Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic methodologies to access nitrogen-containing heterocycles functionalized with trifluoromethyl groups, as these motifs significantly enhance the metabolic stability, lipophilicity, and bioavailability of drug candidates. Patent CN111423381B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds that addresses many of the historical challenges associated with introducing fluorine atoms into heterocyclic scaffolds. This innovative approach utilizes a transition metal palladium-catalyzed carbonylation series reaction, leveraging cheap and easily obtained starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. By operating under remarkably mild conditions at 30°C, this technology offers a reliable pharmaceutical intermediate supplier pathway that minimizes energy consumption while maximizing substrate compatibility. The ability to synthesize diversely substituted imidazole derivatives through simple substrate design underscores the versatility of this method for modern medicinal chemistry applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the direct reaction of specialized synthons with suitable substrates, a process often fraught with significant operational difficulties. Commonly used trifluoromethyl synthons, such as trifluorodiazoethane and various trifluoroethylimidoyl halides, have historically suffered from limited availability, high costs, and safety concerns regarding their handling on a large scale. Furthermore, traditional methods frequently require harsh reaction conditions, including extreme temperatures or the use of hazardous reagents, which can lead to poor functional group tolerance and low overall yields. These limitations create substantial bottlenecks for procurement managers seeking cost reduction in API manufacturing, as the complexity of purification and the instability of intermediates often drive up production expenses. Additionally, the narrow substrate scope of older methodologies restricts the chemical space available to R&D directors, hindering the rapid exploration of structure-activity relationships essential for drug discovery programs.
The Novel Approach
In stark contrast to these legacy techniques, the novel methodology described in the patent employs a sophisticated yet operationally simple palladium-catalyzed multicomponent coupling strategy. This approach ingeniously combines trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts in the presence of a carbon monoxide surrogate generated in situ from formic acid and acetic anhydride. The reaction proceeds efficiently at a mild temperature of 30°C over a period of 16 to 24 hours, utilizing tetrahydrofuran as a preferred organic solvent to ensure high conversion rates. This mild thermal profile is particularly advantageous for preserving sensitive functional groups that might decompose under more vigorous conditions, thereby expanding the utility of the process for complex molecule synthesis. The use of commercially available catalysts like palladium chloride and triphenylphosphine further enhances the economic viability of the process, making it an attractive option for the commercial scale-up of complex pharmaceutical intermediates.
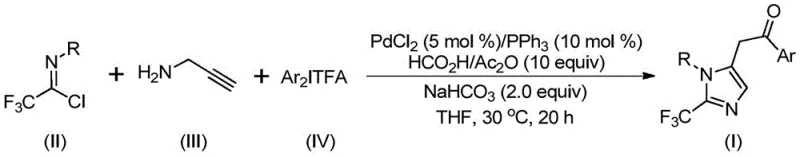
The general reaction scheme illustrates the convergence of three distinct building blocks into a single, highly functionalized heterocyclic product. This convergent synthesis strategy not only simplifies the retrosynthetic analysis for target molecules but also allows for the modular introduction of diversity at multiple positions on the imidazole ring. By varying the substituents on the aryl groups of the starting materials, chemists can rapidly generate libraries of compounds for biological screening without the need for lengthy linear synthetic sequences. This flexibility is a critical asset for supply chain heads concerned with reducing lead time for high-purity pharmaceutical intermediates, as it enables rapid response to changing project requirements.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
A deep understanding of the catalytic cycle is essential for R&D directors aiming to optimize this process for specific applications. The mechanism initiates with the formation of a trifluoroacetamidine compound through an intermolecular carbon-nitrogen bond promotion by alkali, followed by isomerization. Subsequently, the palladium catalyst activates the alkyne moiety of the propargylamine derivative, leading to palladation and the generation of an alkenyl palladium intermediate. This intermediate undergoes further isomerization to form a more stable alkyl palladium species, which is then poised for the crucial carbonylation step. The carbon monoxide required for this transformation is elegantly released from the decomposition of the formic acid and acetic anhydride mixture, serving as a safe and convenient alternative to handling pressurized CO gas. This in situ generation of CO ensures a steady concentration of the carbonyl source, driving the reaction towards the formation of the acyl palladium intermediate.
Following the carbonylation event, the diaryl iodonium salt participates in the cycle by undergoing oxidative addition with the palladium center, generating a high-valent tetravalent palladium intermediate. This step is critical for introducing the aryl group at the appropriate position on the developing heterocycle. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl-substituted imidazole product and regenerates the active palladium catalyst for subsequent turnover. The high yields observed across a range of substrates, as evidenced by the successful synthesis of compounds I-1 through I-5 with yields ranging from 65% to 97%, attest to the robustness of this mechanistic pathway. The tolerance for various substituents, including electron-donating groups like methoxy and tert-butyl, as well as electron-withdrawing groups like nitro and halogens, demonstrates the broad applicability of this catalytic system.
