Advanced Synthesis of Benzophenone Hydrazone for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries constantly seek robust synthetic routes for critical intermediates that balance high purity with economic viability. Patent CN102503853A, published in June 2012, introduces a significant advancement in the preparation of Benzophenone Hydrazone, a pivotal compound utilized extensively in the synthesis of cardiovascular agents and beta-lactam antibiotics. This specific methodology addresses long-standing challenges in hydrazone formation by utilizing a refined solvent system that promotes superior reaction kinetics and product isolation. By leveraging a reflux condensation technique in the presence of ethylene glycol, the process achieves exceptional conversion rates without the need for complex catalytic systems or hazardous reagents. For R&D directors and process chemists, this patent represents a validated pathway to secure a reliable supply of high-purity intermediates essential for downstream drug manufacturing. The technical robustness of this approach ensures that the resulting material meets the stringent quality specifications required for active pharmaceutical ingredient (API) synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for hydrazones often suffer from inconsistent yields and difficult purification protocols that can bottleneck large-scale production. Conventional methods frequently rely on volatile organic solvents like ethanol or methanol, which may require extended reaction times or the use of acid catalysts that complicate the workup procedure. These older techniques often result in the formation of side products or incomplete conversion, necessitating energy-intensive recrystallization steps to achieve the necessary purity levels for pharmaceutical applications. Furthermore, the handling of hydrazine derivatives in low-boiling solvents can present safety hazards due to volatility and potential exothermic risks during scale-up. The variability in crystal morphology and yield in these traditional processes creates uncertainty in supply chain planning, making it difficult for procurement managers to forecast material availability accurately. Consequently, manufacturers face higher operational costs associated with waste disposal and solvent recovery, diminishing the overall economic efficiency of the production line.
The Novel Approach
The innovative method disclosed in the patent overcomes these deficiencies by employing ethylene glycol as a high-boiling reaction medium, which fundamentally alters the thermodynamic landscape of the condensation reaction. By conducting the reaction under reflux conditions for a controlled duration of 1.5 to 2.5 hours, the system ensures complete interaction between the benzophenone and hydrazine hydrate reactants. This thermal environment facilitates the efficient removal of water generated during the condensation, driving the equilibrium strongly towards the formation of the desired hydrazone product. The subsequent cooling protocol, involving natural cooling to room temperature followed by precise chilling below 5°C, induces the formation of well-defined white needle-shaped crystals that are easily separated by filtration. This streamlined workflow eliminates the need for complex extraction procedures, significantly reducing the processing time and labor intensity required for isolation. The result is a process that not only delivers yields consistently above 95% but also produces a product with superior physical characteristics suitable for immediate use in subsequent synthetic steps.
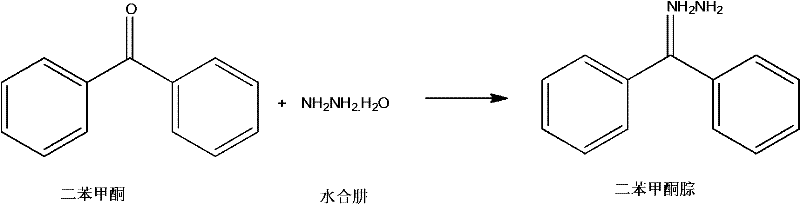
Mechanistic Insights into Ethylene Glycol-Mediated Condensation
The core chemical transformation involves a nucleophilic addition-elimination mechanism where the nitrogen atom of the hydrazine hydrate attacks the electrophilic carbonyl carbon of the benzophenone. In the presence of ethylene glycol, the solvation of the transition state is optimized, lowering the activation energy required for the initial addition step. The high boiling point of the solvent allows the reaction mixture to be maintained at elevated temperatures without significant pressure buildup, ensuring that the kinetic energy of the molecules is sufficient to overcome steric hindrance around the carbonyl group. As the reaction proceeds, the elimination of a water molecule occurs, stabilized by the hydrogen bonding network of the glycol solvent, which effectively sequesters the byproduct and prevents the reverse hydrolysis reaction. This mechanistic pathway is highly selective, minimizing the formation of azine byproducts that are common in hydrazine reactions when stoichiometry is not strictly controlled. The precise control of reaction time between 1.5 and 2.5 hours is critical to maximizing the forward reaction rate while preventing thermal degradation of the sensitive hydrazone linkage.
Impurity control in this synthesis is inherently managed through the crystallization dynamics dictated by the solvent system. As the reaction mixture cools from reflux temperature to below 5°C, the solubility of the benzophenone hydrazone decreases sharply, promoting rapid nucleation and crystal growth. The specific cooling profile ensures that impurities, such as unreacted starting materials or trace hydrazine, remain dissolved in the mother liquor rather than being occluded within the crystal lattice. The use of cold alcohol washing during the filtration step further purifies the surface of the crystals, removing any adhering solvent or soluble contaminants. This rigorous control over the solid-state formation results in a product with a narrow particle size distribution and high chemical purity, which is essential for maintaining consistent reactivity in downstream coupling reactions. For quality assurance teams, this predictable crystallization behavior simplifies the validation of the manufacturing process and ensures batch-to-batch consistency.
