Advanced Chiral Zinc Nitrogen Complexes for Efficient Asymmetric Synthesis and Commercial Scale-up
Advanced Chiral Zinc Nitrogen Complexes for Efficient Asymmetric Synthesis and Commercial Scale-up
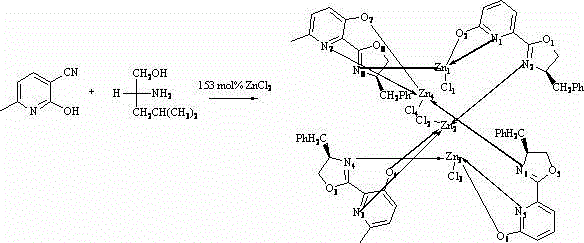
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of novel organometallic architectures that balance high performance with economic viability. Patent CN102863464A details a breakthrough in the development of chiral zinc nitrogen complexes, specifically a tetrameric structure designated as complex (I), which exhibits remarkable catalytic proficiency in carbon-carbon bond-forming reactions. This technology addresses the critical industry demand for reliable chiral catalysts that can operate efficiently without the prohibitive costs associated with precious metals. By leveraging the unique coordination chemistry of zinc with chiral oxazoline-pyridine ligands derived from D-phenylalaninol, this innovation provides a robust platform for the synthesis of high-value pharmaceutical intermediates. The structural integrity and catalytic potential of these complexes offer a compelling value proposition for R&D teams seeking to optimize enantioselective processes while maintaining strict control over impurity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to asymmetric catalysis have long relied on complexes of expensive transition metals such as rhodium, palladium, or iridium, which present substantial supply chain vulnerabilities and cost barriers for large-scale manufacturing. Furthermore, many conventional ligand systems suffer from limited stability under rigorous reaction conditions, often leading to catalyst decomposition and the generation of difficult-to-remove metal impurities in the final active pharmaceutical ingredient (API). The synthesis of chiral ligands themselves can be prohibitively complex, requiring multi-step sequences that erode overall process efficiency and yield. In the context of Henry reactions and cyanosilylation, older catalytic systems frequently struggle to achieve both high conversion rates and excellent enantiomeric excess simultaneously, forcing process chemists to make difficult trade-offs between throughput and stereochemical purity. These limitations necessitate a shift towards more earth-abundant metal centers that do not compromise on catalytic performance.
The Novel Approach
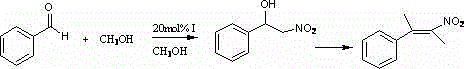
The methodology described in the patent introduces a sophisticated tetrameric zinc complex that overcomes these historical bottlenecks through a unique self-assembly process involving 6-methyl-2-cyano-3-hydroxy pyridine and D-phenylalaninol. This novel approach utilizes zinc chloride, an inexpensive and widely available Lewis acid, coordinated within a rigid chiral environment that effectively directs substrate approach. The resulting complex demonstrates exceptional stability and reactivity, achieving a conversion rate of 81.3 percent in the Henry reaction of benzaldehyde, a benchmark transformation for evaluating nitroaldol catalysts. By shifting the paradigm from precious metals to zinc, this technology drastically simplifies the supply chain logistics and reduces the environmental footprint associated with heavy metal waste disposal. The modular nature of the ligand synthesis allows for easy adaptation to various substrates, providing a versatile tool for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Zn-N Coordination and Catalytic Activity
The catalytic efficacy of this system is rooted in the precise geometric arrangement of the zinc centers within the tetrameric cluster, as elucidated by single-crystal X-ray diffraction data. The structure reveals a complex network of Zn-O and Zn-N bonds that create distinct chiral pockets capable of activating electrophiles while shielding specific faces of the reacting species. In the Henry reaction mechanism, the zinc center acts as a potent Lewis acid to activate the aldehyde carbonyl group, facilitating nucleophilic attack by the nitronate anion generated in situ. The rigidity of the oxazoline-pyridine framework ensures that this activation occurs with high stereocontrol, minimizing the formation of unwanted diastereomers. Understanding these mechanistic nuances is crucial for R&D directors aiming to apply this catalyst to broader substrate scopes, as the steric bulk of the benzyl group on the oxazoline ring plays a pivotal role in defining the enantioselectivity. This level of structural definition allows for rational optimization of reaction parameters to maximize yield and purity.
Impurity control is another critical aspect where this zinc complex excels, primarily due to the homogeneous nature of the catalytic cycle and the stability of the metal-ligand bond. Unlike heterogeneous catalysts that may leach metal ions unpredictably, this well-defined molecular complex maintains its integrity throughout the reaction course, reducing the risk of metal contamination in the product stream. The specific bond lengths, such as the Zn(1)-O(2) distance of 1.968 angstroms and Zn(1)-N(1) distance of 2.001 angstroms, indicate a tight coordination sphere that resists ligand dissociation even under reflux conditions. This stability translates directly to cleaner reaction profiles, simplifying downstream purification processes and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory standards. The ability to predict and control the impurity profile is a significant advantage for process development teams working towards commercial validation.
