Advanced Photoredox and Copper Co-Catalysis for Scalable Polysubstituted Furan Manufacturing
Advanced Photoredox and Copper Co-Catalysis for Scalable Polysubstituted Furan Manufacturing
The landscape of heterocyclic chemistry is continuously evolving, driven by the demand for more efficient and sustainable synthetic routes for bioactive scaffolds. Patent CN112341417A introduces a groundbreaking methodology for the synthesis of polysubstituted furans, a structural motif ubiquitous in natural products and pharmaceutical agents. This innovation leverages a synergistic dual-catalytic system combining visible-light photoredox catalysis with copper co-catalysis to overcome longstanding limitations in substrate scope and reaction conditions. By utilizing carbon-centered radicals as nucleophiles, this technology enables the construction of complex furan rings under exceptionally mild conditions, specifically at room temperature using blue LED irradiation. For R&D directors and process chemists, this represents a significant leap forward in accessing diverse chemical space without the need for harsh thermal activation or expensive noble metal catalysts.
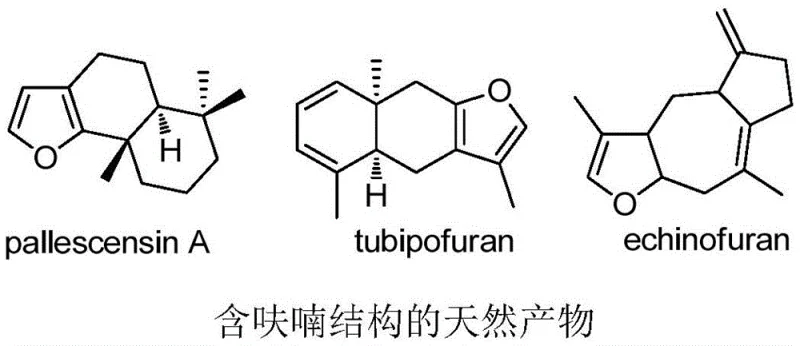
Furan rings serve as a cornerstone in medicinal chemistry, appearing in countless bioactive natural products such as pallescensin A, tubipofuran, and echinofuran, which exhibit potent antiviral, antibacterial, and antitumor activities. The inherent biological significance of these five-membered heterocycles drives continuous interest from the organic synthesis community. However, traditional synthetic strategies often struggle with functional group tolerance and the requirement for specialized, electron-rich nucleophiles. The disclosed invention addresses these challenges by expanding the repertoire of usable nucleophiles to include simple alkyl radicals generated in situ. This capability is crucial for the development of next-generation pharmaceutical intermediates, where structural diversity is key to optimizing drug efficacy and metabolic stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted furans has relied heavily on transition metal-catalyzed alkyne cycloisomerization, with gold catalysis being a prominent example since the seminal work by Larock et al. in 2004. While effective, these conventional gold-catalyzed systems typically necessitate the use of expensive precious metals and often require elevated temperatures or specific activators to proceed efficiently. Furthermore, a critical bottleneck in these traditional methodologies is their limited scope regarding carbon-centered nucleophiles; they are predominantly restricted to electron-rich aromatic compounds and indoles. This restriction severely hampers the ability to introduce aliphatic chains or diverse alkyl groups directly onto the furan ring, limiting the structural variety accessible to medicinal chemists. Additionally, the reliance on thermal conditions can lead to decomposition of sensitive functional groups, complicating the synthesis of complex drug candidates.
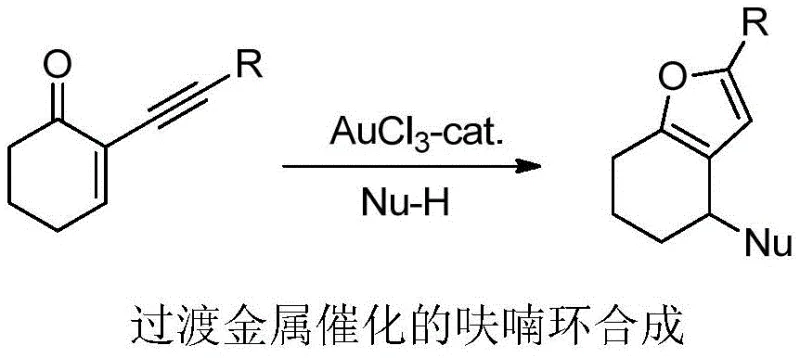
The Novel Approach
In stark contrast to these legacy methods, the patented light/copper co-catalyzed strategy offers a robust and versatile alternative that operates under ambient conditions. By employing a photocatalyst such as Ir[dF(CF3)ppy]2(dtbbpy)PF6 in conjunction with a copper salt like Cu(OTf)2, the system activates 2-alkynyl-2-enones towards radical addition. This approach successfully bypasses the electronic constraints of previous methods, allowing for the use of alkyl bis(catechol)silicates and Hantzsch esters as radical precursors. The result is a highly efficient cyclization that proceeds at room temperature over 24 hours, eliminating the energy costs associated with heating and reducing the risk of thermal degradation. This shift not only broadens the substrate scope significantly but also aligns with green chemistry principles by utilizing visible light as a traceless reagent.
