Advanced Synthesis of 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Derivatives for Pharmaceutical Applications
The landscape of heterocyclic chemistry is continuously evolving, driven by the demand for more efficient and environmentally benign synthetic routes for bioactive scaffolds. A pivotal advancement in this domain is detailed in patent CN113307790B, which discloses a robust preparation method for 3-quinolyl-5-trifluoromethyl substituted 1,2,4-triazole compounds. These structural motifs are not merely academic curiosities; they serve as critical building blocks in the development of next-generation therapeutics and functional materials. As illustrated in the broader context of medicinal chemistry, 1,2,4-triazole rings are ubiquitous in drug discovery, appearing in diverse pharmacological agents ranging from antifungal medications like fluconazole to kinase inhibitors such as sitagliptin. The ability to construct these cores efficiently, particularly when fused or linked with quinoline systems, opens new avenues for creating bidentate ligands and complex API intermediates. This patent represents a significant leap forward by replacing multi-step, low-yield sequences with a direct, oxidative cyclization strategy that operates under mild conditions.
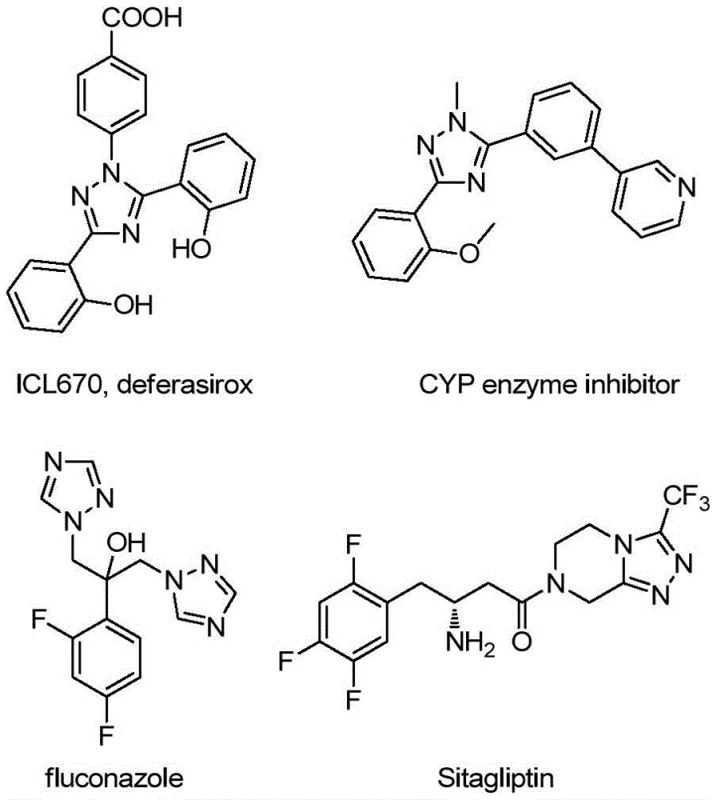
The strategic importance of this technology cannot be overstated for a reliable pharmaceutical intermediate supplier aiming to support the global supply chain. By leveraging a metal-free catalytic system, the process circumvents the stringent purification requirements associated with heavy metal removal, a common bottleneck in GMP manufacturing. The methodology utilizes readily available starting materials, specifically 2-methylquinoline derivatives and trifluoroacetohydrazides, reacting them in the presence of tetrabutylammonium iodide and tert-butyl peroxide. This approach not only simplifies the operational workflow but also enhances the economic viability of producing high-purity OLED material precursors and pharmaceutical intermediates. For R&D teams evaluating new pathways, the tolerance for diverse functional groups on both the quinoline and the hydrazide components suggests a versatile platform for library synthesis and lead optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolyl-substituted 1,2,4-triazoles has been plagued by inefficiencies that hinder large-scale production. Traditional protocols often rely on quinoline-2-carboxylic acid as the primary starting material, necessitating a cumbersome five-step reaction sequence to achieve the final target structure. This linear synthesis is characterized by severe reaction conditions that often require strict anhydrous and anaerobic environments, significantly increasing operational costs and safety risks. Furthermore, the cumulative yield of such multi-step processes is notoriously poor, with literature reporting total yields as low as 17%. From a supply chain perspective, this low atom economy translates to excessive waste generation and higher raw material consumption. The reliance on multiple isolation and purification steps between each transformation further extends the lead time for high-purity pharmaceutical intermediates, making it difficult to respond rapidly to market demands or clinical trial requirements. Additionally, the use of stoichiometric activating agents in older methods often generates substantial amounts of hazardous byproducts, complicating waste treatment and environmental compliance.