Revolutionizing Benzimidazole Production: A Green One-Pot Catalytic Strategy for Industrial Scale
Revolutionizing Benzimidazole Production: A Green One-Pot Catalytic Strategy for Industrial Scale
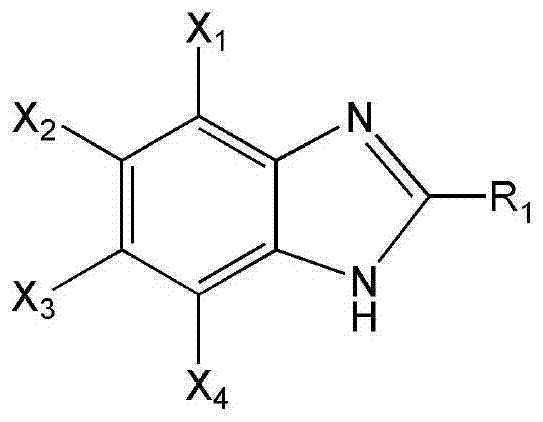
The pharmaceutical and agrochemical industries are constantly seeking more efficient, environmentally benign pathways for synthesizing critical heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN103288743A, which discloses a novel one-pot method for synthesizing benzimidazole compounds expressed by formula (III). This innovative approach utilizes ortho-nitroaniline compounds and fatty alcohols as primary raw materials, employing water as the reaction solvent under the influence of a robust supported metal solid catalyst. The core of this technology lies in the utilization of a specialized Cu-Zn-Pd/Al2O3 catalyst, which facilitates the transformation under shielding gas atmospheres without the need for corrosive liquid acids or volatile organic solvents. This represents a paradigm shift from traditional multi-step processes, offering a streamlined route that aligns perfectly with modern green chemistry principles and industrial sustainability goals. For a reliable benzimidazole supplier, adopting such methodologies ensures not only regulatory compliance but also substantial operational efficiencies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of benzimidazole or substituted benzimidazole derivatives has relied heavily on the condensation of o-phenylenediamine with aldehydes, carboxylic acids, or ester derivatives. These traditional pathways, while chemically sound, are fraught with significant logistical and economic drawbacks that hinder large-scale optimization. Most notably, these reactions typically require the presence of strong acidic catalysts, such as phosphoric acid, polyphosphoric acid, or hydrochloric acid, which necessitate complex neutralization and waste treatment protocols post-reaction. Furthermore, many existing literature methods utilize expensive starting materials like o-phenylenediamine, which drives up the raw material costs significantly compared to nitro-based precursors. Some alternative routes involve the use of hazardous oxidants like hydrogen peroxide or stannous chloride, creating severe environmental burdens and requiring specialized corrosion-resistant equipment. Additionally, the reliance on organic solvents such as tetrahydrofuran or DMF introduces volatility risks and complicates the downstream purification processes, often leading to lower overall yields due to difficult product isolation. Consequently, the industry has long suffered from processes that are neither cost-effective nor environmentally sustainable.
The Novel Approach
In stark contrast, the methodology described in patent CN103288743A offers a transformative solution by leveraging a one-pot synthesis strategy that directly couples o-nitroaniline compounds with fatty alcohols. This novel approach eliminates the need for pre-reduction steps or separate cyclization stages, effectively consolidating the entire synthetic sequence into a single reactor vessel. By employing water as the sole reaction medium, the process inherently avoids the toxicity and flammability issues associated with organic solvents, thereby drastically simplifying the safety profile of the manufacturing plant. The use of a heterogeneous Cu-Zn-Pd/Al2O3 catalyst allows for easy separation via simple filtration, enabling the catalyst to be recovered and reused, which is a critical factor for cost reduction in pharmaceutical intermediates manufacturing. Moreover, the reaction conditions, operating between 150-250°C and 2-8 MPa, are well within the capabilities of standard high-pressure industrial reactors, ensuring that the transition from lab scale to commercial production is seamless. This method not only achieves conversion rates of up to 100% but also delivers selectivity as high as 98%, proving that efficiency and sustainability can coexist in modern chemical synthesis.
Mechanistic Insights into Cu-Zn-Pd/Al2O3 Catalyzed Cyclization
The exceptional performance of this synthesis route is fundamentally rooted in the synergistic catalytic effects provided by the ternary metal system supported on alumina. The Cu-Zn-Pd/Al2O3 catalyst functions through a sophisticated mechanism where the palladium component likely facilitates the initial hydrogenation or activation of the nitro group, while the copper and zinc species promote the subsequent cyclization and dehydration steps required to form the imidazole ring. The specific loading ratios, optimized between 0.5-15wt% for Copper, 0.1-3wt% for Zinc, and 0.5-5wt% for Palladium, create active sites that are highly selective for the desired benzimidazole structure, minimizing the formation of over-reduced byproducts or polymerized tars. This precise tuning of the catalyst surface chemistry ensures that the reaction proceeds through a concerted pathway, where the intermediate species are rapidly converted to the final product before they can degrade. The stability of the catalyst under high-temperature and high-pressure aqueous conditions suggests a strong metal-support interaction that prevents leaching of the precious metals, a common failure mode in heterogeneous catalysis. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate these results, as slight deviations in catalyst preparation, such as the calcination temperature of 240-500°C, could alter the active phase distribution.
