Scalable Palladium-Catalyzed Dehydrogenative Coupling for High-Purity Enamide Intermediates
Scalable Palladium-Catalyzed Dehydrogenative Coupling for High-Purity Enamide Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective synthetic routes. A significant breakthrough in this domain is detailed in patent CN114014802A, which discloses a novel method for synthesizing enamide compounds via palladium-catalyzed dehydrogenative coupling. This technology represents a paradigm shift from traditional stoichiometric approaches to a more elegant catalytic cycle, enabling the direct functionalization of 2-pyridone compounds with acrylic acid derivatives. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition: the ability to access complex nitrogen-containing scaffolds with high purity and yield while bypassing the logistical and economic burdens associated with pre-functionalized starting materials. The methodology leverages the power of transition metal catalysis to forge carbon-nitrogen bonds under remarkably mild conditions, positioning it as a cornerstone technique for the next generation of fine chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of enamide structures from 2-pyridone precursors has been fraught with significant chemical and operational challenges that hinder efficient commercial production. The traditional reliance on nucleophilic substitution reactions necessitates the use of halogenated olefins, which are not only expensive to procure but also require energy-intensive preparation steps prior to the main coupling event. Furthermore, these classical routes typically demand strongly basic reaction conditions to drive the substitution, which often leads to poor atom economy and generates substantial quantities of salt waste that complicate downstream purification and environmental compliance. As illustrated in the conventional pathway below, the requirement for harsh reagents limits the substrate scope, rendering many sensitive functional groups incompatible with the reaction environment.
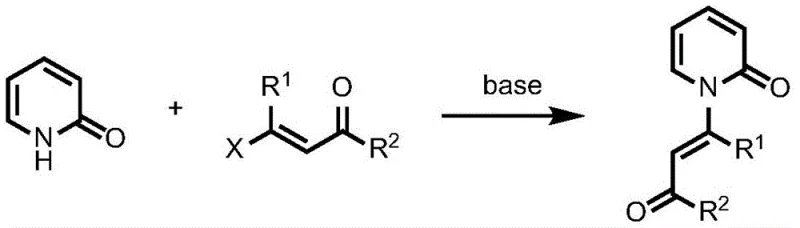
In addition to the issues with halogenated olefins, alternative historical methods involving the addition of propiolic acid derivatives under alkaline conditions present their own set of drawbacks. These processes are often characterized by narrow applicability, failing to accommodate a diverse range of substituents on the pyridone ring, which restricts the chemical space available for drug discovery and development. The combination of expensive starting materials, harsh reaction parameters, and limited generality creates a bottleneck in the supply chain, forcing manufacturers to accept lower yields and higher production costs. Consequently, there has been a persistent industry demand for a more robust and versatile synthetic strategy that can overcome these inherent inefficiencies without compromising on product quality or safety standards.
The Novel Approach
The innovative methodology described in the patent data introduces a transformative solution by employing a palladium-catalyzed dehydrogenative coupling mechanism that directly links 2-pyridones with acrylic acid derivatives. This approach elegantly circumvents the need for pre-halogenated substrates, thereby streamlining the synthetic sequence and reducing the overall material footprint of the process. By utilizing palladium acetate as a catalyst in conjunction with a silver-based oxidant, the reaction proceeds through a catalytic cycle that activates the C-H bond of the pyridone ring, allowing for direct coupling with the alkene moiety of the acrylate. This shift from stoichiometric substitution to catalytic C-H activation not only enhances the atom economy but also significantly broadens the scope of compatible substrates, enabling the synthesis of a wide array of enamide derivatives with diverse electronic and steric properties.
Mechanistic Insights into Pd-Catalyzed Dehydrogenative Coupling
At the heart of this technological advancement lies a sophisticated catalytic cycle driven by the interplay between the palladium center and the oxidant. The reaction initiates with the coordination of the palladium catalyst to the 2-pyridone substrate, facilitating the activation of the specific carbon-hydrogen bond adjacent to the nitrogen atom. This C-H activation step is critical, as it generates a reactive organopalladium intermediate that is poised for subsequent insertion of the acrylic acid derivative. The presence of the silver oxidant plays a dual role: it serves to regenerate the active palladium species from its reduced state, ensuring the continuity of the catalytic turnover, and it assists in the final elimination step to restore aromaticity and release the enamide product. This mechanistic pathway is highly efficient, requiring only catalytic amounts of the metal complex (often as low as 0.1 equivalent of oxidant relative to the catalyst loading) to drive the transformation to completion.
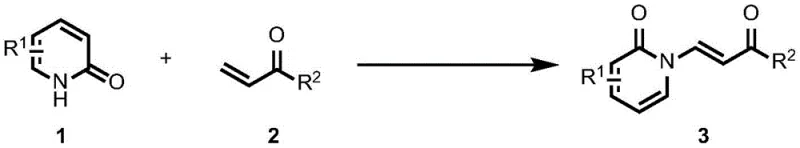
From an impurity control perspective, this mechanism offers distinct advantages over traditional nucleophilic attacks. Because the reaction does not rely on highly reactive nucleophiles or electrophiles that can lead to indiscriminate side reactions, the formation of by-products is inherently minimized. The mild reaction conditions, typically ranging from 40°C to 100°C, further suppress thermal degradation pathways that often plague high-temperature syntheses. Moreover, the regioselectivity of the C-H activation ensures that the coupling occurs specifically at the desired position on the pyridone ring, eliminating the need for complex protecting group strategies or difficult separation processes. This high level of control translates directly into a cleaner crude reaction profile, which simplifies the purification workflow and ensures that the final API intermediate meets the stringent purity specifications required by global regulatory bodies.
