Scalable Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Derivatives for Pharmaceutical Applications
Introduction to Next-Generation Imidazole Synthesis
The strategic incorporation of trifluoromethyl groups into heterocyclic scaffolds represents a cornerstone of modern medicinal chemistry, significantly enhancing the metabolic stability, lipophilicity, and bioavailability of drug candidates. As highlighted in recent literature, imidazole derivatives serve as critical backbones for a vast array of bioactive molecules, ranging from antihistamines to complex natural products like Naamidine A. 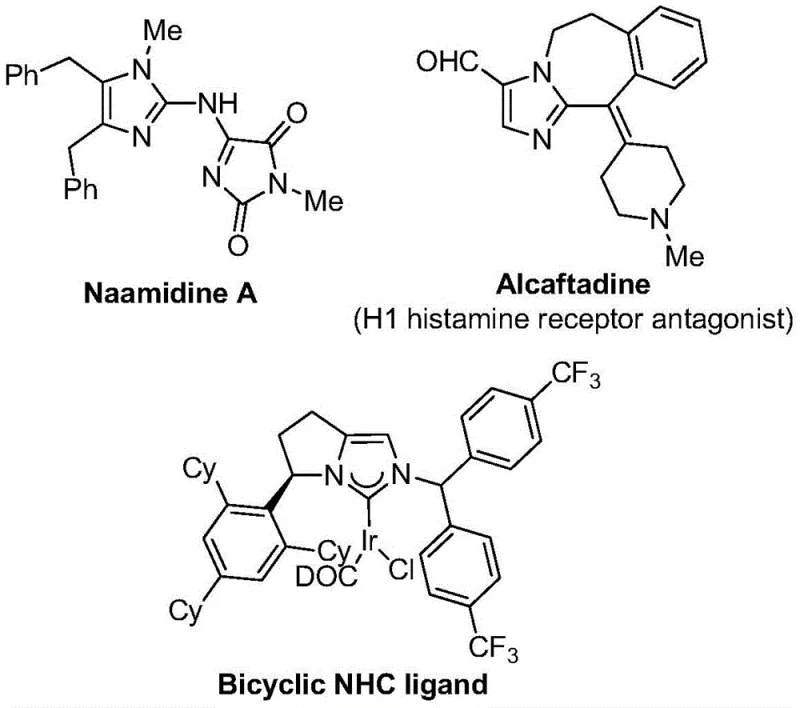 Patent CN111423381B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds that addresses long-standing synthetic challenges. This innovative approach leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as key building blocks. By operating under remarkably mild conditions, this technology not only streamlines the construction of these valuable nitrogen-containing five-membered heterocycles but also opens new avenues for the efficient synthesis of diversified molecular libraries essential for drug discovery pipelines.
Patent CN111423381B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds that addresses long-standing synthetic challenges. This innovative approach leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts as key building blocks. By operating under remarkably mild conditions, this technology not only streamlines the construction of these valuable nitrogen-containing five-membered heterocycles but also opens new avenues for the efficient synthesis of diversified molecular libraries essential for drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted nitrogen-containing heterocycles has been fraught with significant operational hurdles and chemical limitations. Traditional methodologies often rely on the direct reaction of synthons bearing trifluoromethyl groups with suitable substrates, frequently necessitating the use of hazardous reagents such as trifluorodiazoethane or requiring harsh reaction conditions that compromise functional group tolerance. These conventional routes typically suffer from poor atom economy, limited substrate scope, and the generation of complex impurity profiles that are difficult to separate, thereby inflating the cost of goods and extending development timelines. Furthermore, the reliance on specialized trifluoromethylating agents that are not widely commercially available creates supply chain bottlenecks, making it difficult for procurement teams to secure consistent quality raw materials for large-scale manufacturing campaigns.
