Advanced Synthesis of Sulfentrazone Intermediates for Commercial Scale-Up
Advanced Synthesis of Sulfentrazone Intermediates for Commercial Scale-Up
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical agrochemical intermediates. Patent CN114044758A introduces a transformative synthetic methodology for producing 4,5-dihydro-3-methyl-1-(2,4-dichloro-5-aminophenyl)-4-difluoromethyl-1,2,4-triazole-5(1H)-one, a pivotal precursor for the triazolinone herbicide sulfentrazone. This technical disclosure addresses long-standing stability issues associated with the N-difluoromethyl substituted triazolinone ring, which has historically plagued manufacturing processes due to its susceptibility to decomposition under harsh reaction conditions. By fundamentally reordering the synthetic sequence, this approach ensures that the sensitive heterocyclic ring is constructed only after the benzene ring functionalization is complete. This strategic shift not only mitigates the risk of structural degradation but also opens new avenues for optimizing reaction conditions without the constraint of protecting a fragile moiety. For R&D teams and procurement specialists alike, understanding this mechanistic evolution is crucial for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality at scale.
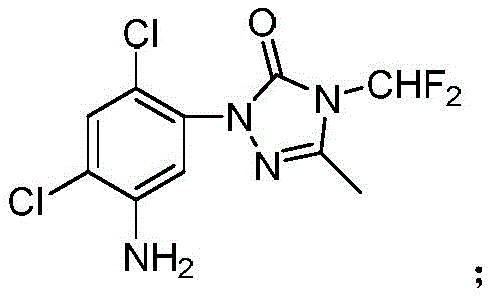
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this key intermediate involved constructing the triazolinone ring early in the process, followed by the nitration and reduction of the benzene ring. This traditional pathway, as illustrated in prior art literature, forces the N-difluoromethyl substituted triazolinone ring to endure aggressive mixed acid nitration environments involving concentrated sulfuric and nitric acids. Under these severe conditions, the large cyclic structural group creates significant steric and electronic instability, leading to decomposition or destruction of the core scaffold. Furthermore, the nitration step in the presence of the pre-formed ring often generates a complex mixture of nitrated isomers and dinitrated byproducts, complicating downstream purification and drastically lowering the overall yield. Reports indicate that total reaction yields using these conventional methods rarely exceed 36.8%, with some pathways performing even lower at approximately 31.0%. This inefficiency translates directly into higher production costs and inconsistent supply availability, posing a significant bottleneck for large-scale herbicide manufacturing.
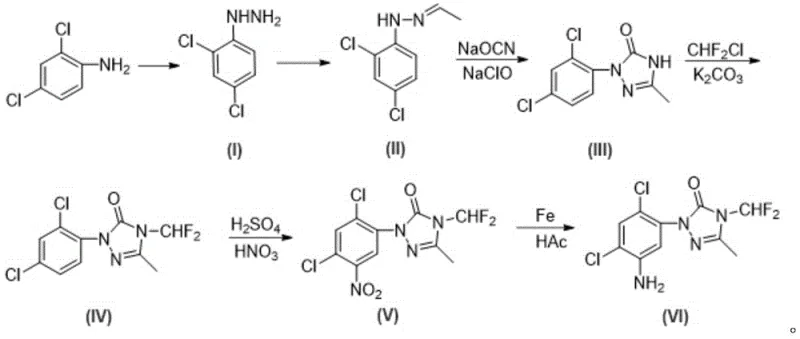
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent employs a "protect-first, cyclize-later" strategy that fundamentally alters the reaction landscape. Instead of subjecting the sensitive triazolinone ring to mixed acid, the process begins with the nitration of the starting material, 2,4-dichloroaniline, followed immediately by acetyl protection of the amino group. This ensures that all harsh electrophilic aromatic substitutions and subsequent reductions occur on a robust aniline derivative rather than a fragile heterocycle. Only after the benzene ring is fully functionalized with the necessary amino group is the triazolinone ring constructed via hydrazine formation and cyclization. Crucially, the difluoromethyl group is introduced at a very late stage, just prior to the final deprotection step. This sequencing effectively isolates the unstable N-difluoromethyl moiety from the harshest reagents in the synthesis, thereby preserving the structural integrity of the molecule and significantly improving the purity profile of the crude product. This approach represents a paradigm shift in cost reduction in herbicide manufacturing by minimizing waste and maximizing throughput.
Mechanistic Insights into Late-Stage Difluoromethylation and DPPA Cyclization
The core of this synthetic advancement lies in the precise orchestration of the cyclization and alkylation steps. Following the formation of the hydrazine intermediate, the process utilizes diphenylphosphoryl azide (DPPA) to effect cyclization in the presence of a base such as triethylamine. This step proceeds through a Curtius-type rearrangement mechanism or direct nucleophilic attack to close the 1,2,4-triazole-5-one ring efficiently. The use of DPPA allows for mild reaction conditions compared to traditional phosgene-based cyclizations, further enhancing safety and scalability. Once the triazolinone scaffold is established, the N-difluoromethylation is performed using chlorodifluoromethane gas in the presence of a phase transfer catalyst like tetrabutylammonium bromide. Performing this alkylation on the already cyclized but still acetyl-protected intermediate ensures that the electron-deficient nitrogen is available for nucleophilic attack without interference from the free aniline amino group. This specific ordering prevents side reactions that could occur if the free amine were present during the alkylation phase, ensuring high regioselectivity for the N4 position of the triazole ring.
