Revolutionizing Furan Production: A Solvent-Free Ti-Catalyzed Route for Commercial Scale-Up
Revolutionizing Furan Production: A Solvent-Free Ti-Catalyzed Route for Commercial Scale-Up
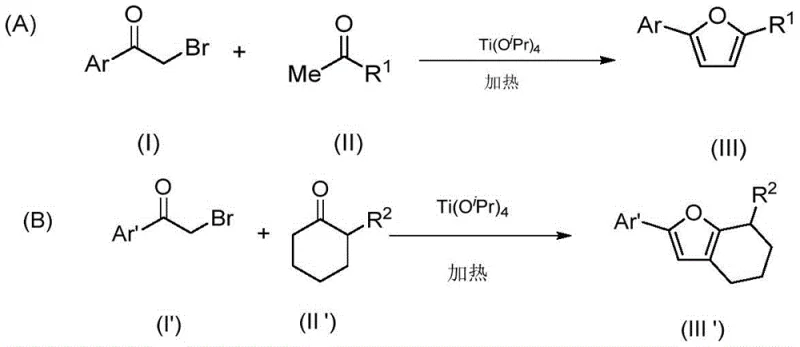
The landscape of heterocyclic chemistry is constantly evolving, driven by the urgent need for greener, more efficient, and cost-effective synthetic methodologies. A groundbreaking advancement in this field is detailed in patent CN112939903A, which discloses a novel method for preparing polysubstituted furan compounds directly from aryl ketone bromides and methyl ketones or cyclic ketones. This technology leverages the catalytic power of tetraisopropyl titanate under strictly solvent-free conditions, representing a significant departure from traditional multi-step syntheses that often rely on hazardous organic solvents and harsh reagents. For R&D directors and process chemists, this patent offers a compelling solution to the longstanding challenges of furan synthesis, providing a pathway that combines high atom economy with operational simplicity. The ability to generate complex furan scaffolds in a single pot without the burden of solvent recovery not only streamlines the workflow but also aligns perfectly with modern green chemistry principles, making it an attractive candidate for immediate adoption in the manufacturing of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
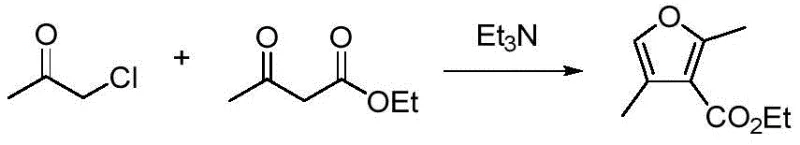
Historically, the synthesis of furan derivatives has been dominated by strategies that, while effective, suffer from significant logistical and economic drawbacks when applied to industrial settings. The classical Feist-Bénary furan synthesis, for instance, relies on the condensation of alpha-halogenated ketones with beta-keto esters under alkaline conditions. While this one-step approach is conceptually simple, it is severely restricted by the requirement for readily enolizable beta-dicarbonyl compounds, which limits the structural diversity of the resulting products primarily to 3-furoates. Furthermore, alternative multi-step strategies involving the conversion of common ketones into stable enol derivatives followed by coupling and Paal-Knorr cyclization introduce unnecessary complexity. These multi-step routes often necessitate strong acids, high temperatures, and prolonged reaction times for the final dehydration step, leading to the formation of difficult-to-separate by-products and a substantially reduced overall yield. Such inefficiencies translate directly into higher production costs and increased waste generation, creating bottlenecks for supply chain managers seeking reliable sources of high-purity intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN112939903A introduces a streamlined, one-pot protocol that bypasses the need for pre-functionalized enol derivatives or specialized beta-keto esters. By utilizing tetraisopropyl titanate as a promoter, the reaction facilitates the direct coupling of aryl ketone bromides with simple methyl ketones or cyclic ketones. This approach eliminates the need for any organic solvent, thereby removing the costs and safety hazards associated with solvent storage, handling, and disposal. The reaction proceeds under mild heating conditions, typically around 80°C, and demonstrates exceptional functional group tolerance, accommodating substrates with chloro, methoxy, and even bulky naphthyl substituents without compromising yield. This robustness ensures that the process can be adapted to synthesize a wide array of furan derivatives, from simple 2,5-diphenylfurans to complex fused bicyclic systems, providing a versatile platform for the rapid generation of chemical libraries.
Mechanistic Insights into Titanium-Catalyzed Cyclization
The efficacy of this synthetic route lies in the unique Lewis acidic properties of tetraisopropyl titanate, which plays a dual role in activating both reaction partners and driving the cyclization forward. Mechanistically, the titanium center coordinates with the carbonyl oxygen of the methyl ketone, enhancing its acidity and promoting the formation of the reactive enol or enolate species in situ. Simultaneously, the catalyst activates the carbonyl group of the alpha-bromo ketone, rendering it more susceptible to nucleophilic attack. This concerted activation lowers the energy barrier for the initial carbon-carbon bond formation, leading to a 1,4-dicarbonyl intermediate which then undergoes rapid intramolecular cyclization. The final dehydration step, crucial for aromatization to the furan ring, is also facilitated by the titanium species, which acts as a water scavenger, effectively pushing the equilibrium towards the product. This elegant mechanistic pathway avoids the formation of stable intermediates that require isolation, thus minimizing material loss and maximizing throughput.