The Novel Approach
In stark contrast to these legacy methods, the novel pathway described in the patent utilizes a highly modular and efficient palladium-catalyzed cascade reaction that fundamentally reshapes the synthetic landscape for these intermediates. 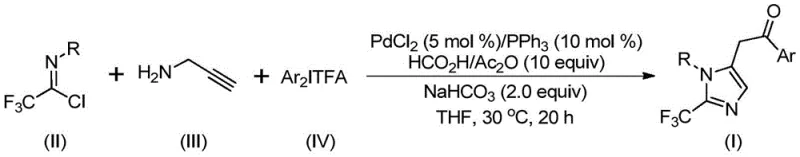 This transformative approach employs cheap and easily obtained starting materials, specifically trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts, which are reacted in the presence of a palladium catalyst and a carbon monoxide surrogate system. The reaction proceeds smoothly at a low temperature of 30°C, demonstrating exceptional compatibility with various functional groups including halogens, alkyls, and nitro groups. This mildness allows for the precise construction of the imidazole core with high regioselectivity and yield, effectively bypassing the safety risks associated with high-pressure CO gas and enabling the rapid generation of diverse analogues for structure-activity relationship (SAR) studies without the need for extensive process optimization.
This transformative approach employs cheap and easily obtained starting materials, specifically trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts, which are reacted in the presence of a palladium catalyst and a carbon monoxide surrogate system. The reaction proceeds smoothly at a low temperature of 30°C, demonstrating exceptional compatibility with various functional groups including halogens, alkyls, and nitro groups. This mildness allows for the precise construction of the imidazole core with high regioselectivity and yield, effectively bypassing the safety risks associated with high-pressure CO gas and enabling the rapid generation of diverse analogues for structure-activity relationship (SAR) studies without the need for extensive process optimization.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The mechanistic elegance of this transformation lies in its intricate multi-step catalytic cycle, which seamlessly integrates C-N bond formation, alkyne activation, and carbonylation into a single operational pot. The process initiates with the base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, generating a trifluoroacetamidine intermediate that subsequently undergoes isomerization. Following this, the palladium catalyst activates the alkyne moiety through palladation to form a vinyl-palladium species, which then isomerizes to a more stable alkyl-palladium intermediate. Crucially, the carbon monoxide required for the carbonylation step is generated in situ from the decomposition of formic acid and acetic anhydride, inserting into the palladium-carbon bond to create an acyl-palladium complex. This sequence avoids the logistical nightmares of handling gaseous CO while ensuring high local concentrations of the carbonyl source at the catalytic center.
The final stages of the catalytic cycle involve the oxidative addition of the diaryl iodonium salt to the acyl-palladium intermediate, forming a high-valent tetravalent palladium species that facilitates the final C-C bond formation. Subsequent reductive elimination releases the desired 2-trifluoromethyl-substituted imidazole product and regenerates the active palladium(0) catalyst to continue the cycle. This sophisticated mechanism ensures high reaction efficiency and minimizes the formation of side products, as the oxidative addition of the hypervalent iodine species is highly selective. The use of sodium bicarbonate as an additive further aids in neutralizing acidic byproducts, maintaining the integrity of the catalytic system and preventing catalyst deactivation, which is critical for achieving the high yields reported across a broad range of electron-rich and electron-deficient substrates.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the catalyst system and the order of reagent addition to maximize conversion. The protocol dictates a precise molar ratio of palladium chloride to triphenylphosphine to sodium bicarbonate, typically optimized at 0.05:0.1:2 relative to the limiting substrate, ensuring sufficient catalytic turnover while minimizing metal loading costs. The reaction is conducted in an aprotic organic solvent, with tetrahydrofuran (THF) identified as the preferred medium due to its ability to effectively dissolve all reactants and promote the reaction kinetics. Detailed standardized synthetic steps, including specific workup procedures involving silica gel mixing and column chromatography purification, are outlined below to ensure reproducibility and high purity of the final isolates.
- Prepare the reaction mixture by adding palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid into an organic solvent such as THF.