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a paradigm shift through a direct oxidative cyclization reaction. By employing 2-methylquinoline and trifluoroacetohydrazide as the foundational building blocks, the process consolidates what was previously a multi-step ordeal into a single, streamlined transformation. The reaction proceeds efficiently at temperatures between 80°C and 100°C in common organic solvents like DMSO, eliminating the need for cryogenic conditions or inert gas manifolds. The use of a catalytic amount of tetrabutylammonium iodide combined with tert-butyl peroxide as the terminal oxidant facilitates the formation of the triazole ring with remarkable selectivity. This novel approach allows for the synthesis of diversified 1,2,4-triazole compounds bearing both quinolyl and trifluoromethyl groups with exceptional ease. The operational simplicity means that the reaction can be scaled from gram-level laboratory experiments to kilogram-level commercial production without fundamental changes to the equipment setup. Moreover, the absence of transition metal catalysts removes the regulatory burden of residual metal testing, a critical advantage for cost reduction in API manufacturing where purity specifications are increasingly stringent.
Mechanistic Insights into TBAI/TBHP Promoted Oxidative Cyclization
The core of this technological breakthrough lies in the unique mechanistic pathway facilitated by the TBAI/TBHP system. The reaction initiates with the oxidation of the methyl group on the 2-methylquinoline substrate. Under the influence of the iodide catalyst and the peroxide oxidant, the methyl group is converted into an aldehyde functionality in situ, generating a 2-quinolinecarbaldehyde intermediate. This reactive species then undergoes a condensation reaction with the trifluoroacetohydrazide to form a dehydrated hydrazone intermediate. Subsequently, the system promotes an oxidative iodination followed by an intramolecular electrophilic substitution. This cyclization step is crucial as it closes the five-membered triazole ring. Finally, an aromatization step yields the stable 3-quinolyl-5-trifluoromethyl-1,2,4-triazole product. Alternatively, the reaction may proceed via a radical mechanism, where radical species generated from the decomposition of TBHP abstract hydrogen atoms to drive the oxidation cascade. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters such as temperature and stoichiometry to maximize conversion rates.
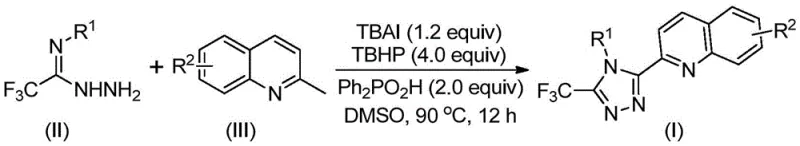
From an impurity control perspective, the mild nature of this oxidative system offers distinct advantages over harsher acidic or basic cyclization methods. The selectivity of the TBAI/TBHP combination minimizes the formation of over-oxidized byproducts or polymeric tars that often contaminate crude reaction mixtures in traditional syntheses. The use of diphenylphosphoric acid as an additive further modulates the reaction environment, likely stabilizing intermediates and promoting the desired cyclization pathway over competing side reactions. This results in a cleaner crude profile, which simplifies downstream purification. For quality control laboratories, this means fewer peaks to monitor and a more robust analytical method for release testing. The ability to tolerate various substituents, such as electron-withdrawing nitro groups or electron-donating methoxy groups, without significant loss of yield indicates that the electronic properties of the substrate do not drastically alter the mechanistic pathway, ensuring consistent product quality across a wide range of analogues. This mechanistic robustness is a key factor in establishing a reliable supply chain for complex heterocyclic intermediates.
How to Synthesize 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a production setting requires careful attention to reagent ratios and thermal management, although the procedure itself is remarkably straightforward. The patent provides a clear blueprint for executing the reaction, emphasizing the use of DMSO as the preferred solvent due to its superior ability to dissolve both the organic substrates and the ionic catalyst. The standard protocol involves mixing the hydrazide, the quinoline derivative, the iodide salt, the peroxide, and the phosphoric acid additive in the solvent before heating. Detailed standardized synthesis steps are provided below to guide process engineers in replicating these high-yielding results.