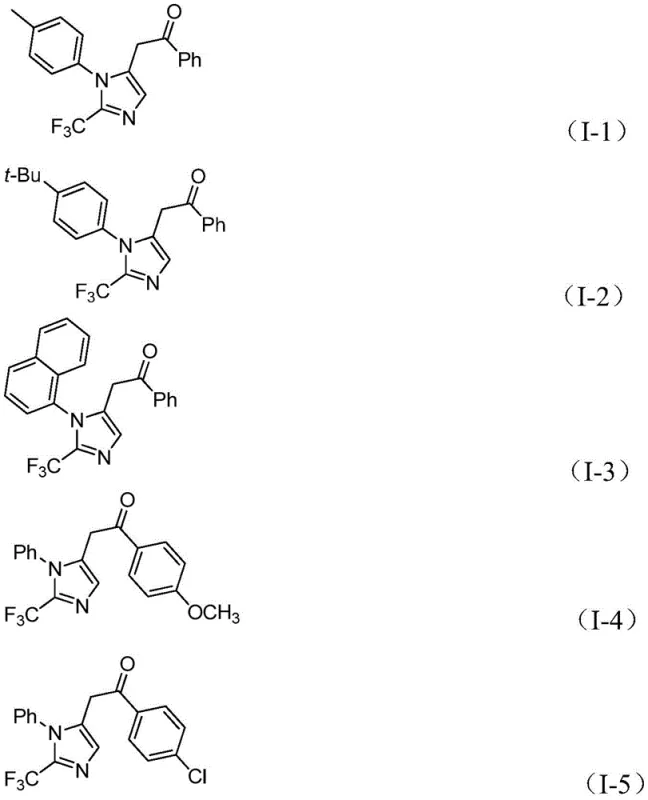
The structural diversity achievable through this method is highlighted by the specific examples provided in the patent data. Compounds such as I-1, featuring a 4-methylphenyl group, and I-2, containing a bulky tert-butyl group, are obtained in excellent yields, confirming that steric hindrance does not significantly impede the reaction efficiency. Similarly, the successful incorporation of naphthyl groups in compound I-3 and various substituted phenyl rings in compounds I-4 and I-5 validates the method's utility for creating complex molecular architectures. This level of control over the final structure is paramount for ensuring the purity and quality of the resulting API intermediates, meeting the stringent specifications required by regulatory bodies.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and order of addition to maximize yield and minimize byproduct formation. The protocol suggests a molar ratio of palladium chloride to triphenylphosphine to sodium bicarbonate of approximately 0.05:0.1:2, ensuring sufficient catalytic activity and base capacity to neutralize acidic byproducts. The reaction mixture typically includes acetic anhydride and formic acid in a 10 equivalent ratio relative to the limiting reagent to drive the carbonylation forward. While the detailed step-by-step operational procedure involves specific mixing and heating protocols, the core principle remains the controlled combination of the three key precursors under inert atmosphere conditions. For those seeking a standardized protocol to replicate these results, the following guide outlines the essential phases of the synthesis.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers compelling advantages that directly address the pain points of procurement managers and supply chain leaders. The reliance on commodity chemicals such as propargylamine and aromatic amines, which are widely available in the global market, mitigates the risk of supply chain disruptions often associated with exotic or proprietary reagents. Furthermore, the elimination of cryogenic conditions and the use of ambient pressure reactions significantly reduce the energy footprint of the manufacturing process, contributing to substantial cost savings in utility expenditures. The simplicity of the post-treatment process, which involves basic filtration and standard column chromatography, streamlines the purification workflow and reduces the demand for specialized equipment or extensive labor hours. These factors collectively enhance the overall economic efficiency of producing high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The use of inexpensive palladium chloride instead of more exotic noble metal catalysts, combined with the avoidance of high-pressure carbon monoxide cylinders, drastically lowers the capital and operational expenditure required for production. The high atom economy of the multicomponent reaction ensures that a significant proportion of the starting material mass is incorporated into the final product, minimizing waste disposal costs. Additionally, the mild reaction conditions reduce the need for expensive cooling or heating infrastructure, allowing for processing in standard glass-lined reactors. These cumulative efficiencies translate into a more competitive pricing structure for the final intermediates without compromising on quality.
- Enhanced Supply Chain Reliability: Since the key starting materials like trifluoroethylimidoyl chloride and diaryl iodonium salts can be synthesized from readily available precursors, the supply chain is less vulnerable to single-source bottlenecks. The robustness of the reaction across a wide range of substrates means that if a specific aryl group becomes unavailable, alternative analogs can be synthesized with minimal process re-optimization. This flexibility ensures continuous production capability even when facing fluctuations in raw material availability. Moreover, the scalability of the process from milligram to gram levels, as demonstrated in the patent examples, provides a clear pathway for scaling up to multi-kilogram or ton quantities to meet commercial demand.
- Scalability and Environmental Compliance: The reaction generates minimal hazardous waste compared to traditional methods that might utilize toxic heavy metals or generate large volumes of saline effluent. The use of tetrahydrofuran as a solvent, which is recyclable, aligns with green chemistry principles and simplifies compliance with increasingly stringent environmental regulations. The absence of gaseous reagents like pure CO eliminates the need for complex gas handling systems and safety protocols related to toxic gas leaks. Consequently, facilities can achieve higher throughput with lower environmental impact, supporting sustainable manufacturing goals while maintaining high production standards.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common inquiries based on the patent specifications. These questions address the practical aspects of implementation, safety considerations, and the potential for customization. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this synthetic route into their existing manufacturing portfolios. The answers provided below reflect the specific data and embodiments disclosed in the intellectual property documentation.
Q: What are the key advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: This method operates under mild conditions (30°C) using readily available starting materials like propargylamine and trifluoroethylimidoyl chloride, avoiding the harsh conditions and limited substrate scope often associated with direct trifluoromethylation reagents.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with high reaction efficiency and simple post-processing, making it viable for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the role of the diaryl iodonium salt in this reaction mechanism?
A: The diaryl iodonium salt acts as an arylating agent that undergoes oxidative addition with the palladium intermediate, facilitating the formation of the final imidazole ring structure through a tetravalent palladium species.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, leveraging advanced catalytic technologies like the one described in CN111423381B to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-trifluoromethyl imidazole meets the highest industry standards for pharmaceutical applications. Our commitment to quality and consistency makes us a trusted partner for global enterprises seeking to secure their supply chains for critical heterocyclic intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative palladium-catalyzed route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this method for your specific target molecules. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on empirical evidence and expert analysis. Let us collaborate to accelerate your drug development timelines through superior chemical manufacturing.