How to Synthesize Benzophenone Hydrazone Efficiently
Implementing this synthesis route requires careful attention to the ratios of reactants and the thermal profile of the reaction vessel to ensure optimal performance. The process begins with the charging of ethylene glycol and hydrazine hydrate into a reactor equipped with efficient stirring and cooling capabilities, followed by the addition of benzophenone. The mixture is then heated to reflux, where it is maintained for a specific window to allow full conversion before initiating the controlled cooling sequence. Detailed standardized operating procedures regarding the exact stirring speeds and cooling rates are essential for reproducing the high yields reported in the patent data.
- Mix Benzophenone and Hydrazine Hydrate (80%) in ethylene glycol solvent under stirring.
- Heat the mixture to reflux temperature and maintain reaction for 1.5 to 2.5 hours to ensure complete conversion.
- Cool naturally to room temperature, then further cool below 5°C to induce crystallization, followed by filtration and vacuum drying at 50°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers substantial advantages that directly impact the bottom line and supply chain resilience for pharmaceutical manufacturers. The elimination of complex catalytic systems and the use of readily available, low-cost solvents like ethylene glycol significantly reduce the raw material expenditure per kilogram of product. The high yield efficiency means that less starting material is wasted, leading to a drastic reduction in the cost of goods sold (COGS) and minimizing the environmental footprint associated with waste treatment. For procurement managers, the simplicity of the process translates to a more reliable supply source, as the manufacturing process is less prone to failure or deviation compared to more complex multi-step syntheses. The ability to produce high-purity material with minimal downstream processing reduces the lead time required to bring batches to market, enhancing the agility of the supply chain in responding to fluctuating demand for antibiotic intermediates.
- Cost Reduction in Manufacturing: The process utilizes ethylene glycol, a commodity chemical with a stable global supply chain, replacing more expensive or specialized solvents often required in traditional hydrazone synthesis. By achieving yields consistently exceeding 95%, the process maximizes the utilization of benzophenone and hydrazine hydrate, effectively lowering the unit cost of production. The simplified workup procedure, which avoids extensive extraction and chromatography, reduces labor costs and energy consumption associated with solvent recovery and distillation. Furthermore, the high purity of the crude product minimizes the need for resource-intensive recrystallization steps, contributing to overall operational savings.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures high batch success rates, reducing the risk of production delays caused by failed reactions or off-specification material. Since the raw materials are bulk chemicals with widespread availability, the supply chain is less vulnerable to shortages of niche reagents or catalysts. The scalability of the process, demonstrated by the use of standard reflux and filtration equipment, allows manufacturers to easily ramp up production volumes to meet surges in demand for downstream antibiotics. This reliability provides a strategic advantage for supply chain heads who need to guarantee continuous material flow for critical drug manufacturing lines.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory glassware to industrial reactors without significant changes in reaction parameters or safety profiles. The use of ethylene glycol, which has a low vapor pressure, reduces volatile organic compound (VOC) emissions compared to processes using alcoholic solvents, aiding in compliance with increasingly strict environmental regulations. The solid waste generated is minimal due to the high conversion efficiency, and the aqueous waste streams are easier to treat due to the absence of heavy metal catalysts or toxic organic residues. This environmental compatibility simplifies the permitting process for new production facilities and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Benzophenone Hydrazone based on the patented technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their specific manufacturing workflows. The answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation.
Q: What is the primary application of Benzophenone Hydrazone in antibiotic synthesis?
A: Benzophenone Hydrazone serves as a critical precursor and protecting group agent in the synthesis of beta-lactam antibiotics such as Cefixime and Tazobactam Sodium, specifically protecting the carboxyl groups of 6-APA or 7-ACA.
Q: How does the ethylene glycol method improve yield compared to traditional solvents?
A: The use of ethylene glycol as a high-boiling solvent facilitates better solubility and reaction kinetics during reflux, allowing the process to achieve consistent yields exceeding 95% while simplifying the post-reaction crystallization process.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's suitability for industrial production due to its simple operation, safety profile, and the ability to recover high-purity white needle-shaped crystals through standard filtration and drying techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzophenone Hydrazone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development and commercialization of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Benzophenone Hydrazone meets the highest industry standards for pharmaceutical applications. Our commitment to quality assurance means that you can rely on us for a steady supply of material that supports your regulatory filings and production schedules without interruption.
We invite you to contact our technical procurement team to discuss how our advanced synthesis capabilities can support your specific project needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that demonstrates how our optimized processes can reduce your overall manufacturing expenses. We encourage you to reach out today to request specific COA data and route feasibility assessments tailored to your unique requirements. Let us be your trusted partner in navigating the complexities of fine chemical sourcing and accelerating your path to market.
