Advancing Pharmaceutical Intermediates: Efficient Synthesis of 2-Trifluoromethyl Benzimidazoles via Recyclable Heterogeneous Catalysis
Advancing Pharmaceutical Intermediates: Efficient Synthesis of 2-Trifluoromethyl Benzimidazoles via Recyclable Heterogeneous Catalysis
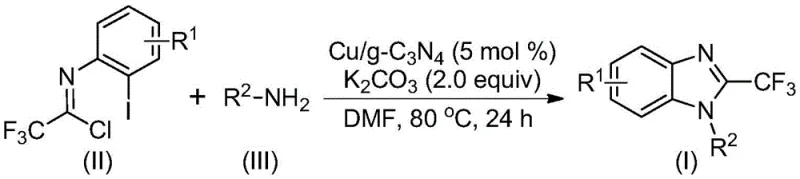
The landscape of pharmaceutical intermediate synthesis is constantly evolving, driven by the need for greener, more efficient, and cost-effective methodologies. A significant breakthrough in this domain is detailed in patent CN113429349A, which discloses a robust preparation method for 2-trifluoromethyl substituted benzimidazole compounds. This class of heterocycles is pivotal in medicinal chemistry, often serving as key scaffolds in bioactive molecules due to the metabolic stability and lipophilicity imparted by the trifluoromethyl group. The patented technology leverages a heterogeneous copper-doped carbon nitride (Cu/g-C3N4) catalyst to facilitate a cascade cyclization reaction between trifluoroacetimidoyl chloride and various amines. This approach represents a paradigm shift from traditional homogeneous catalysis, offering distinct advantages in terms of catalyst recovery, operational simplicity, and environmental compliance. For R&D directors and procurement strategists, understanding the nuances of this technology is crucial for optimizing supply chains and reducing the cost of goods sold (COGS) in complex API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzimidazole core, particularly with trifluoromethyl substitution, has relied on several conventional pathways that present significant logistical and chemical challenges. Traditional methods often involve the condensation of o-phenylenediamine with trifluoroacetic acid or its derivatives, which frequently requires harsh reaction conditions and strong dehydrating agents. Alternatively, reductive cyclization of o-nitroanilines or direct trifluoromethylation of pre-formed benzimidazoles has been employed. However, these routes are often plagued by narrow substrate scopes, poor atom economy, and the necessity for expensive or difficult-to-handle reagents. Furthermore, earlier reports utilizing copper catalysis typically relied on homogeneous systems. While effective in small-scale laboratory settings, homogeneous catalysts pose severe difficulties in industrial applications, primarily due to the challenge of removing trace metal residues from the final product—a critical quality attribute for pharmaceutical intermediates. The inability to easily recover and reuse the catalyst also contributes to higher operational costs and increased heavy metal waste.
The Novel Approach
The methodology described in patent CN113429349A offers a compelling solution to these entrenched problems by utilizing a heterogeneous Cu/g-C3N4 catalyst. This novel approach enables a tandem nucleophilic addition-elimination and cyclization sequence under remarkably mild conditions, typically ranging from 70°C to 90°C in polar aprotic solvents like DMF. The use of readily available starting materials, specifically trifluoroacetimidoyl chloride and diverse amines, coupled with inexpensive inorganic bases like potassium carbonate, streamlines the raw material sourcing process. Crucially, the heterogeneous nature of the catalyst allows for simple filtration post-reaction, facilitating catalyst recycling. Experimental data within the patent indicates that the catalyst retains significant activity even after three recycling runs, demonstrating robustness that is essential for continuous processing. This transition from homogeneous to heterogeneous catalysis not only simplifies the workup procedure but also aligns with green chemistry principles by minimizing waste generation and energy consumption.
Mechanistic Insights into Cu/g-C3N4 Catalyzed Cyclization
Understanding the mechanistic underpinnings of this transformation is vital for R&D teams aiming to adapt this chemistry for specific proprietary molecules. The reaction initiates with a nucleophilic attack of the amine on the imidoyl chloride, resulting in the formation of an amidine intermediate through an addition-elimination pathway. Subsequently, the copper species embedded within the graphitic carbon nitride matrix plays a dual role. It facilitates the oxidative insertion into the carbon-iodine bond of the aryl ring (in substrates where R1 allows or via intramolecular activation), forming a transient organocopper complex. Under the basic conditions provided by potassium carbonate, a molecule of hydrogen iodide is eliminated, promoting the formation of a bivalent or trivalent cyclic copper intermediate. The final step involves a reductive elimination that closes the benzimidazole ring and releases the 2-trifluoromethyl substituted product, regenerating the active copper catalytic site. This intricate dance of electron transfer and bond formation is stabilized by the unique electronic environment of the g-C3N4 support, which prevents the aggregation of copper nanoparticles and maintains high catalytic turnover.
From an impurity control perspective, this mechanism offers distinct advantages. The mild reaction temperatures (70-90°C) minimize thermal degradation of sensitive functional groups, thereby reducing the formation of complex polymeric byproducts often seen in high-temperature condensations. Furthermore, the specificity of the copper-catalyzed C-N bond formation ensures high regioselectivity, limiting the generation of isomeric impurities that are notoriously difficult to separate. The use of a solid-supported catalyst also reduces the likelihood of metal-mediated side reactions that can occur with soluble copper salts. For quality assurance teams, this translates to a cleaner crude reaction profile, which simplifies downstream purification and enhances the overall yield of high-purity material. The ability to tune the electronic properties of the catalyst support further allows for optimization against specific substrate liabilities, ensuring consistent quality across different batches.