From an impurity control perspective, the mild nature of this catalytic system is paramount. Traditional acid-catalyzed dehydrations often lead to polymerization or decomposition of sensitive functional groups, resulting in complex impurity profiles that are costly to purge. However, the neutral to mildly Lewis acidic environment provided by tetraisopropyl titanate preserves the integrity of sensitive moieties such as halogens and ethers. The absence of strong protic acids prevents side reactions like hydrolysis or rearrangement, ensuring that the crude reaction mixture is relatively clean. This high level of chemoselectivity simplifies the downstream purification process, often allowing for straightforward extraction and crystallization rather than demanding chromatographic separations. For quality control teams, this translates to a more consistent impurity profile and a higher probability of meeting stringent pharmacopeial specifications for API intermediates without extensive reprocessing.
How to Synthesize Polysubstituted Furans Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry and addition sequence to maximize the catalytic efficiency of the titanium species. The patent specifies a molar ratio where the ketone and alpha-aryl bromide ketone are mixed in a roughly 1:1 to 1:1.4 ratio, with a slight excess of the titanium catalyst (1 to 2 equivalents) to ensure complete conversion. The process begins by heating the mixture of ketones under an inert atmosphere to prevent oxidation, followed by the addition of the catalyst once the thermal equilibrium is reached. This specific order of operations is critical for maintaining the activity of the titanate and preventing premature hydrolysis. Detailed standardized synthetic steps, including precise quenching protocols and workup procedures to isolate the high-purity furan product, are provided in the technical guide below.
- Mix aryl ketone bromide and methyl ketone (or cyclic ketone) in a reactor under inert gas protection and heat to 60-100°C.
- Add tetraisopropyl titanate catalyst to the heated mixture and maintain stirring for 4 to 24 hours to complete the cyclization.
- Quench the reaction with saturated ammonium chloride, extract with dichloromethane, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this solvent-free methodology offers tangible benefits that extend far beyond the laboratory bench. The most immediate impact is seen in the reduction of raw material costs and waste disposal fees. By eliminating the need for large volumes of organic solvents such as DMF, THF, or toluene, manufacturers can significantly reduce their expenditure on consumables and the associated logistics of solvent recovery or incination. Furthermore, the use of tetraisopropyl titanate, a commercially abundant and inexpensive reagent, replaces more exotic or toxic transition metal catalysts that might require expensive removal steps to meet residual metal limits in pharmaceutical products. This shift not only lowers the direct cost of goods sold but also mitigates regulatory risks associated with heavy metal contamination, ensuring a smoother path through quality assurance audits.
- Cost Reduction in Manufacturing: The economic argument for adopting this technology is robust, driven primarily by the intensification of the reaction process. Operating under solvent-free conditions inherently increases the concentration of reactants, which allows for the use of smaller reactors to produce the same amount of product, effectively increasing the volumetric productivity of existing manufacturing assets. Additionally, the simplified workup procedure, which involves a basic aqueous quench and extraction, reduces the labor hours and utility consumption required for purification. The elimination of solvent distillation steps further contributes to energy savings, making the overall process energetically favorable compared to traditional dilute solution chemistry. These cumulative efficiencies result in a leaner manufacturing process that is less sensitive to fluctuations in solvent prices and availability.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on commodity chemicals as starting materials. Aryl ketone bromides and methyl ketones are widely produced bulk chemicals with stable global supply networks, reducing the risk of shortages that often plague specialized reagents. The robustness of the reaction conditions, which tolerate a wide range of temperatures and minor variations in stoichiometry, ensures consistent batch-to-batch performance even when scaling up from pilot to commercial production. This reliability allows for more accurate demand forecasting and inventory planning, as the lead time for production is shortened by the removal of time-consuming solvent exchange and drying steps. Consequently, manufacturers can respond more agilely to market demands for key furan intermediates used in antiviral, anticancer, and anti-inflammatory drug formulations.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this solvent-free approach inherently mitigates many of these risks. The absence of volatile organic compounds (VOCs) reduces the fire hazard and the load on facility ventilation systems, simplifying the safety case for large-scale reactors. From an environmental compliance standpoint, the drastic reduction in waste solvent generation aligns with increasingly stringent global regulations on industrial emissions and waste disposal. The process generates minimal aqueous waste which is easy to treat, and the titanium byproducts are generally non-toxic and easily managed. This green profile not only enhances the corporate sustainability image but also future-proofs the manufacturing site against tightening environmental legislation, ensuring long-term operational continuity without the need for costly retrofits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this titanium-catalyzed furan synthesis. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic overview of the technology's capabilities and limitations for potential adopters. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the primary advantages of this solvent-free furan synthesis method?
A: The primary advantages include the elimination of organic solvents which reduces environmental pollution and cost, mild reaction conditions (80°C), excellent substrate universality allowing for diverse functional groups, and a simplified one-step operation suitable for industrial scaling.
Q: How does this method compare to the classical Feist-Bénary synthesis?
A: Unlike the Feist-Bénary synthesis which is limited to beta-keto esters and produces 3-furoates, this novel method utilizes readily available methyl ketones and aryl ketone bromides to produce a wider variety of polysubstituted furans with higher flexibility in substrate selection.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly suitable for large-scale production due to its solvent-free nature which simplifies safety and waste management, the use of inexpensive and stable titanium catalysts, and the robust tolerance to various functional groups ensuring high yields across different batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Furan Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the solvent-free titanium-catalyzed synthesis described in CN112939903A for the production of high-value furan intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle solvent-free reactions safely and efficiently, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry. We are committed to delivering not just a chemical product, but a reliable supply solution that enhances your own manufacturing efficiency.
We invite you to explore how this advanced synthetic route can optimize your supply chain and reduce your overall production costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data for our furan derivatives and to discuss route feasibility assessments for your next project. Let us collaborate to bring your next-generation therapeutics to market faster and more sustainably.
