Advanced Synthesis of Geminal Dinitro Compounds Using Nitrate Hydrate for Commercial Scale-up
The landscape of organic synthesis for high-energy and bioactive molecules is undergoing a significant transformation, driven by the urgent need for safer and more sustainable methodologies. A pivotal advancement in this domain is detailed in patent CN109574913B, which discloses a robust method for preparing geminal dinitro compounds using nitrate hydrates. This technology represents a paradigm shift from traditional, hazardous nitration protocols to a streamlined, iron-catalyzed radical cascade process. By utilizing readily available iron nitrate nonahydrate as both a catalyst and a nitro source, this approach addresses critical pain points in the manufacturing of energetic materials and potential pharmaceutical intermediates, specifically those exhibiting nitric oxide release capabilities. The ability to construct complex cyclic gem-dinitro scaffolds directly from 1,6-diyne precursors under mild conditions offers a compelling value proposition for R&D teams seeking to optimize their synthetic routes for scale-up.
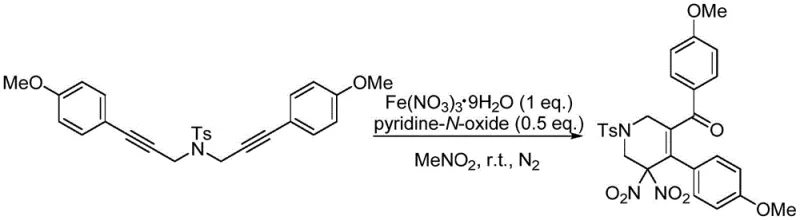
The significance of this innovation cannot be overstated when considering the historical challenges associated with introducing gem-dinitro functionalities into organic frameworks. Conventional strategies often rely on harsh oxidative nitration of nitroalkanes under strongly alkaline conditions or the use of excessive amounts of concentrated nitric acid, which poses severe safety and environmental liabilities. In contrast, the methodology described in the patent leverages a tandem nitration-cyclization-oxidation sequence that proceeds efficiently at temperatures between 25°C and 60°C. This gentle thermal profile not only enhances operational safety but also preserves the integrity of sensitive functional groups on the aromatic rings, thereby expanding the scope of accessible chemical space for drug discovery and material science applications without compromising on yield or purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of geminal dinitro compounds has been plagued by substantial technical and logistical hurdles that hinder efficient commercial production. Traditional pathways typically necessitate the use of strong bases to facilitate the oxidative nitration of nitroalkanes, a process that generates significant amounts of saline waste and requires rigorous pH control to prevent decomposition of the sensitive dinitro moiety. Furthermore, alternative methods involving the serial nitrosation-oxidation of oximes often demand concentrated nitric acid to serve as both the reagent and the solvent. This reliance on corrosive, fuming acids creates a hostile environment for standard stainless steel reactors, necessitating expensive glass-lined equipment and specialized handling protocols that drastically inflate capital expenditure and operational overheads. Additionally, the exothermic nature of these classical nitration reactions poses a constant risk of thermal runaway, requiring sophisticated cooling systems and limiting the feasible batch sizes for safe operation.
The Novel Approach
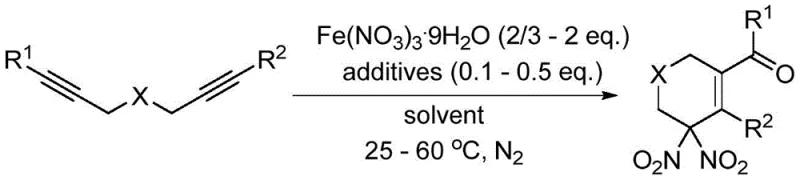
The novel approach outlined in the patent data introduces a transformative strategy that circumvents these legacy issues by employing iron nitrate nonahydrate as a benign yet effective nitrating agent. This method utilizes a radical mechanism where the nitrate salt decomposes in situ to generate nitro radicals, which then engage with the 1,6-diyne substrate to initiate the cyclization cascade. As illustrated in the general reaction scheme below, this process integrates nitration, cyclization, and oxidation into a single pot operation, significantly simplifying the workflow.
The use of additives such as pyridine-N-oxide or benzoquinone further fine-tunes the reaction kinetics, ensuring high conversion rates while minimizing the formation of undesired byproducts. By shifting the reaction medium to common organic solvents like acetonitrile or nitromethane, the process eliminates the need for bulk mineral acids, thereby reducing waste treatment costs and enhancing the overall green chemistry profile. This streamlined protocol not only improves the safety margin for operators but also facilitates easier downstream processing, as the removal of iron residues is straightforward compared to the neutralization of large volumes of acidic waste.
Mechanistic Insights into Fe(NO3)3-Catalyzed Cyclization
At the heart of this technological breakthrough lies a sophisticated radical cascade mechanism that orchestrates the construction of the six-membered ring bearing the gem-dinitro group. The reaction initiates with the thermal decomposition of the iron nitrate hydrate, which serves as a reservoir for nitro radicals (•NO2). These highly reactive species selectively attack the electron-rich triple bonds of the 1,6-diyne substrate, generating vinyl radical intermediates. This initial addition step is crucial as it sets the stereochemical and regiochemical trajectory for the subsequent cyclization. The presence of the linker atom X (which can be oxygen, nitrogen, or carbon) plays a pivotal role in directing the intramolecular radical attack, leading to the formation of a cyclic radical species that is subsequently trapped by a second nitro radical or undergoes oxidation to restore aromaticity or form the double bond within the ring.