How to Synthesize Enamide Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting involves a straightforward protocol that balances reaction efficiency with operational simplicity. The process begins by dissolving the chosen 2-pyridone compound and the acrylic acid derivative in a suitable organic solvent, such as 1,2-dichloroethane, toluene, or chlorobenzene, creating a homogeneous reaction mixture. To this solution, the palladium acetate catalyst and the silver oxidant are added, initiating the dehydrogenative coupling upon heating. The reaction is typically maintained under stirring for a period of approximately 24 hours at temperatures around 60°C, although the patent indicates flexibility within the 40-100°C range depending on the specific substrate reactivity. Following the reaction completion, the mixture is filtered through kieselguhr to remove metal residues, and the filtrate is concentrated and purified via standard column chromatography to isolate the high-purity enamide product.
- Dissolve the 2-pyridone compound and acrylic acid derivative in an organic solvent such as 1,2-dichloroethane or toluene within a reaction vessel.
- Add palladium acetate catalyst and a silver-based oxidant (e.g., silver trifluoroacetate) to the mixture under stirring.
- Heat the reaction mixture to 40-100°C for approximately 24 hours, then filter through kieselguhr and purify the filtrate via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed technology offers tangible benefits that extend far beyond the laboratory bench. The primary advantage lies in the drastic simplification of the raw material supply chain; by eliminating the need for specialized halogenated olefins, manufacturers can source standard, commodity-grade acrylic acid derivatives that are available in bulk quantities from multiple global suppliers. This diversification of the supply base mitigates the risk of shortages and price volatility associated with niche intermediates, ensuring a more stable and predictable production schedule. Furthermore, the removal of strong bases and the reduction in waste generation align perfectly with modern environmental, health, and safety (EHS) mandates, potentially lowering the costs associated with waste disposal and regulatory compliance.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of expensive pre-functionalized starting materials and the reduction in processing steps. By avoiding the synthesis of halogenated olefins, companies save on both the direct material costs and the operational expenses related to their preparation and handling. Additionally, the high atom economy of the dehydrogenative coupling means that a greater proportion of the input mass is converted into the desired product, reducing the overall consumption of raw materials per kilogram of output. The mild reaction conditions also contribute to energy savings, as the process does not require extreme heating or cooling, further optimizing the utility costs associated with large-scale manufacturing operations.
- Enhanced Supply Chain Reliability: The reliance on widely available 2-pyridones and acrylic acid derivatives significantly enhances the resilience of the supply chain. Unlike proprietary or hard-to-source reagents, these building blocks are produced by numerous chemical manufacturers worldwide, reducing dependency on single-source suppliers. This abundance ensures that production timelines are not held hostage by external supply disruptions, allowing for consistent delivery of critical pharmaceutical intermediates. Moreover, the robustness of the reaction across a wide range of substrates means that the same process platform can be adapted for various analogues without requiring extensive re-optimization, accelerating the time-to-market for new drug candidates.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage levels is facilitated by its operational simplicity and the use of common industrial solvents. The absence of hazardous reagents like strong alkalis or unstable halides simplifies the engineering controls required for safe operation, making it easier to transfer the process from R&D to commercial production facilities. From an environmental standpoint, the generation of less chemical waste and the potential for catalyst recycling contribute to a greener manufacturing footprint. This alignment with sustainability goals is increasingly important for maintaining partnerships with major pharmaceutical companies that prioritize green chemistry principles in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed enamide synthesis. These insights are derived directly from the experimental data and technical disclosures found in the patent literature, providing a reliable foundation for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production workflows and for assessing its potential impact on project timelines and budgets.
Q: What are the primary advantages of this Pd-catalyzed method over traditional nucleophilic substitution?
A: Unlike traditional methods requiring harsh basic conditions and pre-functionalized halogenated olefins, this palladium-catalyzed approach utilizes readily available acrylic acid derivatives under mild conditions, significantly improving atom economy and substrate compatibility.
Q: Does this process require expensive or exotic reagents?
A: No, the process relies on commercially accessible raw materials such as 2-pyridones and standard acrylic acid derivatives, utilizing catalytic amounts of palladium acetate and silver salts which can be optimized for cost-efficiency in large-scale production.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the reaction operates at moderate temperatures (40-100°C) and uses common organic solvents, making it highly amenable to commercial scale-up with simplified downstream processing compared to multi-step traditional routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enamide Supplier
As the demand for high-quality enamide intermediates continues to grow within the pharmaceutical and fine chemical sectors, partnering with an experienced CDMO becomes a strategic imperative. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to reality. Our technical team is adept at optimizing catalytic cycles like the one described in CN114014802A, guaranteeing stringent purity specifications and rigorous QC labs oversight to meet the highest international standards. We understand that consistency and reliability are paramount in the supply of critical intermediates, and our infrastructure is designed to deliver exactly that.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits specific to your target molecule. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your supply chain with efficient, scalable, and cost-effective solutions.
