Revolutionizing Taxane Synthesis: A Deep Dive into Selective C(10) Derivatization for Commercial Scale-up
Introduction to Advanced Taxane Modification Technologies
The landscape of anticancer drug manufacturing is continuously evolving, driven by the need for more efficient synthetic routes to complex molecules like paclitaxel and docetaxel. Patent CN1205196C introduces a groundbreaking methodology for the selective derivatization of taxanes, specifically targeting the C(10) and C(7) hydroxyl groups of 10-deacetylbaccatin III (10-DAB). Historically, the conversion of 10-DAB, a critical natural precursor extracted from yew needles, into active pharmaceutical ingredients has been bottlenecked by the similar reactivity of its multiple hydroxyl groups. This patent discloses a novel process that fundamentally shifts the paradigm by enabling the selective protection or derivatization of the C(10) hydroxyl group prior to the C(7) position. This inversion of conventional reactivity order offers profound implications for process chemistry, allowing for streamlined synthesis pathways that reduce waste and improve overall throughput. For industry stakeholders, understanding this technology is crucial for optimizing the supply chain of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for paclitaxel rely heavily on the observation that the C(7) hydroxyl group of 10-DAB is inherently more nucleophilic than the C(10) hydroxyl group. Consequently, standard protocols mandate the protection of the C(7) position, often using triethylsilyl chloride, before any manipulation at C(10) can occur. This sequential necessity introduces significant inefficiencies. The initial silylation step requires strict control to prevent over-reaction or protection of other hydroxyls, and the subsequent deprotection steps add to the step count. Furthermore, the conventional approach often suffers from moderate yields and the generation of difficult-to-separate impurities, which complicates downstream processing. For a procurement manager, these inefficiencies translate into higher raw material consumption and increased costs associated with solvent recovery and waste disposal, ultimately impacting the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The innovation detailed in CN1205196C circumvents these historical constraints by employing Lewis acid catalysts to reverse the natural reactivity order. By introducing catalysts such as zinc chloride (ZnCl2) or cerium trichloride (CeCl3), the process selectively activates the C(10) hydroxyl group for acylation or silylation, even in the presence of a free C(7) hydroxyl. This allows for the direct conversion of 10-DAB to Baccatin III or other C(10) protected derivatives in a single, highly efficient step. 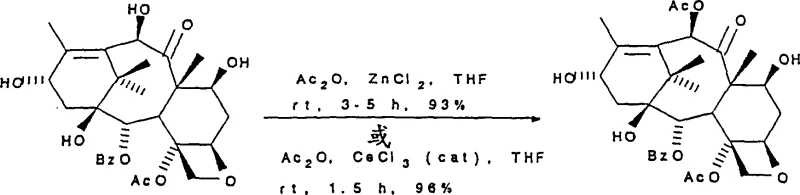 This shift eliminates the need for the preliminary C(7) protection step, drastically shortening the synthetic route. The ability to install diverse functional groups at C(10) using anhydrides, dicarbonates, or isocyanates under mild conditions provides chemists with unprecedented flexibility. This novel approach not only enhances the purity profile of the resulting intermediates but also significantly simplifies the operational workflow, making it an attractive option for reliable pharmaceutical intermediates supplier networks aiming to optimize their production capabilities.
This shift eliminates the need for the preliminary C(7) protection step, drastically shortening the synthetic route. The ability to install diverse functional groups at C(10) using anhydrides, dicarbonates, or isocyanates under mild conditions provides chemists with unprecedented flexibility. This novel approach not only enhances the purity profile of the resulting intermediates but also significantly simplifies the operational workflow, making it an attractive option for reliable pharmaceutical intermediates supplier networks aiming to optimize their production capabilities.
Mechanistic Insights into Lewis Acid-Catalyzed Selective Acylation
The core of this technological advancement lies in the mechanistic interaction between the Lewis acid catalyst and the taxane substrate. In the absence of a catalyst, the steric and electronic environment of the 10-DAB molecule favors attack at the C(7) position. However, when a Lewis acid like ZnCl2 is introduced, it coordinates with the carbonyl oxygen atoms of the existing ester groups on the taxane core, particularly the C(4) acetate and the C(2) benzoate. This coordination induces a conformational change or an electronic redistribution that effectively enhances the nucleophilicity of the C(10) hydroxyl group relative to the C(7) hydroxyl. Alternatively, the catalyst may coordinate directly with the C(7) hydroxyl, temporarily reducing its reactivity.  This subtle yet powerful modulation of reactivity allows acylating agents to preferentially react at the C(10) position. The patent data indicates that this selectivity is robust across a range of acylating agents, including acetic anhydride and dibenzyl dicarbonate. Furthermore, the use of silylamides in the presence of catalytic bases like LiHMDS offers a parallel pathway for selective C(10) silylation. Understanding these mechanistic nuances is vital for R&D directors focused on impurity control, as the high selectivity minimizes the formation of regioisomers that are notoriously difficult to remove, thereby ensuring stringent purity specifications are met with less rigorous purification efforts.
This subtle yet powerful modulation of reactivity allows acylating agents to preferentially react at the C(10) position. The patent data indicates that this selectivity is robust across a range of acylating agents, including acetic anhydride and dibenzyl dicarbonate. Furthermore, the use of silylamides in the presence of catalytic bases like LiHMDS offers a parallel pathway for selective C(10) silylation. Understanding these mechanistic nuances is vital for R&D directors focused on impurity control, as the high selectivity minimizes the formation of regioisomers that are notoriously difficult to remove, thereby ensuring stringent purity specifications are met with less rigorous purification efforts.
