Advanced Catalytic Method for Replacing Fluorine with Hydrogen on Olefin Double Bonds for Industrial Scale-Up
The landscape of fluorine chemistry is constantly evolving, driven by the critical need for high-purity intermediates in the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is documented in patent CN107759440B, which discloses a novel method for replacing fluorine on the double bond of fluorine-containing olefins with hydrogen. This technology addresses a longstanding challenge in organic synthesis: achieving high selectivity in defluorination without compromising the structural integrity of the olefinic backbone. Traditional methods often struggle with harsh conditions or poor selectivity, leading to complex impurity profiles that are costly to remove. By introducing a two-step catalytic sequence involving carbon tetrachloride and hydrogen, this invention offers a robust pathway to valuable fluoroolefins. For R&D directors and procurement managers alike, understanding this mechanistic shift is crucial for securing a reliable fluorine-containing olefin supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of specific fluoroolefins often relied on gas-phase reactions involving hydrogen chloride (HCl), as seen in earlier patents like US20138487144. While effective in generating chlorinated intermediates, the reliance on corrosive HCl gas presents severe engineering and safety challenges. The handling of anhydrous HCl requires specialized, highly corrosion-resistant reactor materials, significantly driving up capital expenditure (CAPEX) for manufacturing facilities. Furthermore, the collection and treatment of effluent gases containing unreacted HCl demand sophisticated scrubbing systems to meet environmental regulations. From a supply chain perspective, the logistical complexity of transporting and storing large quantities of hazardous acidic gases introduces potential bottlenecks. These factors collectively limit the industrial viability of conventional routes, making cost reduction in fluorochemical manufacturing difficult to achieve without compromising safety standards.
The Novel Approach
The method described in CN107759440B circumvents these issues by utilizing carbon tetrachloride (CCl4) as the chlorine source in the first step, followed by a hydrogen substitution step. This strategic shift eliminates the need for corrosive HCl gas entirely, thereby simplifying equipment requirements and enhancing operational safety. The process operates in the gas phase over solid catalysts, specifically transitioning from a fluorine-to-chlorine exchange to a chlorine-to-hydrogen exchange. This modular approach allows for precise control over reaction parameters, such as temperature and contact time, ensuring high selectivity for the desired mono-substituted products. By avoiding the pitfalls of direct fluorine-to-hydrogen replacement, which can be energetically demanding and non-selective, this novel route provides a cleaner, more efficient pathway. The result is a process that is not only chemically elegant but also commercially attractive due to reduced waste generation and simplified downstream processing.
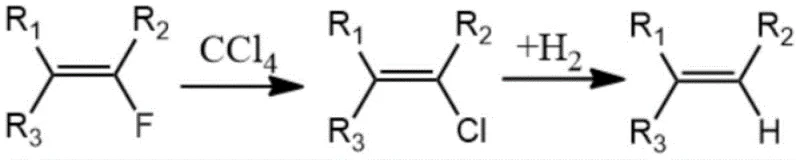
Mechanistic Insights into Gas-Phase Catalytic Halogen Exchange
The core of this technology lies in the dual-catalyst system designed to facilitate sequential halogen exchanges with high fidelity. In the first stage, the fluorine-containing olefin reacts with carbon tetrachloride over a metal-based catalyst, such as a Cr-Zn-Mg or Cr-Mg-Co composite. This catalyst promotes the substitution of the vinylic fluorine atom with a chlorine atom, generating a chlorofluoroolefin intermediate. The choice of transition metals like Chromium and Zinc is critical, as they provide the necessary Lewis acidity to activate the C-F bond while maintaining the stability of the double bond. The reaction typically proceeds at temperatures between 100°C and 600°C, with contact times ranging from 0.1 to 20 seconds. This rapid gas-phase interaction minimizes side reactions such as polymerization or over-chlorination, which are common pitfalls in liquid-phase halogenations.
Following the initial halogen exchange, the chlorofluoroolefin undergoes a hydrogenolysis reaction in the second stage. Here, a palladium on carbon (Pd/C) catalyst is employed to replace the newly introduced chlorine atom with hydrogen. This step is remarkably selective, operating effectively between 80°C and 400°C. The use of Pd/C ensures that the C-Cl bond is cleaved preferentially over the C-F bond, preserving the remaining fluorine substituents which are often essential for the biological activity of the final pharmaceutical or agrochemical product. The mechanism likely involves the oxidative addition of the C-Cl bond to the palladium center, followed by reductive elimination with hydrogen. This precise control over bond dissociation energies allows for the synthesis of complex fluoroolefins that were previously difficult to access, providing R&D teams with new tools for molecular design.
How to Synthesize Fluoroolefins Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and activation protocols to ensure optimal performance. The patent details specific procedures for creating the Cr-Zn-Mg catalyst via co-precipitation, followed by a rigorous drying and activation sequence involving hydrogen fluoride. Similarly, the Pd/C catalyst must be prepared via impregnation and activated under a hydrogen stream to generate active metallic sites. These preparatory steps are fundamental to achieving the high yields reported in the examples, such as the 90% selectivity for the chloro-intermediate and 76% yield for the final hydro-product in Example 1. For process chemists looking to replicate or scale this technology, adhering to these activation parameters is non-negotiable. The detailed standardized synthesis steps see the guide below.