How to Synthesize 2-Trifluoromethyl Benzimidazole Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize efficiency and yield. The standard protocol involves charging a reaction vessel, such as a Schlenk tube, with the heterogeneous catalyst (Cu/g-C3N4), the base (K2CO3), the trifluoroacetimidoyl chloride substrate, and the chosen amine. The molar ratio is critical; typically, a slight excess of the amine (1.5 equivalents) relative to the imidoyl chloride is preferred to drive the reaction to completion. The reaction is conducted in an organic solvent, with DMF showing superior performance in dissolving both organic substrates and inorganic salts while maintaining catalyst stability. Heating the mixture to 80°C for approximately 24 hours generally provides optimal conversion. Following the reaction, the solid catalyst is removed by filtration, and the product is isolated via standard chromatographic techniques. For a detailed, step-by-step operational guide tailored to your specific facility requirements, please refer to the standardized protocol below.
- Prepare the reaction mixture by adding copper-doped carbon nitride (Cu/g-C3N4), potassium carbonate, trifluoroacetimidoyl chloride, and the corresponding amine into an organic solvent such as DMF.
- Heat the reaction mixture to a temperature range of 70-90°C and maintain stirring for a duration of 18 to 30 hours to ensure complete conversion.
- Upon completion, filter the reaction mixture to recover the heterogeneous catalyst, mix the filtrate with silica gel, and purify via column chromatography to isolate the target benzimidazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this heterogeneous catalytic process offers tangible strategic benefits that extend beyond mere chemical yield. The shift towards a recyclable catalyst system directly addresses the escalating costs associated with precious metal usage and waste disposal in fine chemical manufacturing. By eliminating the need for expensive ligand systems and complex metal scavenging steps required for homogeneous catalysts, the overall process mass intensity (PMI) is significantly improved. This reduction in processing steps translates to shorter cycle times and lower utility consumption, providing a competitive edge in cost reduction in pharmaceutical intermediate manufacturing. Moreover, the reliance on commodity chemicals like potassium carbonate and widely available amines mitigates supply chain risks associated with specialized reagents, ensuring greater continuity of supply.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of costly downstream purification steps. In traditional homogeneous catalysis, removing trace copper to meet ppm-level specifications often requires specialized resins or multiple recrystallizations, which erodes yield and increases solvent usage. The heterogeneous nature of Cu/g-C3N4 allows for physical separation via filtration, drastically simplifying the workup. Additionally, the catalyst's recyclability means that the effective cost of the catalyst per kilogram of product decreases with each reuse cycle. This structural change in the process flow leads to substantial cost savings without compromising on the quality or purity of the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, non-sensitive reagents. Unlike air- or moisture-sensitive catalysts that require inert atmosphere handling and specialized storage, the Cu/g-C3N4 catalyst and the associated reagents are stable and easy to handle. This reduces the logistical burden and the risk of batch failures due to reagent degradation. Furthermore, the broad substrate tolerance of the reaction means that a single catalytic platform can be used to synthesize a wide library of analogues. This flexibility allows manufacturers to respond rapidly to changing market demands or clinical trial requirements without needing to requalify entirely new synthetic routes, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often exposes hidden inefficiencies, but this methodology is inherently scalable. The use of common solvents like DMF and moderate temperatures fits well within existing reactor infrastructure, avoiding the need for cryogenic cooling or high-pressure equipment. From an environmental standpoint, the reduction in heavy metal waste aligns with increasingly stringent global regulations on effluent discharge. The ability to recycle the catalyst minimizes the generation of hazardous solid waste, supporting corporate sustainability goals. This ease of commercial scale-up of complex heterocyclic compounds ensures that production can be ramped up quickly to meet commercial launch timelines while maintaining strict environmental compliance standards.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries regarding the reaction scope, catalyst handling, and product quality. These insights are derived directly from the experimental data and technical disclosures found in the patent literature. Understanding these nuances is essential for making informed decisions about process adoption and technology transfer. We encourage potential partners to review these details closely to assess the alignment with their current manufacturing capabilities.
Q: What are the advantages of using Cu/g-C3N4 over traditional homogeneous copper catalysts?
A: The primary advantage lies in the heterogeneity of the catalyst, which allows for easy separation via simple filtration. Unlike homogeneous catalysts that require complex removal processes and leave metal residues, Cu/g-C3N4 can be recycled multiple times with only partial loss of efficiency, significantly reducing waste and purification costs.
Q: What is the substrate scope for this trifluoromethylation cyclization reaction?
A: The method demonstrates broad substrate tolerance. It accommodates various substituents on the aromatic ring (R1) including hydrogen, methyl, chloro, bromo, and trifluoromethyl groups. Furthermore, it accepts diverse amine inputs (R2) ranging from aliphatic chains like n-butyl and cyclohexyl to aromatic groups like phenyl and benzyl.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its mild reaction conditions (70-90°C) and the use of inexpensive, commercially available reagents like potassium carbonate and DMF. The ability to recycle the catalyst and the simple post-treatment workflow make it economically viable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN113429349A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs equipped with state-of-the-art analytical instrumentation, guaranteeing that every batch of 2-trifluoromethyl benzimidazole meets the exacting standards required by global regulatory bodies. We understand that consistency and reliability are paramount in the pharmaceutical supply chain, and our infrastructure is designed to deliver exactly that.
We invite you to collaborate with us to leverage this efficient synthetic route for your next project. Whether you are looking to optimize an existing process or develop a new candidate from scratch, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume and purity requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a wealth of chemical expertise and manufacturing capacity dedicated to accelerating your drug development timeline while maximizing cost efficiency.