How to Synthesize C(10) Acylated Taxanes Efficiently
Implementing this selective derivatization process requires precise control over reaction parameters to maximize yield and selectivity. The general procedure involves dissolving 10-DAB in an ether solvent such as tetrahydrofuran (THF) and adding the Lewis acid catalyst. The choice of catalyst loading can vary from stoichiometric to catalytic amounts depending on the specific acylating agent used. Once the catalyst is integrated, the acylating agent is introduced, and the mixture is stirred at room temperature or slightly elevated temperatures. This straightforward protocol avoids the need for cryogenic conditions often associated with sensitive organometallic reactions, facilitating easier handling and safer operations in a manufacturing environment. The workup typically involves simple aqueous washes and chromatographic purification, yielding the desired C(10) derivative in high purity.
- Prepare a reaction mixture containing 10-DAB and a stoichiometric or catalytic amount of a Lewis acid such as ZnCl2 or CeCl3 in an ether solvent like THF.
- Add the acylating agent, such as acetic anhydride or dibenzyl dicarbonate, to the mixture while maintaining the temperature at room temperature or slightly elevated conditions.
- Stir the reaction until completion as monitored by TLC, then quench with aqueous bicarbonate, extract with ethyl acetate, and purify the C(10) acylated product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this C(10) selective derivatization technology offers tangible strategic benefits beyond mere chemical elegance. The primary advantage is the significant simplification of the synthetic route. By removing the mandatory C(7) protection step required in conventional methods, the total number of unit operations is reduced. This reduction directly correlates to lower operational expenditures, as fewer reactors are tied up for shorter periods, and less labor is required for monitoring and handling. Additionally, the high yields reported in the patent examples, often exceeding 90%, mean that less starting material (10-DAB) is wasted. Since 10-DAB is a natural product with supply constraints, maximizing atom economy is critical for ensuring supply continuity and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of the initial C(7) silylation step removes the cost associated with silylating reagents and the subsequent removal of those protecting groups. Furthermore, the use of inexpensive and readily available Lewis acids like zinc chloride or cerium chloride replaces more exotic or expensive catalysts. This shift in reagent profile leads to substantial cost savings in raw material procurement. The simplified purification process also reduces the volume of solvents and silica gel required for chromatography, further driving down the cost of goods sold (COGS) without compromising quality.
- Enhanced Supply Chain Reliability: Dependence on complex multi-step syntheses increases the risk of supply disruptions due to failure at any single stage. By shortening the synthesis of key intermediates like Baccatin III, the overall process robustness is improved. The ability to source C(10) protected intermediates through a more direct route diversifies the supply base and mitigates risks associated with the volatility of natural product extraction. This reliability is essential for maintaining consistent production schedules for final API manufacturing, ensuring that downstream partners receive materials on time.
- Scalability and Environmental Compliance: The reaction conditions described, primarily utilizing THF at ambient temperatures, are highly amenable to scale-up from kilogram to metric ton scales. The avoidance of extreme temperatures or pressures simplifies engineering requirements for large-scale reactors. Moreover, the higher selectivity results in a cleaner crude product, which reduces the burden on waste treatment facilities. Less hazardous waste is generated per kilogram of product, aligning with increasingly stringent environmental regulations and supporting sustainability goals within the fine chemical industry.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding this patent technology, we have compiled answers based on the experimental data and claims provided in CN1205196C. These insights are intended to clarify the practical application of the selective derivatization method for potential partners and licensees. Understanding these details helps in assessing the feasibility of integrating this chemistry into existing production lines or new process development projects.
Q: How does this process reverse the conventional reactivity of 10-DAB hydroxyl groups?
A: Conventionally, the C(7) hydroxyl is more reactive than C(10). This patent demonstrates that using specific Lewis acids like ZnCl2 or CeCl3 coordinates with the carbonyl oxygens, effectively enhancing the nucleophilicity of the C(10) position or sterically hindering C(7), allowing for selective C(10) derivatization without prior C(7) protection.
Q: What are the primary commercial benefits of this C(10) first strategy?
A: By protecting C(10) first, the subsequent protection of C(7) becomes significantly more selective and efficient. This reduces the number of purification steps, minimizes the formation of di-protected byproducts, and improves the overall yield of key intermediates like Baccatin III, leading to substantial cost savings in large-scale manufacturing.
Q: Is this method scalable for industrial production of paclitaxel intermediates?
A: Yes, the method utilizes common solvents like THF and commercially available Lewis acid catalysts at room temperature. The absence of cryogenic conditions and the high selectivity reported (often exceeding 90% yield) make it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 10-DAB Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the selective C(10) derivatization technology disclosed in CN1205196C. As a leading CDMO and supplier in the fine chemical sector, we possess the technical expertise to adapt and optimize these patented routes for commercial production. Our facilities are equipped to handle complex organic syntheses, ensuring that we can deliver high-purity taxane intermediates that meet the rigorous demands of the global pharmaceutical market. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to maintain stringent purity specifications for every batch we release.
We invite international partners to collaborate with us to leverage this advanced chemistry for your supply chain needs. Whether you require custom synthesis of specific C(10) acylated taxanes or large-scale production of Baccatin III, our team is ready to provide a Customized Cost-Saving Analysis tailored to your project. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive edge in the anticancer drug market.
