Advanced Nickel-Catalyzed Synthesis of Alpha Beta Unsaturated Thioesters for Pharmaceutical Intermediates
The landscape of organic synthesis for complex molecular scaffolds is undergoing a significant transformation, driven by the urgent need for more sustainable and cost-effective methodologies. Patent CN114773242A introduces a groundbreaking preparation method for α,β-unsaturated thioester compounds, a class of molecules pivotal in the construction of natural products and pharmaceutical intermediates. These compounds serve as versatile building blocks for Diels-Alder reactions, conjugate additions, and cascade cyclizations, yet their traditional synthesis has often been plagued by inefficiencies. The disclosed technology leverages a sophisticated nickel-catalyzed thiocarbonylation strategy that replaces expensive precious metals with earth-abundant nickel, while simultaneously utilizing arylsulfonyl chlorides as a superior sulfur source. This innovation not only addresses the economic constraints of using rhodium or palladium but also solves the operational hazards associated with volatile thiols, marking a substantial leap forward for reliable pharmaceutical intermediate supplier networks seeking robust synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α,β-unsaturated thioesters has relied heavily on condensation reactions or transition metal-catalyzed thiocarbonylation using noble metals such as rhodium, platinum, and palladium. While these precious metal catalysts exhibit high activity, their prohibitive cost and scarcity create significant bottlenecks in cost reduction in pharmaceutical intermediates manufacturing. More critically, conventional thiocarbonylation protocols frequently necessitate the use of thiols as sulfur sources. Thiols are chemically problematic; they possess extremely unpleasant odors that complicate laboratory and plant operations, and perhaps more detrimentally, they act as potent catalyst poisons. This catalyst deactivation leads to inconsistent reaction yields, requiring higher catalyst loadings and generating excessive heavy metal waste, which complicates downstream purification and environmental compliance for any commercial scale-up of complex fine chemicals.
The Novel Approach
The methodology outlined in CN114773242A fundamentally re-engineers this synthetic pathway by introducing a nickel-catalyzed system that utilizes arylsulfonyl chlorides and alkenyl trifluoromethanesulfonates. This approach elegantly bypasses the need for malodorous thiols, thereby eliminating catalyst poisoning risks and improving the overall reaction efficiency. By employing molybdenum hexacarbonyl [Mo(CO)₆] as both the carbonyl source and a reducing agent, the process avoids the handling of toxic carbon monoxide gas, enhancing safety profiles significantly. The reaction operates under relatively mild thermal conditions (90–110°C) in ethylene glycol dimethyl ether (DME), demonstrating exceptional functional group tolerance. This allows for the synthesis of diverse derivatives, including those with halogen, alkyl, and alkoxy substituents, making it an ideal solution for reducing lead time for high-purity pharmaceutical intermediates where structural diversity is paramount.
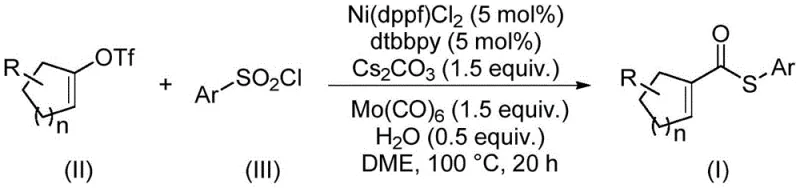
Mechanistically, this transformation represents a masterful orchestration of organometallic chemistry. The catalytic cycle initiates with the reduction of the nickel(II) precatalyst, likely facilitated by the Mo(CO)₆, to generate the active nickel(0) species. Unlike traditional carbonylations that rely on external CO pressure, the in situ release of CO from the molybdenum complex ensures a steady, controlled concentration of the carbonyl source, mitigating the formation of inactive nickel tetracarbonyl species that often plague nickel-catalyzed carbonylations. The oxidative addition of the arylsulfonyl chloride to the nickel center is a key differentiator; the sulfur-chlorine bond is activated to introduce the sulfur moiety without the side reactions typical of thiol-based systems. Subsequent migratory insertion of the alkenyl group and reductive elimination forge the critical carbon-sulfur and carbon-carbonyl bonds. This precise control over the catalytic cycle ensures high selectivity for the α,β-unsaturated thioester product, minimizing byproduct formation and simplifying the impurity profile, which is a critical metric for any high-purity OLED material or API intermediate production.
Furthermore, the impurity control mechanism inherent in this design is robust. The use of cesium carbonate as a base provides a mild yet effective environment for neutralizing the hydrochloric acid byproduct generated from the sulfonyl chloride, preventing acid-catalyzed degradation of the sensitive unsaturated ester product. The wide substrate scope demonstrated in the patent, accommodating cyclopentenyl, cyclohexenyl, and cycloheptenyl rings, indicates that steric hindrance is well-managed by the bulky dppf ligand on the nickel center. This steric bulk protects the active site from non-productive binding events, ensuring that the reaction proceeds efficiently even with sterically demanding substrates. For R&D teams, this means a predictable and reproducible pathway that can be adapted for various analogues without extensive re-optimization, streamlining the development of new drug candidates.