- Introduce the substrates: trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt into the catalytic system under stirring.
- Maintain the reaction at 30°C for 16-24 hours, followed by filtration and column chromatography purification to isolate the target imidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic benefits that extend far beyond simple chemical yield improvements. The shift towards using commodity chemicals like propargylamine and readily synthesizable diaryl iodonium salts drastically reduces the dependency on exotic, high-cost fluorinating reagents that often plague the supply chains of fluorinated pharmaceutical intermediates. By eliminating the need for specialized high-pressure equipment required for traditional carbonylation reactions, capital expenditure for manufacturing facilities is significantly reduced, allowing for more flexible production scheduling and lower overhead costs. Furthermore, the mild reaction temperature of 30°C translates to lower energy consumption during the reaction phase, contributing to a greener manufacturing profile and reduced utility expenses over the lifecycle of the product.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive palladium chloride as the catalyst source, which is markedly cheaper than many specialized palladium complexes used in cross-coupling reactions. The elimination of hazardous gaseous carbon monoxide removes the need for expensive safety infrastructure and monitoring systems, leading to substantial cost savings in both CAPEX and OPEX. Additionally, the high atom economy and selectivity of the reaction minimize waste generation, reducing the costs associated with waste disposal and environmental compliance, thereby optimizing the overall cost structure for API manufacturing.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved as the key starting materials, including various substituted aromatic amines and aryl boronic acids used to prepare the precursors, are widely available from global chemical suppliers. This diversity in sourcing options mitigates the risk of single-supplier dependency and ensures business continuity even during market fluctuations. The robustness of the reaction conditions means that production is less susceptible to variations in raw material quality, providing a stable and predictable output that allows supply chain planners to maintain leaner inventory levels without compromising on delivery commitments to downstream customers.
- Scalability and Environmental Compliance: The simplicity of the post-treatment procedure, which involves basic filtration and standard chromatographic techniques, facilitates easy scale-up from gram to kilogram and eventually to ton-scale production without encountering significant engineering barriers. The process aligns well with modern environmental, health, and safety (EHS) standards by avoiding the use of toxic gases and minimizing the generation of heavy metal waste, as the palladium loading is kept low. This environmental friendliness simplifies the regulatory approval process for new drug applications and supports corporate sustainability goals, making it an attractive option for companies aiming to reduce their carbon footprint in fine chemical synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is critical for R&D teams evaluating the feasibility of integrating this route into their existing process development workflows. The answers provided reflect the specific conditions and outcomes observed during the validation of this method, ensuring that expectations regarding yield and purity are grounded in empirical evidence.
Q: What are the primary advantages of this palladium-catalyzed method over traditional synthesis?
A: This method operates under exceptionally mild conditions (30°C) compared to traditional high-temperature cyclizations, utilizes cheap and readily available starting materials like propargylamine, and offers excellent functional group tolerance for diverse substrate design.
Q: What is the role of formic acid and acetic anhydride in this reaction?
A: Formic acid and acetic anhydride serve as an in situ carbon monoxide (CO) surrogate system, generating the necessary CO for the carbonylation step without requiring hazardous high-pressure CO gas cylinders, thereby enhancing operational safety.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the protocol is designed for scalability, utilizing simple post-treatment procedures like filtration and standard column chromatography, and employs inexpensive catalysts like PdCl2, making it viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality fluorinated heterocycles play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-trifluoromethyl imidazole intermediate meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to navigate the complexities of palladium-catalyzed reactions, delivering products with consistent quality and minimal impurity profiles.
We invite pharmaceutical partners to leverage our expertise for their upcoming projects by requesting a Customized Cost-Saving Analysis tailored to their specific volume requirements. Our technical procurement team is ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative synthesis can accelerate your development timelines. By collaborating with us, you gain access to a reliable supply chain partner dedicated to driving down costs while maintaining the highest levels of chemical integrity and service reliability.