Understanding the impurity profile is essential for R&D directors aiming to implement this chemistry in a GMP environment. The controlled generation of nitro radicals ensures that over-nitration of the aromatic rings is minimized, a common side reaction in electrophilic aromatic substitution processes. The additives, acting as oxidants or radical promoters, help to drive the reaction to completion and suppress the accumulation of partially nitrated intermediates or polymerization byproducts that could arise from uncontrolled radical propagation. Furthermore, the mild acidic nature of the iron salt solution helps to protonate basic sites if present, preventing catalyst deactivation and ensuring consistent reaction performance across different batches. This mechanistic clarity allows for precise tuning of reaction parameters, such as temperature and stoichiometry, to maximize the yield of the desired gem-dinitro scaffold while maintaining a clean impurity spectrum.
How to Synthesize Geminal Dinitro Compound Efficiently
To successfully implement this synthesis route in a laboratory or pilot plant setting, strict adherence to the optimized protocol is required to ensure reproducibility and safety. The process begins with the careful weighing of the 1,6-diyne substrate and the iron nitrate nonahydrate, typically in a molar ratio ranging from 2:1 to 2:3, along with a catalytic amount of an additive like pyridine-N-oxide. The reaction is conducted under an inert nitrogen atmosphere to prevent interference from atmospheric oxygen, which could lead to unpredictable radical termination pathways. Detailed standardized operating procedures regarding solvent selection, addition rates, and workup techniques are critical for achieving the high yields reported in the patent examples.
- Prepare the reaction mixture by combining 1,6-diyne substrate, Fe(NO3)3·9H2O, and an additive such as pyridine-N-oxide or benzoquinone in an organic solvent like acetonitrile or nitromethane.
- Conduct the reaction under a nitrogen atmosphere at temperatures ranging from 25°C to 60°C, stirring for 4 to 26 hours depending on the substrate reactivity.
- Upon completion, filter the mixture through celite to remove insolubles, concentrate the filtrate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nitrate hydrate-based methodology offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material supply chain. Iron nitrate nonahydrate is a commodity chemical that is inexpensive, stable, and readily available in bulk quantities globally, unlike specialized nitrating agents or exotic catalysts that may suffer from supply volatility. This stability in raw material sourcing translates directly into enhanced supply chain reliability, ensuring that production schedules are not disrupted by vendor delays or geopolitical constraints affecting niche reagents. Moreover, the elimination of concentrated nitric acid as a bulk solvent reduces the regulatory burden associated with the storage and transport of hazardous corrosive materials, streamlining logistics and lowering insurance premiums.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the reduction in waste disposal costs and equipment maintenance. By avoiding the use of large volumes of strong mineral acids, the facility saves significantly on neutralization agents and wastewater treatment fees, which are often hidden costs in traditional nitration processes. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower overall carbon footprint and operational expenditure. The high atom economy of the tandem reaction means less raw material is wasted on side products, further driving down the cost per kilogram of the final active pharmaceutical ingredient or energetic material precursor.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent output quality, which is vital for maintaining long-term contracts with downstream customers. The tolerance for various functional groups means that a single platform technology can be used to synthesize a diverse library of analogues, allowing for rapid response to market demands without the need for extensive process re-validation. This flexibility reduces the lead time for new product introductions and strengthens the manufacturer's position as a reliable partner capable of adapting to changing project requirements swiftly and efficiently.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by the absence of highly exothermic steps that typically limit batch sizes in nitration chemistry. The use of solid nitrate salts allows for precise dosing and better heat management in large reactors, mitigating the risk of thermal incidents. From an environmental perspective, the process aligns with modern green chemistry principles by reducing the E-factor (mass of waste per mass of product) and minimizing the use of volatile organic compounds where possible. This compliance with stringent environmental regulations future-proofs the manufacturing site against tightening legislation and enhances the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders evaluating this technology for their supply chains.
Q: What are the primary advantages of using nitrate hydrate over concentrated nitric acid?
A: Using nitrate hydrates like Fe(NO3)3·9H2O eliminates the need for large volumes of concentrated nitric acid as a solvent, significantly reducing corrosion risks, waste disposal costs, and safety hazards associated with handling strong oxidizers in industrial settings.
Q: Can this method be applied to substrates with sensitive functional groups?
A: Yes, the mild reaction conditions (25-60°C) and the use of iron salts allow for excellent tolerance of various functional groups including esters, ethers, and halogens, making it suitable for complex pharmaceutical intermediate synthesis.
Q: What is the typical purity profile of the resulting geminal dinitro compounds?
A: The process yields high-purity products after standard column chromatography purification, with minimal byproduct formation due to the selective radical mechanism, ensuring compliance with stringent quality specifications for downstream applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Geminal Dinitro Compound Supplier
As the global demand for specialized pharmaceutical intermediates and high-performance energetic materials continues to rise, the ability to deliver complex molecules with consistent quality and competitive pricing is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the nitrate hydrate-mediated cyclization to meet the evolving needs of our clients. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of geminal dinitro compound meets the highest standards required for downstream drug development or material fabrication.
We invite you to collaborate with us to explore the full potential of this innovative synthesis route for your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this greener, safer process can optimize your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to accelerate your path to market with reliable, high-quality chemical solutions.
