Revolutionizing Acid Chloride Production: A Deep Dive into Catalytic Hydrolysis for Commercial Scale-Up
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more efficient, selective, and scalable synthetic routes. A pivotal advancement in this domain is documented in patent CN1109876A, which discloses a sophisticated process for the preparation of acid chloride compounds. This technology represents a significant departure from traditional stoichiometric methods, leveraging the power of Lewis acid catalysis to achieve high selectivity in the hydrolysis of trichloromethyl or gem-dichloro precursors. For R&D directors and procurement specialists alike, understanding this mechanism is crucial, as it offers a pathway to high-purity intermediates essential for the synthesis of complex pharmaceuticals and agrochemicals. The core innovation lies in the specific interplay between a transition metal halide catalyst, such as Ferric Chloride (FeCl3), and a carefully selected chlorocarbon solvent system.
By utilizing this catalytic approach, manufacturers can bypass the harsh conditions and waste generation associated with older thionyl chloride or oxalyl chloride protocols. The patent explicitly highlights the ability to convert compounds of general formula Ar-C(L1)2Cl directly into the corresponding acid chlorides with remarkable efficiency. This capability is not merely a laboratory curiosity but a robust industrial solution that addresses critical pain points in supply chain reliability and cost structure. As we delve deeper into the technical specifics, it becomes evident that this methodology provides a reliable pharmaceutical intermediates supplier with the tools necessary to deliver consistent quality at scale, ensuring that downstream processes, such as amide bond formation, proceed with minimal interruption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acid chlorides from methyl-substituted aromatics or related precursors has been fraught with challenges regarding selectivity and waste management. Traditional methods often rely on stoichiometric amounts of chlorinating agents or involve multi-step sequences that generate significant quantities of hazardous byproducts. For instance, prior art such as J.C.S. Chem. Comm. (1977) describes methods involving hexamethyldisiloxane which produce trimethylchlorosilane, necessitating complex recycling loops to regenerate the siloxane reagent. Furthermore, uncontrolled hydrolysis of trichloromethyl groups frequently leads to the formation of carboxylic acids rather than the desired acid chlorides, drastically reducing yield and complicating purification. These inefficiencies translate directly into higher operational costs and extended lead times, creating bottlenecks for companies seeking cost reduction in pharmaceutical intermediates manufacturing. The reliance on stoichiometric reagents also imposes a heavy burden on waste treatment facilities, conflicting with modern environmental compliance standards.
The Novel Approach
In stark contrast, the methodology outlined in CN1109876A introduces a catalytic cycle that fundamentally alters the reaction thermodynamics and kinetics. By employing a weak Lewis acid, preferably a transition metal halide like FeCl3, in conjunction with a specific chlorocarbon solvent, the process directs the hydrolysis exclusively towards the acid chloride functionality. The use of solvents like 1,2-dichloroethane is particularly ingenious; their low solubility in water allows for the precise, dropwise addition of water via a Dean-Stark apparatus. This controlled hydration prevents the over-hydrolysis that typically plagues such reactions. The result is a streamlined process where the catalyst can be used in sub-stoichiometric amounts (molar ratio less than 0.1), significantly lowering material costs. This novel approach not only enhances the atomic economy of the reaction but also simplifies the downstream workup, as the catalyst can often be removed by simple filtration, leaving a clean product stream ready for immediate conversion into high-value derivatives.
