Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Drug Discovery
Scalable Metal-Free Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazoles for Advanced Drug Discovery
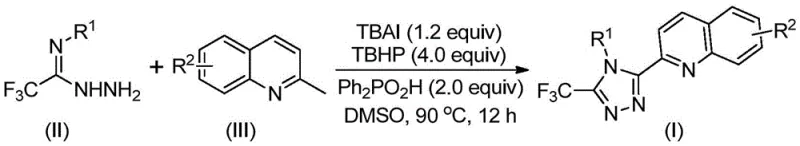
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds that serve as critical building blocks in active pharmaceutical ingredients (APIs). Patent CN113307790B introduces a groundbreaking preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing long-standing inefficiencies in heterocyclic synthesis. This technology leverages a metal-free oxidative cyclization strategy that not only simplifies the operational workflow but also significantly enhances the economic viability of producing these high-value intermediates. By utilizing readily available starting materials such as 2-methylquinoline and trifluoroacetimidoyl hydrazide, the process circumvents the need for complex multi-step sequences traditionally associated with quinoline functionalization. For R&D directors and process chemists, this represents a pivotal shift towards greener, more atom-economical methodologies that align with modern sustainability goals while maintaining rigorous purity standards required for downstream drug development applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficient multi-step protocols that rely on quinoline-2-carboxylic acid as a primary feedstock. These traditional pathways typically necessitate a cumbersome five-step reaction sequence to achieve the final heterocyclic core, resulting in a dismal overall yield of approximately 17%. Such low efficiency is compounded by the requirement for severe reaction conditions, including stringent temperature controls and the use of hazardous reagents that complicate waste management and safety protocols. Furthermore, the reliance on carboxylic acid derivatives often involves activation steps that generate stoichiometric amounts of byproducts, thereby increasing the burden on purification processes and driving up the cost of goods sold (COGS). For supply chain managers, these factors translate into longer lead times, higher raw material consumption, and increased vulnerability to supply disruptions for specialized precursors, making large-scale commercial production economically unfeasible for many organizations seeking reliable agrochemical intermediate suppliers.
The Novel Approach
In stark contrast, the methodology disclosed in CN113307790B revolutionizes the landscape by employing a direct oxidative coupling strategy that merges 2-methylquinoline and trifluoroacetimidoyl hydrazide in a single reaction vessel. This innovative route eliminates the need for pre-functionalized carboxylic acids, instead activating the methyl group of the quinoline ring in situ through a catalytic oxidation process. The reaction proceeds under relatively mild thermal conditions, typically between 80°C and 100°C, and crucially, does not require anhydrous or oxygen-free environments, which drastically reduces operational complexity. By avoiding the use of toxic heavy metal catalysts, the process not only mitigates environmental hazards but also simplifies the removal of metal residues, a critical quality attribute for pharmaceutical intermediates. This streamlined approach allows for the rapid generation of diverse libraries of triazole derivatives by simply varying the substituents on the aryl rings, offering unparalleled flexibility for structure-activity relationship (SAR) studies without compromising on scalability or cost reduction in electronic chemical manufacturing contexts.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this transformative synthesis lies in the synergistic interaction between tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), which acts as a potent oxidant to drive the formation of the triazole ring. Mechanistically, the reaction initiates with the oxidation of the methyl group on the 2-methylquinoline substrate to generate a reactive 2-quinolinecarbaldehyde intermediate. This aldehyde species then undergoes a condensation reaction with the trifluoroacetimidoyl hydrazide to form a dehydrated hydrazone intermediate, setting the stage for the subsequent cyclization event. The presence of diphenylphosphinic acid serves as a crucial additive that likely stabilizes the transition states or facilitates proton transfer during the ring-closing step. Following the formation of the hydrazone, the system undergoes oxidative iodination and intramolecular electrophilic substitution, ultimately leading to aromatization and the formation of the stable 1,2,4-triazole core. This cascade of reactions occurs seamlessly in polar aprotic solvents like DMSO, which effectively solubilizes both the organic substrates and the ionic catalyst species, ensuring high conversion rates and minimizing side reactions.