- Combine tetrabutylammonium iodide (TBAI), tert-butyl peroxide (TBHP) aqueous solution, diphenylphosphoric acid, trifluoroacetohydrazide, and 2-methylquinoline in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature range of 80-100°C and maintain stirring for 8 to 14 hours to ensure complete oxidative cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-quinolyl-5-trifluoromethyl-1,2,4-triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The elimination of precious metal catalysts such as palladium or copper fundamentally alters the cost structure of the manufacturing process. Without the need for expensive ligands and the subsequent scavenging resins required to remove metal residues, the overall cost of goods sold is significantly reduced. This cost reduction in pharmaceutical intermediate manufacturing is achieved not just through cheaper raw materials, but through the simplification of the entire production workflow. The reaction does not require specialized equipment for handling air-sensitive reagents, allowing it to be run in standard glass-lined reactors commonly found in multipurpose chemical plants. This flexibility enhances supply chain reliability by enabling production to be shifted between different facilities without extensive retrofitting.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is profound. By removing the dependency on transition metals, manufacturers avoid the volatility associated with the pricing of commodities like palladium. Furthermore, the high atom economy of the direct cyclization means that less raw material is wasted, leading to substantial cost savings on a per-kilogram basis. The simplified workup procedure, which often involves basic filtration and chromatography rather than complex extractions or distillations, reduces labor hours and solvent consumption. These factors combine to create a highly competitive cost profile for the final intermediate, allowing downstream drug developers to optimize their own budget allocations for clinical trials and commercial launches.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, namely 2-methylquinolines and trifluoroacetohydrazides, are commodity chemicals that are widely available from multiple global suppliers. This abundance mitigates the risk of supply disruptions that can occur with proprietary or niche reagents. The robustness of the reaction conditions, which tolerate moisture and oxygen to a reasonable degree compared to organometallic couplings, ensures that batch-to-batch variability is minimized. Consistent production schedules can be maintained even in facilities with varying levels of environmental control. This reliability is crucial for maintaining the continuity of supply for critical API intermediates, preventing costly delays in the drug development timeline.
- Scalability and Environmental Compliance: Scaling this reaction from the bench to the plant floor is facilitated by the use of common solvents and reagents that behave predictably at larger volumes. The absence of heavy metals simplifies the environmental permitting process, as wastewater treatment does not require specialized protocols for metal precipitation and disposal. The waste stream is primarily organic and can often be incinerated for energy recovery, aligning with modern green chemistry principles. This environmental compliance reduces the regulatory burden on the manufacturing site and lowers the long-term liability associated with hazardous waste storage. The ability to produce these complex heterocycles sustainably makes the supply chain more resilient against tightening environmental regulations globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative cyclization technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing portfolios.
Q: Does this synthesis method require toxic heavy metal catalysts?
A: No, the method described in patent CN113307790B utilizes a metal-free catalytic system involving tetrabutylammonium iodide (TBAI) and tert-butyl peroxide (TBHP), eliminating the need for expensive and toxic transition metals often found in traditional cross-coupling reactions.
Q: How does the yield compare to conventional quinoline-2-carboxylic acid routes?
A: Traditional methods using quinoline-2-carboxylic acid typically suffer from low total yields around 17% over five steps. In contrast, this novel oxidative cyclization approach achieves significantly higher yields, with specific examples demonstrating isolated yields up to 97% in a single operational step.
Q: What is the substrate scope for the aryl group on the hydrazide?
A: The process exhibits excellent functional group tolerance, successfully accommodating various substituents on the aryl ring including methyl, methoxy, halogens (fluorine, bromine), nitro, and trifluoromethyl groups at ortho, meta, or para positions without compromising reaction efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Quinolyl-5-Trifluoromethyl-1,2,4-Triazole Supplier
As the pharmaceutical industry continues to demand more sophisticated heterocyclic building blocks, the ability to deliver high-quality intermediates with speed and precision becomes a key differentiator. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in CN113307790B to serve our global clientele. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met whether you are in the early discovery phase or preparing for market launch. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify the identity and purity of every batch we produce.
We invite you to explore how our technical expertise can accelerate your drug development programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to optimize your supply chain without compromising quality. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in turning complex chemical challenges into commercial successes.