Furthermore, the choice of water as a solvent plays a non-trivial role in the reaction mechanism, potentially acting as a proton shuttle that facilitates the dehydration steps essential for ring closure. The high pressure of 2-8 MPa maintains the water in a liquid or supercritical state at elevated temperatures, enhancing its solvating power for organic substrates and improving mass transfer rates within the reactor. This environment suppresses side reactions that might occur in dry organic media, such as the formation of Schiff bases that do not cyclize efficiently. The result is a remarkably clean impurity profile, where the primary challenge shifts from removing complex byproducts to simply separating the product from the aqueous phase. For quality control laboratories, this translates to simpler analytical methods and higher confidence in the purity of the high-purity OLED material or pharmaceutical intermediate produced. The ability to achieve such high selectivity without the use of stoichiometric oxidants or reductants underscores the elegance of this catalytic design.
How to Synthesize Benzimidazole Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst and the control of reaction parameters to ensure reproducibility and safety. The process begins with the meticulous impregnation of the alumina support with precise concentrations of palladium, zinc, and copper salts, followed by pH adjustment and thermal treatment to activate the metal sites. Once the catalyst is prepared, the reaction is conducted in a high-pressure autoclave where the molar ratios of o-nitroaniline to fatty alcohol and water are strictly maintained to maximize conversion. Detailed standardized synthesis steps, including specific heating ramps and stirring speeds, are critical for scaling this technology from gram-scale experiments to ton-level production. The following guide outlines the essential procedural framework derived from the patent data to assist technical teams in adopting this methodology.
- Prepare the Cu-Zn-Pd/Al2O3 catalyst by impregnating alumina with palladium, zinc, and copper solutions, followed by drying, calcination at 240-500°C, and hydrogen reduction.
- Load the o-nitroaniline compound, fatty alcohol, water, and the prepared catalyst into a high-pressure autoclave under an inert nitrogen or argon atmosphere.
- Heat the mixture to 150-250°C at a pressure of 2-8 MPa for 6-24 hours, then filter to recover the catalyst and distill the filtrate to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic technology offers profound benefits that extend far beyond the laboratory, directly impacting the bottom line and supply chain resilience. By shifting the raw material base from expensive o-phenylenediamine to more commodity-grade o-nitroanilines and fatty alcohols, manufacturers can achieve significant cost reduction in pharmaceutical intermediates manufacturing without compromising on quality. The elimination of liquid acids and organic solvents removes the need for costly waste disposal services and reduces the regulatory burden associated with handling hazardous chemicals, further driving down operational expenditures. Additionally, the heterogeneous nature of the catalyst means that precious metals are not lost in the product stream, allowing for their recovery and reuse, which stabilizes the cost of goods sold against fluctuations in metal prices. These factors combine to create a robust economic model that supports competitive pricing strategies in the global market.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the substitution of high-cost starting materials with readily available nitro-compounds and alcohols, which are generally produced at much larger scales and lower prices. By avoiding the use of stoichiometric amounts of strong acids or expensive oxidizing agents, the process significantly reduces the consumption of auxiliary chemicals that contribute heavily to the variable costs of production. The ability to reuse the solid catalyst multiple times without regeneration further amortizes the initial investment in precious metals like palladium, leading to substantial long-term savings. Moreover, the simplified work-up procedure, which involves mere filtration and distillation, reduces energy consumption and labor hours compared to complex extraction and neutralization sequences found in traditional methods.
- Enhanced Supply Chain Reliability: Utilizing water as a solvent and stable solid catalysts mitigates many of the supply chain risks associated with volatile organic solvents and corrosive liquids that often face transportation restrictions. The raw materials, o-nitroanilines and fatty alcohols, are widely available from multiple global suppliers, reducing the risk of single-source dependency and ensuring continuous production even during market disruptions. The robustness of the catalyst under harsh reaction conditions implies a longer operational lifespan, reducing the frequency of catalyst replenishment orders and minimizing downtime for reactor cleaning or changeovers. This reliability is crucial for maintaining consistent delivery schedules to downstream customers who depend on just-in-time inventory models for their own manufacturing lines.
- Scalability and Environmental Compliance: The process is inherently scalable because it operates in a homogeneous liquid phase (water) with a suspended solid catalyst, which behaves predictably when moving from pilot plants to full-scale commercial reactors. The absence of toxic organic solvents and strong acids simplifies the environmental permitting process and reduces the capital expenditure required for waste treatment infrastructure, making it easier to establish new production facilities in regions with strict environmental regulations. The high atom economy of the reaction, coupled with the potential for recycling the aqueous phase, aligns with corporate sustainability targets and enhances the brand reputation of the manufacturer as a responsible chemical producer. This environmental advantage is increasingly becoming a key differentiator when qualifying as a supplier for major multinational corporations with rigorous green procurement policies.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this benzimidazole synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide clarity for engineering and procurement teams. Understanding these details is essential for assessing the feasibility of integrating this process into existing manufacturing workflows.
Q: Can the Cu-Zn-Pd/Al2O3 catalyst be reused in benzimidazole synthesis?
A: Yes, the patent data indicates that the supported metal solid catalyst maintains high activity and stability, allowing it to be separated by filtration and reused multiple times without significant loss in conversion or selectivity.
Q: What are the advantages of using water as a solvent in this reaction?
A: Using water eliminates the need for volatile organic solvents and strong liquid acids, significantly reducing environmental impact, simplifying waste treatment, and lowering overall production costs compared to traditional organic solvent-based methods.
Q: How does this method compare to traditional o-phenylenediamine routes?
A: This method utilizes cheaper o-nitroaniline and fatty alcohol raw materials instead of expensive o-phenylenediamine and aldehydes, while achieving higher yields (up to 98%) and avoiding harsh acidic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN103288743A for producing high-value benzimidazole derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to market. Our facility is equipped with state-of-the-art high-pressure reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required for pharmaceutical and agrochemical applications. We are committed to leveraging such innovative green chemistry routes to deliver superior products that balance performance with sustainability.
We invite you to collaborate with us to explore how this efficient synthesis method can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals.
