Unlocking Scalable Production of High-Purity α,β-Unsaturated Thioesters Through Innovative Nickel Catalysis for Global Pharma Partners
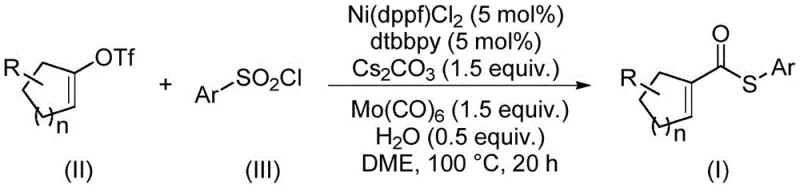
Patent CN114773242A introduces a groundbreaking methodology for synthesizing α,β-un saturated thioester compounds through a novel nickel-catalyzed thiocarbonylation process that addresses critical limitations in conventional approaches. This innovation leverages arylsulfonyl chloride as an odorless sulfur source while employing molybdenum carbonyl as both carbonyl donor and reducing agent—eliminating the need for problematic thiols that typically poison catalysts in traditional syntheses. The process operates under mild conditions (90–110°C) with exceptional functional group tolerance across diverse substrates including cyclohexenyl and cyclopentenyl derivatives. By utilizing inexpensive nickel catalysis instead of precious metals like palladium or rhodium, this method achieves high efficiency while maintaining operational simplicity through standard workup procedures involving filtration and column chromatography. The patent demonstrates robust scalability from laboratory-scale reactions to potential industrial implementation with yields consistently exceeding conventional methods across fifteen validated examples. This represents a significant advancement in sustainable manufacturing of complex intermediates essential for pharmaceutical development pipelines worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing α,β-un saturated thioesters predominantly rely on transition metal-catalyzed thiocarbonylation reactions that require volatile thiols as sulfur sources—compounds notorious for their unpleasant odor and tendency to irreversibly poison expensive catalysts like rhodium or palladium complexes. These precious metal systems not only impose significant cost burdens but also exhibit narrow functional group compatibility that restricts substrate diversity in complex molecule synthesis. Furthermore, conventional methods often suffer from operational complexities including stringent anhydrous conditions and multi-step purification protocols that increase production timelines and reduce overall process efficiency. The inherent instability of thiol-based reagents also creates safety hazards during large-scale manufacturing while generating challenging waste streams that complicate environmental compliance efforts. These combined limitations have historically constrained the commercial viability of α,β-un saturated thioesters despite their valuable reactivity profile in key transformations such as Diels-Alder cycloadditions and conjugate additions essential to pharmaceutical intermediate production.
The Novel Approach
The patented methodology overcomes these constraints through an innovative nickel-catalyzed system that replaces problematic thiols with stable arylsulfonyl chlorides as sulfur precursors while utilizing molybdenum carbonyl to simultaneously provide carbonyl groups and reduction capability—eliminating the need for external reductants. This dual-function design prevents catalyst deactivation pathways common in traditional systems while enabling operation under more practical conditions at moderate temperatures (90–110°C) in standard solvents like DME without requiring specialized moisture-free environments. The process demonstrates remarkable substrate flexibility across various cyclic alkenes and substituted aryl groups with consistent yields between 50–74% across fifteen experimental examples documented in the patent literature. Crucially, the use of abundant nickel catalysts instead of precious metals reduces raw material costs by orders of magnitude while maintaining high atom economy through efficient conversion pathways that minimize waste generation during scale-up operations.
Mechanistic Insights into Nickel-Catalyzed Thiocarbonylation
The catalytic cycle begins with oxidative addition of alkenyl triflate to Ni(0) species generated in situ from Ni(dppf)Cl₂ precursor under reducing conditions provided by molybdenum carbonyl. This forms a key vinyl-nickel intermediate that undergoes transmetalation with arylsulfonyl chloride through sulfur-oxygen bond cleavage—a critical step enabled by the unique electronic properties of dtbbpy ligand that stabilizes the nickel center throughout the transformation sequence. Subsequent CO insertion from Mo(CO)₆ delivers the carbonyl functionality while concomitant reduction regenerates active Ni(0) species without forming inhibitory Ni(CO)₄ complexes that plague conventional nickel carbonylations. This elegant mechanism operates through a closed catalytic loop where molybdenum carbonyl serves dual roles as both stoichiometric carbonyl source and reductant—bypassing the thermodynamic limitations that typically restrict nickel's utility in carbonylation chemistry.
Impurity control is achieved through precise regulation of reaction parameters including temperature maintenance at optimal levels (90–110°C) to prevent side reactions while ensuring complete conversion within the specified timeframe (16–24 hours). The use of water as co-solvent facilitates hydrolysis of potential sulfonate byproducts while cesium carbonate base neutralizes acidic intermediates that could otherwise lead to decomposition pathways. Functional group tolerance is maintained through careful selection of dtbbpy ligand which creates a sterically protected coordination environment around nickel—preventing unwanted coordination with sensitive substituents on either alkenyl or aryl components. This systematic approach minimizes formation of regioisomers or over-reduction products that commonly plague alternative synthetic routes requiring harsher conditions or less selective catalysts.