- Prepare a fluorine-to-chlorine catalyst (e.g., Cr-Zn-Mg or Cr-Mg-Co) via co-precipitation, followed by drying at 400°C and activation with hydrogen fluoride.
- Conduct the gas-phase fluorination reaction by mixing the fluorine-containing olefin with carbon tetrachloride at 100-600°C over the catalyst to generate chlorofluoroolefin.
- Perform the hydrogen substitution reaction by passing the chlorofluoroolefin and hydrogen gas over a Pd/C catalyst at 80-400°C to yield the final fluoroolefin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic method represents a significant opportunity to optimize the supply of high-purity fluorochemical intermediates. The elimination of corrosive hydrogen chloride gas from the process flow drastically reduces the maintenance burden on production equipment. Standard stainless steel reactors can often be used instead of exotic alloys required for HCl service, leading to substantial cost savings in both initial setup and long-term maintenance. Furthermore, the use of carbon tetrachloride and hydrogen as reagents simplifies the raw material supply chain, as these are commodity chemicals with stable global availability. This stability mitigates the risk of supply disruptions that can occur with specialized or hazardous reagents, ensuring a more reliable fluorine-containing olefin supplier network for downstream manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the reaction infrastructure and the efficiency of the catalysts. By removing the need for expensive corrosion-resistant materials and complex acid gas scrubbing systems, the overall capital intensity of the plant is significantly reduced. Additionally, the high selectivity of the catalysts minimizes the formation of by-products, which reduces the load on purification units like distillation columns. This efficiency translates directly into lower operating expenses (OPEX) per kilogram of product. The ability to operate in a continuous gas-phase mode further enhances throughput, allowing for economies of scale that are difficult to achieve with batch processes. Consequently, the total cost of ownership for producing these valuable intermediates is markedly lower compared to traditional methods.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, heterogeneous catalysts that have long operational lifetimes. The patent describes activation procedures that stabilize the catalyst structure, ensuring consistent performance over extended periods. This reliability reduces the frequency of catalyst change-outs and associated production downtime. Moreover, the raw materials involved, such as carbon tetrachloride and hydrogen, are widely produced industrial gases and solvents, reducing dependency on niche suppliers. This broad availability ensures that production schedules can be maintained even during fluctuations in the global chemical market. For supply chain planners, this means more predictable lead times and the ability to commit to larger volume contracts with confidence, knowing that the underlying technology is stable and scalable.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method offers distinct advantages that facilitate smoother permitting and operation. The reduction in hazardous waste streams, particularly the absence of acidic wastewater from HCl scrubbing, simplifies effluent treatment. The process generates fewer industrial wastes, aligning with increasingly stringent global environmental standards. Scalability is inherent in the design, as gas-phase fixed-bed reactors are easily expanded by numbering up or increasing reactor diameter without changing the fundamental chemistry. This modularity allows manufacturers to respond quickly to market demand increases. The combination of lower waste generation and easier scalability makes this technology an ideal candidate for green chemistry initiatives, potentially qualifying for regulatory incentives or preferred vendor status with environmentally conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this halogen exchange technology. These insights are derived directly from the experimental data and claims within patent CN107759440B, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of adopting this route for specific target molecules. The answers reflect the balance between chemical performance and practical engineering considerations.
Q: Why is the two-step halogen exchange method superior to direct hydrogenation?
A: Direct hydrogenation often leads to over-reduction or requires harsh conditions. This patent's method first selectively replaces fluorine with chlorine using CCl4, which is then easily hydrogenolyzed to hydrogen, preserving the double bond integrity with high selectivity.
Q: What are the safety advantages of this process compared to prior art?
A: Unlike previous methods requiring corrosive hydrogen chloride (HCl) gas, this process utilizes carbon tetrachloride and hydrogen. This eliminates the need for highly corrosion-resistant equipment and reduces risks associated with handling toxic acidic gases.
Q: Can this process be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the process utilizes gas-phase fixed-bed reactors, which are standard in the chemical industry. The catalysts are robust, and the reaction conditions (100-600°C) are compatible with existing industrial infrastructure, facilitating easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluoroolefin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methods like the one described in CN107759440B for the production of next-generation fluorinated building blocks. Our team of expert process chemists is well-versed in adapting such innovative pathways to meet the rigorous demands of the pharmaceutical and agrochemical industries. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our facilities are equipped with state-of-the-art gas-phase reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch meets the highest international standards.
We invite you to collaborate with us to leverage this technology for your specific project needs. Whether you require custom synthesis of complex fluoroolefins or optimization of an existing route, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how we can best support your supply chain goals with high-quality, cost-effective solutions.