From an impurity control perspective, the metal-free nature of this catalytic system offers distinct advantages over transition-metal catalyzed alternatives. Traditional methods involving copper or palladium often leave behind trace metal contaminants that are difficult to remove and can catalyze degradation pathways in the final API. By utilizing an organic iodide catalyst, the impurity profile is significantly cleaner, consisting primarily of organic byproducts that are more easily separated via standard chromatographic techniques or crystallization. The tolerance of this system to various functional groups, including halogens, alkyls, and electron-donating methoxy groups, further demonstrates its robustness. For instance, substrates bearing electron-withdrawing groups like trifluoromethyl or nitro groups on the phenyl ring of the hydrazide component are well-tolerated, yielding the corresponding triazoles in high purity. This broad substrate scope ensures that the process is adaptable for the synthesis of complex molecular architectures required in next-generation therapeutics, providing a reliable foundation for commercial scale-up of complex polymer additives or pharmaceutical intermediates.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and reproducibility. The protocol dictates a molar ratio where the oxidant (TBHP) is used in excess relative to the substrate to ensure complete conversion of the methyl group, while the iodide catalyst is employed in sub-stoichiometric or catalytic amounts to drive the cycle. The choice of solvent is critical, with dimethyl sulfoxide (DMSO) identified as the optimal medium due to its ability to stabilize the polar intermediates and facilitate the oxidative process. Operators should monitor the reaction progress via TLC or HPLC, typically observing completion within an 8 to 14-hour window depending on the specific electronic nature of the substituents. Once the reaction is deemed complete, the workup procedure is remarkably straightforward, involving simple filtration followed by silica gel treatment and column chromatography, bypassing the need for complex aqueous extractions or distillation steps often required in older methodologies.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl hydroperoxide (TBHP), diphenylphosphinic acid, trifluoroacetimidoyl hydrazide, and 2-methylquinoline in DMSO solvent.
- Heat the reaction mixture to 80-100°C and stir for 8-14 hours to facilitate oxidative cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits that extend beyond mere technical feasibility. The elimination of precious metal catalysts removes a significant cost driver from the bill of materials, as well as the associated costs of metal scavenging and validation testing required for regulatory compliance. Furthermore, the use of commodity chemicals like 2-methylquinoline and TBHP ensures a stable and resilient supply chain, reducing the risk of bottlenecks associated with specialized reagents. The simplicity of the workup procedure also translates to reduced processing time and lower energy consumption, contributing to a smaller carbon footprint and enhanced sustainability metrics for the manufacturing site. These factors collectively position this technology as a highly attractive option for organizations seeking cost reduction in pharmaceutical intermediate manufacturing while maintaining high standards of quality and safety.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the replacement of expensive transition metal catalysts with inexpensive organic salts like tetrabutylammonium iodide. This shift not only lowers the direct material costs but also eliminates the downstream expenses associated with removing trace metals to meet strict pharmacopeial limits. Additionally, the high atom economy of the one-pot reaction minimizes waste generation, reducing the costs related to waste disposal and solvent recovery. The ability to achieve high yields in a single step compared to the traditional five-step sequence dramatically improves the overall process mass intensity (PMI), leading to significant savings in raw material usage and operational overheads.
- Enhanced Supply Chain Reliability: By relying on widely available bulk chemicals such as 2-methylquinoline and tert-butyl hydroperoxide, manufacturers can mitigate the risks associated with sourcing niche reagents that may have limited suppliers or long lead times. The robustness of the reaction conditions, which do not require inert atmospheres or ultra-dry solvents, further simplifies logistics and storage requirements, allowing for more flexible production scheduling. This reliability is crucial for maintaining continuous supply to downstream customers, especially in the volatile landscape of global chemical distribution where disruptions can have cascading effects on production timelines.
- Scalability and Environmental Compliance: The absence of heavy metals simplifies the regulatory pathway for commercial production, as there is no need for extensive validation of metal clearance procedures. The reaction's tolerance to water and oxygen makes it inherently safer and easier to scale from gram-scale laboratory experiments to multi-ton industrial reactors without significant re-engineering of the process equipment. Moreover, the use of DMSO, a solvent with favorable recycling properties, aligns with green chemistry principles, helping companies meet increasingly stringent environmental regulations and corporate sustainability goals without sacrificing productivity or yield.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios or for procurement specialists assessing the long-term value proposition of this synthetic approach.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method utilizes an organic catalytic system based on tetrabutylammonium iodide (TBAI) and tert-butyl hydroperoxide (TBHP), completely eliminating the need for toxic or costly heavy metal catalysts.
Q: What are the typical reaction conditions for this oxidative cyclization?
A: The reaction is typically conducted in DMSO at temperatures between 80°C and 100°C for a duration of 8 to 14 hours, without requiring strict anhydrous or anaerobic environments.
Q: How does this method compare to traditional quinoline-2-carboxylic acid routes?
A: Traditional routes often involve five steps with low overall yields (around 17%) and harsh conditions. This novel approach achieves high yields (up to 97% in optimized examples) in a single pot using cheap, commercially available starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced heterocyclic intermediates play in the development of next-generation therapeutics and agrochemicals. Our team of expert process chemists has extensively evaluated the methodology described in CN113307790B and confirmed its potential for robust commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of these valuable building blocks. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-quinolyl-5-trifluoromethyl-1,2,4-triazole delivered meets the exacting standards required for global pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in metal-free catalysis can drive efficiency and innovation in your supply chain.