Impurity control is inherently built into this mechanistic design. By avoiding the exposure of the triazolinone ring to mixed acid, the formation of ring-opened byproducts and fluorine-loss degradation products is virtually eliminated. In conventional routes, the acidic environment can protonate the ring nitrogen, facilitating the loss of the difluoromethyl group as HF or other fluorinated species. The new route circumvents this by keeping the ring neutral and protected during the acidic nitration phase. Additionally, the acetyl protection group on the aniline nitrogen serves a dual purpose: it directs the nitration to the desired 5-position through steric and electronic effects, and it prevents the aniline from participating in unwanted side reactions during the hydrazine formation and cyclization steps. The final hydrolysis step removes this acetyl group cleanly under mild acidic conditions, yielding the target amine with minimal contamination from over-nitrated or reduced impurities, thus simplifying the crystallization process and enhancing the final assay value.
How to Synthesize 4,5-dihydro-3-methyl-1-(2,4-dichloro-5-aminophenyl)-4-difluoromethyl-1,2,4-triazole-5(1H)-one Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing temperature control and stoichiometric precision to maximize yield. The process begins with the careful nitration of 2,4-dichloroaniline at low temperatures to prevent polynitration, followed by acetylation to lock in the substitution pattern. Subsequent reduction of the nitro group to an amine, followed by diazotization and reduction to the hydrazine, requires strict pH control to ensure complete conversion without decomposing the sensitive hydrazine intermediate. The cyclization step using DPPA must be conducted under reflux in a non-polar solvent like toluene to drive the equilibrium towards ring closure. Finally, the introduction of the difluoromethyl group requires a sealed system or continuous gas flow setup to handle the gaseous reagent safely, followed by a final hydrolysis to reveal the active pharmaceutical ingredient precursor. Detailed standardized synthesis steps are provided in the guide below for technical reference.
- Perform nitration on 2,4-dichloroaniline followed by acetyl protection to stabilize the amine group before ring formation.
- Reduce the nitro group and convert to hydrazine, then react with pyruvic acid to form the hydrazone precursor.
- Execute DPPA-mediated cyclization to form the triazolinone ring, followed by late-stage difluoromethylation and hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the purification train. Because the new method avoids the generation of complex nitrated isomers and ring-degradation byproducts, the number of recrystallization steps required to meet specification is significantly reduced. This reduction in downstream processing time translates directly into shorter batch cycles and increased facility throughput. Furthermore, the elimination of harsh conditions acting on the final ring structure means that equipment corrosion is minimized, leading to lower maintenance costs and extended reactor lifespan. The stability of the intermediates throughout the process also reduces the risk of batch failures, ensuring a more predictable and reliable supply of the critical intermediate for downstream herbicide formulation.
- Cost Reduction in Manufacturing: The improved stability of the intermediates allows for the use of more cost-effective reagents and solvents that would otherwise be incompatible with the sensitive triazolinone ring. By eliminating the need for extensive chromatographic purification to remove ring-degradation impurities, the overall consumption of silica gel and organic solvents is substantially decreased. Additionally, the higher overall yield means that less starting material is required to produce the same amount of final product, directly lowering the raw material cost per kilogram. The avoidance of expensive heavy metal catalysts or specialized reagents for stabilizing the ring further contributes to a leaner cost structure, making the final herbicide more competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic pathway ensures that production is less susceptible to variations in raw material quality or minor fluctuations in reaction parameters. Since the critical ring-forming steps occur under milder conditions, the process is easier to control and scale, reducing the likelihood of off-spec batches that disrupt supply schedules. The use of common, commercially available starting materials like 2,4-dichloroaniline and acetyl chloride ensures that the supply chain is not dependent on exotic or single-source reagents. This diversification of the supply base mitigates the risk of shortages and allows for more flexible sourcing strategies, ensuring continuous availability of the intermediate for year-round herbicide production.
- Scalability and Environmental Compliance: From an environmental perspective, the new route generates fewer hazardous waste streams associated with ring decomposition and over-nitration. The ability to perform nitration on a simpler aniline derivative allows for better management of acid waste and easier neutralization protocols. The late-stage introduction of the difluoromethyl group minimizes the release of fluorinated volatile organic compounds (VOCs) that might occur if the group were subjected to high-temperature acidic workups. This alignment with green chemistry principles facilitates easier regulatory compliance and reduces the environmental footprint of the manufacturing site, which is increasingly important for maintaining operational licenses and meeting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the operational benefits and chemical rationale behind the new process. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer or for procurement teams assessing the long-term viability of this supply source.
Q: Why is the new synthesis route for sulfentrazone intermediates more stable than conventional methods?
A: Conventional methods perform nitration and reduction after the triazolinone ring is formed, exposing the unstable N-difluoromethyl group to harsh mixed acid conditions. The new route performs these steps on the aniline ring first, avoiding decomposition risks.
Q: What represents the key technological breakthrough in patent CN114044758A?
A: The key breakthrough is the strategic reordering of synthetic steps, specifically delaying the introduction of the difluoromethyl group until after the harsh nitration and reduction phases are complete, thereby preserving structural integrity.
Q: How does this synthesis method impact the purity profile of the final herbicide?
A: By avoiding the degradation of the triazolinone ring during nitration, the new method significantly reduces the formation of nitrated isomers and dinitrated byproducts, leading to a cleaner impurity profile and higher overall yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfentrazone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis route are fully realized in a manufacturing setting. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards. Our capability to handle complex fluorinated chemistry and sensitive heterocyclic formations positions us as a strategic partner for companies seeking to optimize their herbicide supply chains.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your existing supply network. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this more stable intermediate can reduce your total landed costs. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a sustainable and efficient supply of high-purity agrochemical intermediates that drive your business forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