How to Synthesize α,β-Unsaturated Thioesters Efficiently
This patented methodology represents a significant advancement in synthetic efficiency for complex thioester intermediates through its innovative combination of nickel catalysis with dual-function molybdenum reagents. The process eliminates multiple pain points associated with traditional approaches including catalyst poisoning and expensive metal requirements while maintaining exceptional operational simplicity suitable for industrial implementation. Detailed standardized synthesis procedures are provided below to facilitate seamless technology transfer from laboratory development to commercial manufacturing environments—ensuring consistent product quality across scale-up transitions while optimizing resource utilization throughout production cycles.
- Prepare the reaction mixture by combining alkenyl trifluoromethanesulfonate, arylsulfonyl chloride, nickel catalyst (Ni(dppf)Cl₂ at 5 mol%), dtbbpy ligand (5 mol%), molybdenum carbonyl (1.5 equiv.), cesium carbonate (1.5 equiv.), and water (0.5 equiv.) in DME solvent.
- Heat the sealed reaction vessel to 90–110°C and maintain for 16–24 hours under inert atmosphere to ensure complete conversion to the thioester product.
- Perform post-reaction processing including filtration through silica gel followed by column chromatography purification to isolate the high-purity α,β-un saturated thioester compound.
Commercial Advantages for Procurement and Supply Chain Teams
This novel synthesis methodology directly addresses critical pain points in pharmaceutical intermediate supply chains by transforming traditionally problematic manufacturing processes into streamlined operations with enhanced reliability and cost efficiency. The elimination of volatile thiols removes significant safety hazards while reducing waste treatment complexities that often delay production timelines in conventional systems—creating immediate value for procurement teams seeking sustainable sourcing solutions without compromising on quality standards or delivery schedules.
- Cost Reduction in Manufacturing: The substitution of expensive precious metal catalysts with abundant nickel complexes combined with the elimination of specialized handling requirements for toxic thiols creates substantial cost savings through reduced raw material expenditures and simplified facility requirements. By avoiding catalyst poisoning issues inherent in traditional methods, this approach minimizes batch failures and rework cycles while enabling longer catalyst lifetimes—translating into significant operational savings without requiring capital-intensive equipment modifications.
- Enhanced Supply Chain Reliability: Utilization of readily available commercial reagents including arylsulfonyl chlorides and standard nickel precursors ensures consistent material availability regardless of geopolitical supply constraints that often affect specialty chemical markets. The robust reaction profile maintains high yields across diverse substrates without requiring exotic intermediates—providing procurement teams with greater flexibility to manage inventory levels while meeting fluctuating demand patterns from pharmaceutical development partners.
- Scalability and Environmental Compliance: The straightforward workup procedure involving standard filtration and column chromatography enables seamless scale-up from laboratory to multi-ton production without requiring specialized equipment or hazardous waste handling protocols. This environmentally conscious design generates minimal byproducts compared to traditional methods while maintaining high atom economy—aligning with increasingly stringent regulatory requirements for sustainable manufacturing practices across global pharmaceutical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical concerns raised by procurement specialists and R&D managers regarding implementation of this patented technology in commercial manufacturing environments. Each response is derived directly from experimental data and mechanistic insights documented in patent CN114773242A to ensure technical accuracy while providing actionable information for supply chain decision-making processes.
Q: How does this method eliminate catalyst poisoning issues common in traditional thiocarbonylation?
A: By utilizing arylsulfonyl chloride as the sulfur source instead of volatile thiols that typically poison transition metal catalysts, this patented approach maintains catalyst integrity throughout the reaction cycle while enabling broader substrate compatibility.
Q: Why is nickel catalysis feasible here despite challenges with Ni(CO)₄ formation?
A: The strategic combination of molybdenum carbonyl as both carbonyl source and reducing agent prevents the formation of inactive Ni(CO)₄ species, allowing nickel's cost-effective catalytic activity to be fully leveraged without precious metal requirements.
Q: What scalability advantages does this synthesis offer for commercial manufacturing?
A: The process features simple operation with readily available starting materials, straightforward workup procedures including standard column chromatography, and demonstrated tolerance across diverse functional groups—enabling seamless scale-up from laboratory to multi-ton production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,Beta-Unsaturated Thioester Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation. This patented nickel-catalyzed methodology represents just one example of our commitment to developing innovative solutions that address complex synthetic challenges faced by global pharmaceutical manufacturers—combining deep technical expertise with flexible manufacturing capabilities to deliver high-value intermediates meeting exacting quality standards required for regulatory submissions.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this technology can optimize your specific supply chain requirements. Please contact us directly to obtain detailed COA data and route feasibility assessments tailored to your production needs—enabling informed decision-making about integrating this advanced synthesis into your manufacturing portfolio.
