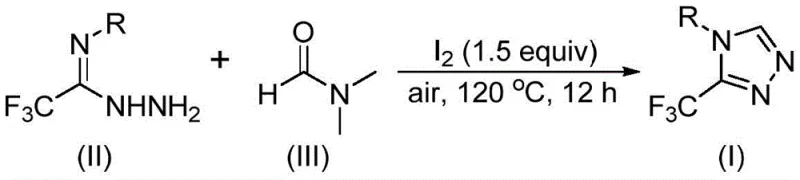
In stark contrast, the methodology disclosed in CN114920707B represents a paradigm shift towards green and economical synthesis. The core innovation lies in the utilization of DMF as a dual-function reagent: it acts as the polar aprotic solvent while simultaneously providing the necessary carbon atom for ring closure. This eliminates the need for external formylating agents. The reaction is promoted by molecular iodine, a cheap and non-toxic halogen source, and proceeds smoothly under an air atmosphere at temperatures between 110°C and 130°C. This tolerance to oxygen and moisture drastically simplifies the engineering requirements for reactors, removing the need for complex inert gas manifolds or glovebox operations. As shown in the general reaction scheme below, the transformation converts trifluoroethyliminohydrazide directly into the target triazole in a single pot, demonstrating exceptional atom economy and operational simplicity.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
Understanding the mechanistic pathway is crucial for R&D directors evaluating the robustness of this synthesis. The patent elucidates two potential pathways depending on which part of the DMF molecule participates as the carbon source, though the N-methyl pathway appears dominant in generating the specific azadiene intermediates required for aromatization. Initially, molecular iodine activates the DMF solvent, facilitating the cleavage of the C-N bond. When the N-methyl group acts as the carbon source, DMF reacts with iodine to form an amine salt intermediate. Following the elimination of hydrogen iodide, this activated species undergoes nucleophilic addition with the trifluoroethyliminohydrazide substrate. This step is critical as it forms the backbone of the future heterocyclic ring. Subsequent elimination of N-methylformamide generates a reactive azadiene intermediate, which then undergoes intramolecular nucleophilic attack followed by oxidative aromatization to yield the stable 1,2,4-triazole ring system. This intricate cascade highlights the elegance of using a simple solvent to drive complex bond formations.
From an impurity control perspective, this mechanism offers distinct advantages. Because the reaction relies on a tandem cyclization rather than discrete isolation of unstable intermediates, the exposure of reactive species to potential degradative pathways is minimized. The use of iodine as a mild oxidant ensures that the final aromatization step proceeds cleanly without over-oxidation of the sensitive trifluoromethyl group or the aromatic substituents. Furthermore, the byproduct profile is relatively benign, primarily consisting of dimethylamine salts or formamide derivatives which are easily removed during the aqueous workup and silica gel purification steps described in the patent examples. This clean reaction profile is essential for maintaining the high purity specifications required for commercial scale-up of complex pharmaceutical intermediates, ensuring that downstream processing remains efficient and cost-effective.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical implementation of this synthesis is designed for ease of execution in standard laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the trifluoroethyliminohydrazide precursor, molecular iodine, and DMF. The mixture is then heated to the optimal range of 110-130°C for a duration of 10 to 15 hours. The detailed standardized synthesis steps, including precise molar ratios and workup procedures, are outlined in the guide below to ensure reproducibility and safety during operation.
- Combine trifluoroethyliminohydrazide, molecular iodine (1.5 equivalents), and DMF solvent in a reaction vessel.
- Heat the mixture to 110-130°C under an air atmosphere and stir for 10-15 hours to facilitate tandem cyclization.
- Upon completion, perform standard post-treatment including filtration, washing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this technology are profound. The shift away from exotic reagents and harsh conditions translates directly into improved margins and supply security. By leveraging commodity chemicals like DMF and iodine, manufacturers can decouple their production costs from the volatility associated with specialized fine chemical suppliers. The elimination of inert atmosphere requirements further reduces capital expenditure on equipment and lowers the energy consumption associated with nitrogen or argon generation. This process optimization supports cost reduction in API manufacturing by streamlining the entire production workflow from raw material intake to final isolation.
- Cost Reduction in Manufacturing: The most significant driver of cost savings is the dual role of DMF. By serving as both solvent and reactant, the process eliminates the purchase of separate C1 synthons, which are often expensive and hazardous to handle. Additionally, the use of molecular iodine instead of precious metal catalysts removes the need for expensive metal scavenging resins and the associated validation testing for heavy metal residues. The simplified workup, involving basic filtration and chromatography, reduces labor hours and solvent consumption during purification, leading to substantial operational expenditure reductions without compromising yield.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of globally available, commodity-grade starting materials. DMF and iodine are produced at massive scales for various industries, ensuring consistent availability and price stability compared to niche heterocyclic building blocks. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failure related to complex inert gas systems. This reliability allows for more accurate forecasting and reducing lead time for high-purity pharmaceutical intermediates, enabling faster response to market demands for active pharmaceutical ingredients.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is straightforward due to the absence of sensitive reagents. The reaction does not generate excessive hazardous waste streams typical of traditional cyclization methods, aligning with modern green chemistry principles. The ability to run the reaction under air simplifies reactor design, allowing for the use of standard glass-lined or stainless steel vessels without specialized modifications. This ease of scale-up ensures that supply can be rapidly increased to meet commercial demand while maintaining strict adherence to environmental regulations regarding solvent usage and waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, offering clarity on reaction parameters and substrate compatibility for potential licensees or manufacturing partners.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the critical carbon source (C1 synthon), eliminating the need for separate, expensive formylating agents and significantly simplifying the reaction stoichiometry.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, a key innovation of patent CN114920707B is that the reaction proceeds efficiently under standard air atmosphere without the need for rigorous drying of solvents or inert gas protection, greatly reducing operational complexity.
Q: What is the substrate scope for the R group in this triazole synthesis?
A: The method demonstrates broad compatibility, successfully accommodating various substituted aryl groups including those with electron-donating (methyl, methoxy) and electron-withdrawing (fluoro, chloro, trifluoromethyl) substituents at ortho, meta, and para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in the development of next-generation therapeutics. Our technical team has extensively analyzed the capabilities presented in CN114920707B and is fully prepared to integrate this iodine-promoted cyclization technology into our production portfolio. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from benchtop discovery to full-scale manufacturing. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this innovative synthetic route for your specific drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data for our available triazole derivatives and to discuss route feasibility assessments for your custom synthesis needs, ensuring a secure and efficient supply chain for your critical pharmaceutical intermediates.
