Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Production
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds exhibiting potent biological activities. Patent CN112239456B introduces a groundbreaking preparation method for substituted 2,3-dihydroquinolone compounds, utilizing a transition metal palladium-catalyzed carbonylation reaction. This technology addresses critical challenges in the synthesis of these valuable pharmacophores, which are known for their significant anti-cancer and analgesic properties, as illustrated by the bioactive structures in the prior art. By leveraging N-pyridine sulfonyl-o-iodoaniline and various olefins as starting materials, this novel approach offers a robust pathway for generating complex molecular architectures essential for drug discovery pipelines.
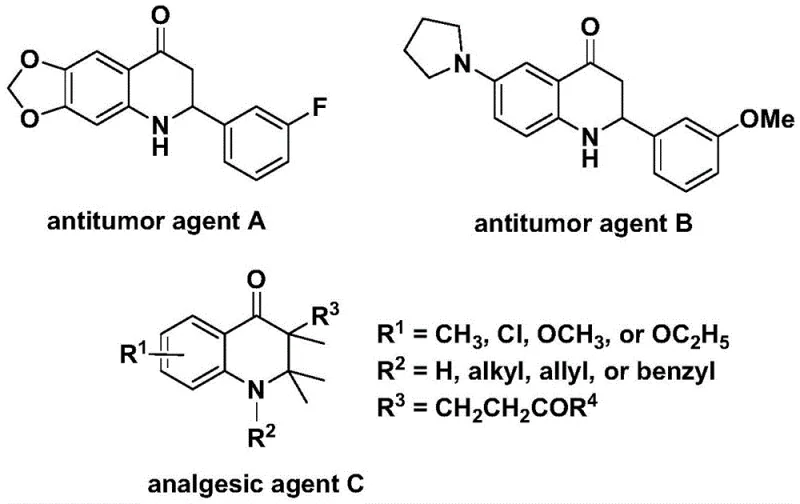
The strategic importance of the 2,3-dihydroquinolone skeleton cannot be overstated, given its prevalence in molecules with profound therapeutic potential. Historical data indicates that specific derivatives, such as those depicted in the reference structures, have demonstrated remarkable efficacy against human cancer cells and pain management applications. However, traditional synthetic methodologies often struggle with harsh conditions, limited substrate scope, or the use of hazardous gaseous reagents. The innovation disclosed in this patent provides a sophisticated solution by employing a solid carbon monoxide substitute, thereby eliminating the safety risks associated with handling toxic CO gas while maintaining high reaction efficiency and atom economy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydroquinolone core via carbonylation has been hindered by significant operational and safety barriers. Conventional protocols frequently rely on the direct use of carbon monoxide gas, which poses severe toxicity risks and requires specialized high-pressure equipment, making scale-up prohibitively expensive and dangerous for many facilities. Furthermore, existing methods often suffer from poor functional group tolerance, limiting the diversity of substituents that can be introduced at the 2-aryl and 3-alkyl positions without compromising yield. These limitations create bottlenecks in the rapid generation of analog libraries needed for structure-activity relationship (SAR) studies, ultimately slowing down the drug development timeline for potential new medicines targeting oncology and neurological disorders.
The Novel Approach
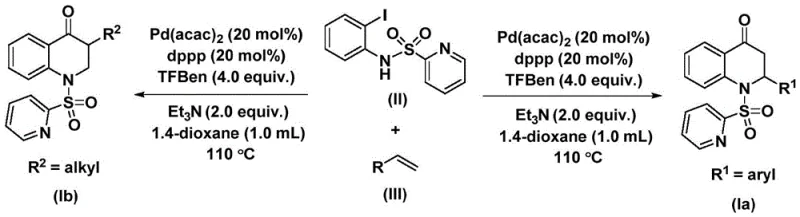
The methodology outlined in patent CN112239456B represents a paradigm shift by replacing gaseous CO with 1,3,5-trimesic acid phenol ester, a stable and easy-to-handle solid surrogate. This substitution not only drastically enhances laboratory and plant safety but also simplifies the reaction setup, allowing it to proceed under standard atmospheric pressure conditions. The process utilizes a palladium catalyst system comprising bis(acetylacetone)palladium and a dppp ligand, which facilitates the smooth insertion of the carbonyl group into the carbon-nitrogen bond framework. This innovative strategy enables the synthesis of a wide array of substituted derivatives with high yields, demonstrating exceptional versatility in accommodating both electron-rich and electron-deficient substrates.
Mechanistic Insights into Palladium-Catalyzed Carbonylative Cyclization
The catalytic cycle driving this transformation is a masterpiece of organometallic chemistry, initiated by the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This critical step generates a reactive aryl-palladium intermediate, which subsequently undergoes migratory insertion with carbon monoxide released in situ from the phenol ester source. The resulting acyl-palladium species is then poised for coordination with the olefin reactant, leading to the formation of a palladium-alkyl intermediate through alkene insertion. Finally, a reductive elimination step closes the catalytic loop, releasing the desired substituted 2,3-dihydroquinolone product and regenerating the active palladium catalyst for further turnover.
