Advanced Chemical Synthesis of Cephradine for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust synthetic routes that balance high purity with operational simplicity, particularly for critical antibiotics like Cephradine. Patent CN101643477B introduces a transformative synthesis method that addresses long-standing stability and crystallinity issues inherent in previous manufacturing techniques. This innovation utilizes a carbonyldiimidazole-mediated activation strategy to couple dihydrophenyl glycine sodium salt with 7-aminodeacetoxycephalosporanic acid (7-ADCA). Unlike traditional enzymatic or complex chemical pathways that require stringent protection groups, this novel approach streamlines the condensation process significantly. By operating under controlled low-temperature conditions in anhydrous solvents, the method ensures the formation of an active amide intermediate that reacts efficiently without degrading the sensitive beta-lactam ring. For procurement and technical teams evaluating reliable Cephradine supplier options, this patent represents a significant leap forward in process reliability and product quality assurance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
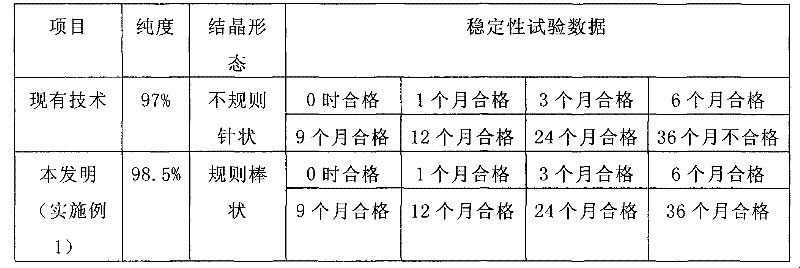
Historically, the industrial production of Cephradine has been plagued by the limitations of both enzymatic and traditional chemical synthesis routes. Enzymatic methods, while specific, rely on penicillin acylase which is prohibitively expensive and highly sensitive to reaction parameters such as pH, temperature, and ionic strength. Furthermore, enzyme activity is often inhibited by by-products, rendering these processes unsuitable for large-scale industrial consistency. On the chemical synthesis front, conventional methods necessitate the protection of the C4-carboxyl and C7-amino groups of 7-ADCA using organic bases or silane reagents like HMDS or TMCS. These protective groups inevitably introduce impurities into the hydrolysis solution during the final deprotection stages. Consequently, the resulting product often exhibits irregular needle-shaped crystals with poor stability, leading to significant challenges in filtration, drying, and long-term storage for high-purity antibiotic intermediates.
The Novel Approach
The methodology disclosed in CN101643477B fundamentally reengineers the synthesis pathway by eliminating the need for preliminary protection of the 7-ADCA substrate. Instead of relying on costly enzymes or cumbersome silane protection, the process activates dihydrophenyl glycine sodium salt directly using carbonyldiimidazole (CDI) in an anhydrous environment. This activation creates a highly reactive amide species that couples seamlessly with 7-ADCA at temperatures ranging from -15°C to -25°C. By bypassing the introduction of extraneous organic bases and silica reagents, the hydrolysis solution remains exceptionally pure, facilitating the growth of uniform, regular rod-shaped crystals. This structural integrity is crucial for cost reduction in antibiotic manufacturing, as it drastically improves downstream processing efficiency and ensures the final API meets rigorous pharmacopeial standards for stability and purity without extensive recrystallization.
Mechanistic Insights into CDI-Mediated Amide Activation
The core chemical innovation lies in the precise activation of the amino acid side chain using N,N'-carbonyldiimidazole. In the first stage, dihydrophenyl glycine sodium salt reacts with CDI in solvents such as methylene dichloride or DMF at temperatures between -20°C and -30°C. This reaction generates an active acyl imidazole intermediate, which is significantly more stable and selective than the mixed acid anhydrides or acyl chlorides used in older protocols. The imidazole leaving group is non-nucleophilic and easily removed, minimizing side reactions that could lead to polymerization or ring-opening of the beta-lactam core. This mechanistic precision ensures that when the 7-ADCA is introduced, the nucleophilic attack occurs exclusively at the desired amino position, preserving the stereochemical integrity of the molecule. Such control is vital for maintaining the biological activity of the final Cephradine product and reducing the burden on purification systems.