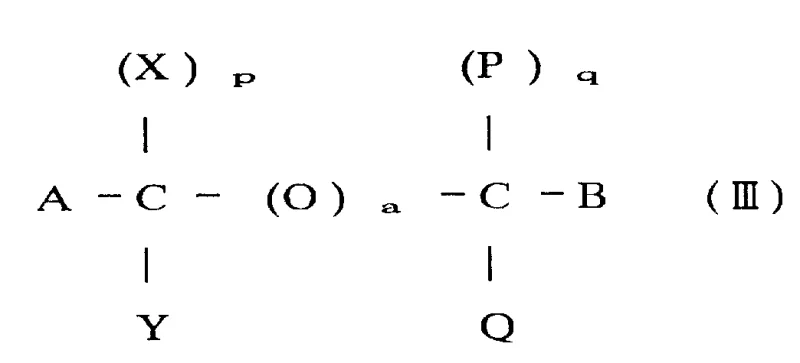
Mechanistic Insights into FeCl3-Catalyzed Hydrolysis
To fully appreciate the value of this technology for a reliable pharmaceutical intermediates supplier, one must understand the mechanistic underpinnings that drive its success. The reaction is believed to proceed through a transition state where the chlorine atoms of the chlorocarbon solvent play a active role, potentially coordinating with the Lewis acid to stabilize the developing positive charge on the carbonyl carbon during the hydrolysis event. The Lewis acid, specifically anhydrous Ferric Chloride, acts as a potent activator for the C-Cl bonds in the trichloromethyl precursor, making them susceptible to nucleophilic attack by water molecules. However, the key to success is the solvent environment. The chlorocarbon solvent, possessing at least two carbon atoms with adjacent chlorine atoms, creates a microenvironment that favors the retention of the acid chloride group over its hydrolysis to the acid. This delicate balance ensures that even with the presence of water, the equilibrium shifts decisively towards the formation of the acid chloride (Formula I), rather than the carboxylic acid.
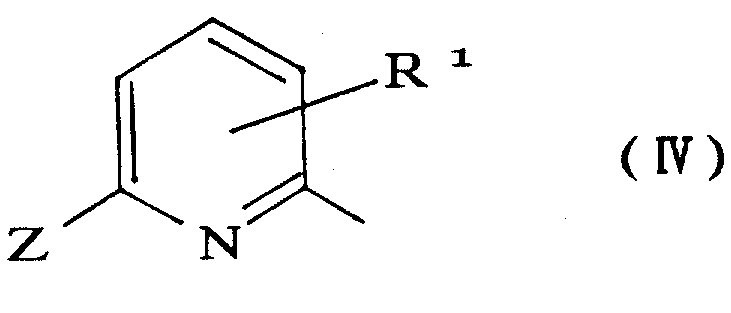
Furthermore, the impurity profile of the resulting product is exceptionally clean, a critical factor for R&D teams focusing on high-purity OLED material or API precursors. The mechanism inherently suppresses side reactions such as ring chlorination or polymerization, which are common in harsher acidic environments. For example, when synthesizing heteroaryl acid chlorides, such as the pyridine derivatives shown in the patent, the mildness of the catalytic system preserves sensitive functional groups on the aromatic ring. This selectivity is paramount when dealing with complex substrates where multiple reactive sites might exist. The ability to tune the Lewis acid strength and the solvent polarity allows chemists to optimize the reaction for specific substrates, ensuring that the commercial scale-up of complex pharmaceutical intermediates proceeds without unexpected deviations in quality or yield.
How to Synthesize 2-Chloro-6-Pyridinecarbonyl Chloride Efficiently
The practical implementation of this patented technology is best illustrated through the synthesis of key intermediates like 2-chloro-6-pyridinecarbonyl chloride, a vital building block for various herbicides and pharmaceuticals. The process begins by charging a reactor with the trichloromethyl precursor, such as Nitrapyrin, and a catalytic quantity of anhydrous FeCl3 in 1,2-dichloroethane. The mixture is brought to reflux, establishing the thermal energy required for the catalytic cycle to initiate. Water is then introduced slowly, often using a Dean-Stark trap to manage the azeotropic removal of water and maintain the precise stoichiometry needed for mono-hydrolysis. This careful control is what differentiates a lab-scale experiment from a robust manufacturing process. Once the hydrolysis is complete, indicated by the exhaustion of water in the trap, the reaction mixture contains the desired acid chloride in high concentration, ready for the next step.
- Combine the trichloromethyl compound (e.g., Nitrapyrin) with a catalytic amount of anhydrous Ferric Chloride (FeCl3) in a chlorocarbon solvent like 1,2-dichloroethane.
- Reflux the mixture and slowly introduce water using a Dean-Stark apparatus to ensure controlled hydrolysis without forming carboxylic acids.