From an impurity control perspective, the choice of ligands and the specific reaction temperature window of 100-120 °C are paramount in suppressing side reactions such as homocoupling or beta-hydride elimination. The use of triethylamine as an additive helps to neutralize acidic byproducts and maintain the optimal pH environment for the catalytic cycle to proceed efficiently. Moreover, the structural integrity of the pyridine sulfonyl protecting group ensures that the nitrogen atom remains available for cyclization without undergoing unwanted decomposition, thereby securing a clean reaction profile that simplifies downstream purification processes significantly.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
To implement this high-yielding synthesis in a practical setting, precise adherence to the molar ratios and reaction parameters described in the patent is essential for maximizing output. The protocol involves charging a reaction vessel with the iodoaniline derivative, the chosen olefin, the palladium catalyst, the phosphine ligand, and the solid CO source in an aprotic solvent like dioxane. Maintaining the temperature at 110 °C for a duration of 48 hours ensures complete conversion of the starting materials, after which standard workup procedures involving filtration and silica gel chromatography afford the pure product. Detailed standardized synthesis steps are provided in the guide below for technical teams to follow.
- Combine N-pyridine sulfonyl-o-iodoaniline, olefin, palladium catalyst, ligand, and solid CO source in an organic solvent.
- Heat the reaction mixture to 100-120 °C and stir for 24-48 hours under inert atmosphere.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits regarding cost stability and operational reliability. By eliminating the need for high-pressure carbon monoxide infrastructure, facilities can avoid substantial capital expenditures on safety systems and specialized reactors, leading to significant cost reduction in pharmaceutical intermediate manufacturing. The reliance on commercially available starting materials, such as simple olefins and readily synthesized iodoanilines, ensures a robust supply chain that is less susceptible to the volatility often seen with exotic or highly regulated reagents. This accessibility translates directly into shorter lead times and more predictable delivery schedules for bulk orders.
- Cost Reduction in Manufacturing: The replacement of toxic gas with a solid ester surrogate removes the logistical and regulatory burdens associated with hazardous gas handling, effectively lowering overhead costs related to safety compliance and waste disposal. Additionally, the high catalytic efficiency minimizes the loading of expensive palladium metals required per batch, optimizing the overall material cost structure. The simplified post-treatment process, which avoids complex extraction sequences, further reduces labor and solvent consumption, contributing to a leaner and more economical production model.
- Enhanced Supply Chain Reliability: Since the key reagents like 1,3-bis(diphenylphosphino)propane and bis(acetylacetone)palladium are standard catalog items, sourcing risks are minimized compared to custom-synthesized catalysts. The broad substrate scope allows manufacturers to pivot quickly between different product variants (e.g., switching from aryl to alkyl substituents) without retooling the entire production line, providing agility in responding to market demands. This flexibility ensures continuous supply continuity even when specific raw material markets experience temporary fluctuations.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, having been demonstrated to work effectively from gram to multi-gram levels without loss of efficiency, paving the way for commercial scale-up of complex pharmaceutical intermediates. The use of dioxane, a common industrial solvent, facilitates straightforward recycling and recovery, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. The absence of heavy metal contamination issues typically associated with difficult-to-remove catalysts simplifies the path to meeting stringent regulatory purity specifications.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this carbonylation technology. Understanding these nuances is vital for R&D teams planning to integrate this method into their existing workflows or for quality assurance personnel establishing control strategies. The answers are derived directly from the experimental data and mechanistic understanding presented in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: What is the primary advantage of using 1,3,5-trimesic acid phenol ester in this synthesis?
A: It serves as a solid, safe substitute for toxic carbon monoxide gas, significantly improving operational safety and handling convenience in industrial settings.
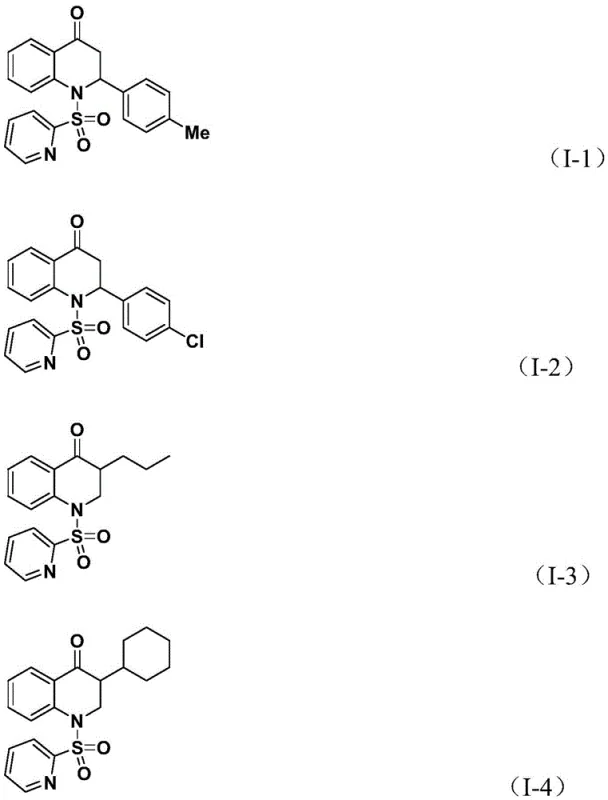
Q: Can this method accommodate diverse functional groups on the olefin substrate?
A: Yes, the protocol demonstrates excellent substrate compatibility, successfully tolerating aryl, alkyl, and silyl groups with various substituents like halogens and alkoxy groups.
Q: What are the typical reaction conditions required for high conversion?
A: Optimal results are achieved using dioxane as the solvent at temperatures between 100-120 °C for a duration of 24 to 48 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with the utmost precision, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality. We understand the critical nature of supply chain consistency in the pharmaceutical industry and are committed to delivering high-purity intermediates that meet global regulatory standards.
We invite you to contact our technical procurement team to discuss how this advanced synthesis route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient methodology. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process and accelerate your time to market.