How to Synthesize Alpha,Beta-Unsaturated Thioesters Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it highly attractive for process chemistry teams aiming for rapid technology transfer. The protocol involves charging a sealed vessel with the nickel catalyst, ligand, molybdenum source, base, and substrates in DME solvent. The mixture is then heated to promote the thiocarbonylation event. Following the reaction, the workup is straightforward, involving filtration to remove inorganic salts and metal residues, followed by standard silica gel chromatography. This simplicity reduces the technical barrier for adoption and facilitates the commercial scale-up of complex polymer additives or pharma intermediates. For detailed standardized operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the technical guide below.
- Combine nickel catalyst (Ni(dppf)Cl2), ligand (dtbbpy), molybdenum hexacarbonyl, cesium carbonate, water, alkenyl triflate, and arylsulfonyl chloride in a sealed vessel with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 20 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the target alpha,beta-unsaturated thioester.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this patented technology offers compelling advantages that directly impact the bottom line and supply chain resilience. The shift from precious metals to nickel represents a fundamental change in the cost structure of the synthesis. Nickel is orders of magnitude cheaper than palladium or rhodium, and its abundance ensures long-term price stability, shielding manufacturers from the volatile fluctuations common in the precious metals market. Additionally, the replacement of thiols with arylsulfonyl chlorides eliminates the need for specialized odor-control infrastructure and hazardous waste disposal protocols associated with sulfur-containing volatiles. This results in substantial cost savings in facility maintenance and regulatory compliance, allowing for more competitive pricing strategies in the global marketplace.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the substitution of expensive catalysts and hazardous reagents. By utilizing nickel, a base metal, the direct material cost of the catalyst system is drastically reduced compared to traditional palladium-mediated processes. Moreover, the use of solid Mo(CO)₆ eliminates the capital expenditure and safety costs associated with high-pressure CO gas reactors. The simplified workup procedure, which avoids complex extraction or distillation steps often required to remove smelly thiol residues, further lowers processing costs. These factors combine to create a highly efficient manufacturing process that maximizes yield while minimizing overhead, delivering significant value to procurement managers focused on margin optimization.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of commodity-grade starting materials. Alkenyl trifluoromethanesulfonates and arylsulfonyl chlorides are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of functional groups and moisture (via the added water component), ensures consistent batch-to-batch quality even with slight variations in raw material grades. This reliability is crucial for maintaining uninterrupted production schedules for critical pharmaceutical intermediates, ensuring that downstream customers receive their materials on time without quality deviations that could halt their own production lines.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is significantly lighter than conventional methods, aligning with modern green chemistry principles. The absence of volatile thiols reduces atmospheric emissions and worker exposure risks, simplifying EHS (Environment, Health, and Safety) compliance. The reaction generates fewer heavy metal wastes due to the lower toxicity and cost of nickel, facilitating easier disposal or recycling. From a scalability standpoint, the use of standard heating and stirring equipment without the need for specialized high-pressure gas handling makes the transition from gram-scale R&D to multi-ton commercial production seamless. This ease of scale-up ensures that supply can be rapidly ramped up to meet market demand without extensive engineering modifications.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the reaction scope, safety, and scalability. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these nuances is essential for integrating this methodology into existing production workflows effectively.
Q: Why is arylsulfonyl chloride preferred over thiols in this synthesis?
A: Traditional thiocarbonylation often relies on thiols, which are notorious for their unpleasant odor and tendency to poison transition metal catalysts. This patent utilizes arylsulfonyl chlorides as a stable, odorless, and non-poisonous sulfur source, significantly improving operational safety and catalyst longevity.
Q: What is the dual function of Molybdenum Hexacarbonyl (Mo(CO)6) in this reaction?
A: Mo(CO)6 serves a critical dual role: it acts as the solid-state source of carbon monoxide (CO) required for the carbonylation step, eliminating the need for handling hazardous CO gas, and simultaneously functions as a reducing agent to regenerate the active low-valent nickel species.
Q: Is this nickel-catalyzed method suitable for large-scale manufacturing?
A: Yes, the process is highly scalable due to the use of inexpensive, earth-abundant nickel catalysts instead of precious metals like palladium or rhodium. Furthermore, the reaction conditions (100°C in DME) and simple workup procedures facilitate easy commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Unsaturated Thioester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the nickel-catalyzed thiocarbonylation technology described in CN114773242A for the synthesis of high-value thioester intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of α,β-unsaturated thioester delivered meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in nickel catalysis can drive efficiency and reduce costs in your supply chain.