Furthermore, the absence of protecting groups on the 7-ADCA nucleus plays a pivotal role in the crystallization mechanism. In traditional routes, residual silanes or amines from deprotection steps interfere with crystal lattice formation, resulting in irregular needles that trap solvent and impurities. In contrast, the clean reaction profile of this novel method allows for the formation of dense, regular rod-shaped crystals. As demonstrated in the comparative stability data, this morphological advantage translates directly into enhanced physical stability. The product maintains its quality specifications for over 36 months, whereas conventional products often degrade or fail stability tests within the same timeframe. This mechanistic clarity provides R&D directors with confidence in the reproducibility and robustness of the synthesis route for commercial scale-up of complex beta-lactams.
How to Synthesize Cephradine Efficiently
The synthesis protocol outlined in the patent offers a clear, step-by-step framework for producing high-quality Cephradine suitable for immediate industrial adoption. The process begins with the preparation of the active amide followed by direct condensation, hydrolysis, and a carefully controlled crystallization sequence using triethylamine to adjust pH levels. This streamlined workflow eliminates unit operations associated with protection and deprotection, thereby reducing the overall cycle time and equipment footprint. For technical teams looking to implement this route, the detailed standardized synthesis steps provided below offer a comprehensive guide to replicating these results in a pilot or production setting, ensuring consistent batch-to-batch quality.
- React dihydrophenyl glycine sodium salt with carbonyldiimidazole in an anhydrous solvent at low temperatures (-20 to -30°C) to form the active amide intermediate.
- Condense the active amide directly with 7-ADCA without prior protection of the C4-carboxyl or C7-amino groups.
- Perform hydrolysis and controlled crystallization using triethylamine to obtain regular rod-shaped Cephradine crystals with high stability.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this synthesis method offers substantial strategic benefits beyond mere technical superiority. The elimination of expensive enzymatic catalysts and specialized silane reagents directly impacts the bill of materials, leading to significant cost optimization in the production of antibiotic intermediates. Moreover, the simplified process flow reduces the number of reaction vessels and separation units required, which lowers capital expenditure and operational overheads. For supply chain heads focused on continuity, the robustness of this chemical route means less susceptibility to raw material shortages associated with niche biocatalysts. The ability to produce a stable product with a prolonged shelf life also enhances inventory management capabilities, reducing the risk of write-offs due to expiration.
- Cost Reduction in Manufacturing: The removal of protection and deprotection steps eliminates the need for costly silane reagents and organic bases, while also reducing solvent consumption and waste treatment volumes. This streamlined chemistry inherently lowers the variable cost per kilogram of production, allowing for more competitive pricing structures in the global API market without compromising on quality margins.
- Enhanced Supply Chain Reliability: By shifting away from enzyme-dependent processes which are sensitive to supply fluctuations and storage conditions, manufacturers can secure a more stable supply of raw materials. The use of commodity chemicals like CDI and standard organic solvents ensures that production schedules are not disrupted by the availability of specialized biocatalysts, thereby reducing lead time for high-purity antibiotic intermediates.
- Scalability and Environmental Compliance: The process generates fewer by-products and avoids the discharge of silicon-containing waste streams, simplifying environmental compliance and wastewater treatment. The regular crystal morphology improves filtration rates and drying efficiency, facilitating easier scale-up from pilot batches to multi-ton commercial production runs while adhering to stricter green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and experimental data, providing a factual basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: How does this synthesis method improve Cephradine stability compared to traditional methods?
A: By eliminating the use of organic bases and silane reagents for protecting groups, this method prevents impurities from entering the hydrolysis solution, resulting in regular rod-shaped crystals that remain stable for over 36 months.
Q: What are the key advantages of using Carbonyldiimidazole (CDI) in this process?
A: CDI allows for the direct activation of dihydrophenyl glycine sodium salt under mild conditions, avoiding the harsh reagents like phosphorus pentachloride used in older methods, which simplifies purification and improves yield.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process avoids expensive enzymatic catalysts and complex protection/deprotection steps, making it highly scalable and cost-effective for commercial manufacturing of antibiotic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephradine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the CDI-mediated Cephradine synthesis can be seamlessly transitioned from the laboratory to full-scale manufacturing. We are committed to delivering products with stringent purity specifications and supporting our partners with rigorous QC labs that validate every batch against the highest international standards. Our infrastructure is designed to handle complex beta-lactam chemistry with the utmost safety and efficiency.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this superior method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs, ensuring a partnership built on transparency, quality, and mutual growth.