How to Synthesize Chiral Zinc Complexes Efficiently
The synthesis of this advanced catalytic material is designed to be operationally simple yet chemically rigorous, ensuring reproducibility across different manufacturing scales. The process begins with the careful preparation of anhydrous conditions, as the presence of moisture can disrupt the coordination equilibrium and lead to hydrolysis of the zinc chloride precursor. By reacting the pyridine derivative with the chiral amino alcohol in chlorobenzene at elevated temperatures, the system drives the formation of the thermodynamically stable tetrameric architecture. The detailed standardized synthesis steps provided below outline the precise stoichiometry and workup procedures required to isolate the complex in high purity, serving as a foundational protocol for technical teams looking to implement this technology.
- Prepare the reaction mixture by combining anhydrous ZnCl2, 6-methyl-2-cyano-3-hydroxy pyridine, and D-phenylalaninol in chlorobenzene solvent under strict anhydrous and oxygen-free conditions.
- Heat the mixture to reflux temperature for approximately 48 to 60 hours to ensure complete coordination and formation of the tetrameric zinc complex structure.
- Purify the crude product by removing the solvent, dissolving the residue in water, extracting with chloroform, and performing column chromatography using petroleum ether and methylene chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-based catalytic technology represents a strategic opportunity to de-risk production schedules and optimize raw material expenditures. The shift away from precious metal catalysts eliminates exposure to the volatile pricing markets of rhodium and palladium, providing a more predictable cost structure for long-term project planning. Additionally, the starting materials for the ligand synthesis, such as 6-methyl-2-cyano-3-hydroxy pyridine and D-phenylalaninol, are commercially available commodity chemicals with robust global supply chains, ensuring continuity of supply even during market disruptions. This reliability is essential for maintaining uninterrupted production lines for critical API intermediates.
- Cost Reduction in Manufacturing: The primary economic driver for this technology is the substitution of expensive noble metals with zinc, which is orders of magnitude cheaper and more abundant. This fundamental change in the catalyst composition leads to substantial cost savings in the bill of materials, particularly for processes requiring high catalyst loadings. Furthermore, the simplified purification workflow, enabled by the stability of the complex, reduces the consumption of solvents and chromatography media, further driving down the operational expenses associated with manufacturing. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS) for the final chiral intermediates.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the use of earth-abundant zinc salts and readily available organic building blocks, reducing dependency on single-source suppliers of specialized ligands or rare metals. The synthetic route is robust and tolerant of minor variations in feedstock quality, which enhances the resilience of the supply chain against quality fluctuations. By establishing a manufacturing process based on widely accessible chemicals, companies can secure multiple sourcing options, thereby mitigating the risk of supply shortages that often plague the fine chemical industry. This stability ensures consistent delivery timelines for downstream customers.
- Scalability and Environmental Compliance: The process utilizes standard unit operations such as reflux and liquid-liquid extraction, which are easily scalable from kilogram to tonne quantities without the need for specialized high-pressure equipment. From an environmental perspective, the use of zinc minimizes the generation of toxic heavy metal waste, simplifying effluent treatment and reducing the regulatory burden associated with hazardous waste disposal. The high atom economy of the catalytic cycle and the potential for catalyst recycling further contribute to a greener manufacturing profile, aligning with modern sustainability goals and corporate responsibility initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral zinc complex technology. These insights are derived directly from the experimental data and structural analysis presented in the patent documentation, providing a clear understanding of the catalyst's capabilities and limitations. By addressing these key areas, we aim to facilitate informed decision-making for technical leaders evaluating this solution for their specific synthetic challenges.
Q: What is the catalytic efficiency of this chiral zinc complex in Henry reactions?
A: According to patent CN102863464A, the complex demonstrates significant catalytic activity in the Henry reaction of benzaldehyde, achieving a conversion rate of 81.3 percent under optimized conditions.
Q: Why is this zinc-based catalyst preferred over traditional transition metal catalysts?
A: Zinc is significantly more abundant and less toxic than precious metals like palladium or rhodium. This complex offers a cost-effective alternative for asymmetric synthesis while maintaining high stereocontrol through its unique tetrameric structure.
Q: Can this synthesis process be scaled for industrial production?
A: Yes, the synthesis utilizes standard reflux conditions in chlorobenzene and common purification techniques like column chromatography, making it highly amenable to scale-up from laboratory to commercial manufacturing volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Zinc Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organometallic catalysts in accelerating drug discovery and process development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify the structural integrity and catalytic activity of every batch. Our capability to handle complex coordination chemistry allows us to support clients in bringing innovative chiral intermediates to market faster.
We invite you to collaborate with us to leverage this cutting-edge zinc catalysis technology for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific process requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our chiral zinc complexes can enhance the efficiency and profitability of your manufacturing operations.