- Upon completion, filter off the Lewis acid catalyst and either isolate the acid chloride or proceed directly to amidation with an amine substrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the implications of adopting this catalytic hydrolysis technology extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for stoichiometric chlorinating agents like thionyl chloride, which are subject to volatile pricing and strict regulatory controls, manufacturers can achieve significant cost reduction in agrochemical intermediates manufacturing. The use of catalytic amounts of iron salts, which are inexpensive and readily available, replaces costly reagents, directly improving the gross margin of the final product. Moreover, the simplified workup procedure, which often involves mere filtration to remove the catalyst, reduces the consumption of solvents and energy associated with extensive washing and distillation steps. This efficiency translates into shorter batch cycles and increased throughput, allowing suppliers to respond more agilely to market demands.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the shift from stoichiometric to catalytic reagents. Traditional methods often require large excesses of chlorinating agents, generating equimolar amounts of corrosive waste gases like HCl and SO2. In contrast, the FeCl3-catalyzed hydrolysis uses water as the oxygen source and generates HCl as the primary byproduct, which can be easily scrubbed or recovered. The elimination of expensive siloxane reagents or specialized chlorinating agents removes a major cost center. Additionally, the ability to recycle the chlorocarbon solvent, such as 1,2-dichloroethane, further drives down the variable cost per kilogram. This lean manufacturing approach ensures that the final high-purity acid chloride is produced at a competitive price point without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is heavily dependent on the availability and stability of raw materials. The reagents required for this process—water, iron chloride, and common chlorinated solvents—are commodity chemicals with robust global supply networks. Unlike specialized reagents that may face shortages or long lead times, these inputs are consistently available, reducing lead time for high-purity intermediates. Furthermore, the robustness of the reaction conditions (reflux in standard solvents) means that the process is less susceptible to minor fluctuations in temperature or pressure, ensuring consistent batch-to-batch reproducibility. This reliability is critical for long-term contracts with multinational corporations that demand uninterrupted supply of critical intermediates for their own production lines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and waste management. This catalytic hydrolysis method is inherently scalable because it operates under relatively mild conditions compared to high-pressure carbonylation or cryogenic chlorination. The use of a Dean-Stark apparatus for water management is a unit operation well-understood in pilot and plant-scale reactors. From an environmental perspective, the process aligns with green chemistry principles by minimizing waste generation. The absence of heavy metal contaminants in the final product (due to easy filtration of the FeCl3) reduces the burden on wastewater treatment plants. This compliance with stringent environmental regulations facilitates smoother permitting processes and reduces the risk of production shutdowns due to regulatory non-compliance, securing the long-term viability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this acid chloride synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN1109876A, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the balance between theoretical potential and practical execution observed in the patent examples.
Q: Why is 1,2-dichloroethane preferred as the solvent in this acid chloride synthesis?
A: According to patent CN1109876A, 1,2-dichloroethane offers unique advantages in selectivity. Its low water solubility (approx. 0.15g/100g at 25°C) allows for precise water management via Dean-Stark distillation, preventing over-hydrolysis to carboxylic acids and ensuring high yields of the desired acid chloride.
Q: How is the Lewis acid catalyst removed after the reaction?
A: The process utilizes heterogeneous or easily precipitable Lewis acids like Ferric Chloride (FeCl3). Post-reaction, the catalyst can be simply removed by filtration, such as through a Hyflo filter bed, simplifying the workup and reducing heavy metal contamination in the final product.
Q: What purity levels can be achieved with this hydrolysis method?
A: The patent data demonstrates exceptional purity control. In Example 2, the subsequent amide product derived from the acid chloride intermediate achieved a gas chromatography purity of 99.9%, indicating that the catalytic hydrolysis route minimizes side reactions and impurity formation effectively.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Acid Chloride Supplier
The technological advancements detailed in CN1109876A underscore the complexity and sophistication required to produce high-quality acid chloride intermediates efficiently. At NINGBO INNO PHARMCHEM, we recognize that translating such patented methodologies into commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of catalytic hydrolysis are fully realized at an industrial scale. We are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of acid chloride or downstream amide meets the exacting standards of the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Whether you require custom synthesis of complex heteroaryl acid chlorides or large-scale production of commodity intermediates, our technical team is ready to assist. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce your overall manufacturing costs.