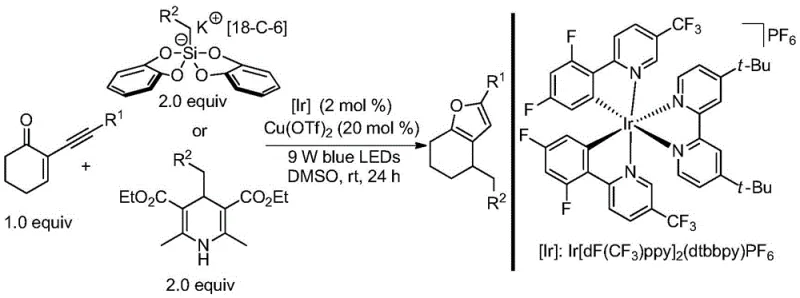
Mechanistic Insights into Photoredox and Copper Co-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the seamless integration of two distinct catalytic cycles. The photoredox cycle initiates the process by absorbing blue light photons to reach an excited state, which then engages in single-electron transfer (SET) with the radical precursor. This oxidation event generates a reactive carbon-centered radical while regenerating the ground-state photocatalyst. Simultaneously, the copper co-catalyst coordinates with the 2-alkynyl-2-enone substrate, activating the alkyne moiety towards nucleophilic attack. The in situ generated alkyl radical adds to the activated alkyne, triggering a cascade of intramolecular cyclization and subsequent protonation or elimination steps to forge the furan ring. This dual-catalytic manifold ensures high turnover numbers and exceptional selectivity, minimizing the formation of unwanted byproducts that often plague radical reactions.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining product purity. Traditional thermal methods often promote side reactions such as polymerization of the enone or oligomerization of the radical species due to high energy inputs. By contrast, the room-temperature operation of this photochemical process kinetically favors the desired cyclization pathway over competing decomposition routes. Furthermore, the use of specific ligand environments around the copper center helps to direct the regioselectivity of the radical addition, ensuring that the polysubstituted furan is formed with high fidelity. This level of control is essential for high-purity pharmaceutical intermediates, where stringent regulatory standards demand minimal levels of genotoxic impurities and closely related structural analogs.
How to Synthesize Polysubstituted Furans Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the stoichiometry of the catalytic components and the quality of the solvent system. The standard protocol involves charging a reaction vessel with the photosensitizer (2 mol%), the 2-alkynyl-2-enone substrate (1.0 equiv), the radical precursor (2.0 equiv), and the copper salt (20 mol%). The reaction is conducted in dry dimethyl sulfoxide (DMSO) under an inert nitrogen atmosphere to prevent quenching of the radical species by oxygen. Following a 24-hour irradiation period with 9W blue LEDs, the crude mixture undergoes a straightforward aqueous workup involving sodium carbonate and ethyl acetate extraction. For detailed operational parameters and specific purification techniques, please refer to the standardized synthesis guide below.
- Combine the photosensitizer Ir[dF(CF3)ppy]2(dtbbpy)PF6, 2-alkynyl-2-enone substrate, radical precursor (such as alkyl bis(catechol)silicate), and copper trifluoromethanesulfonate in a reaction vessel.
- Add dry dimethyl sulfoxide (DMSO) as the solvent under a nitrogen atmosphere and seal the reaction tube.
- Irradiate the mixture with 9W blue LEDs at room temperature for 24 hours, followed by aqueous workup and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photo/copper co-catalyzed technology presents compelling economic and logistical benefits. The most immediate advantage is the drastic reduction in raw material costs achieved by replacing expensive gold catalysts with abundant and inexpensive copper salts. This substitution does not compromise reaction efficiency; rather, it enhances the overall process economics by lowering the cost of goods sold (COGS) for the final furan intermediates. Moreover, the elimination of high-temperature heating requirements translates directly into reduced energy consumption and lower utility overheads, contributing to a more sustainable and cost-effective manufacturing footprint. These factors collectively strengthen the supply chain resilience by making the production of these critical building blocks less susceptible to fluctuations in precious metal markets.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with earth-abundant copper significantly lowers the direct material costs associated with the synthesis. Since the copper salt is used in catalytic amounts and is far cheaper than gold or iridium alternatives often found in similar transformations, the overall catalyst cost per kilogram of product is minimized. Additionally, the ambient temperature operation removes the need for specialized heating equipment and the associated energy expenditure, leading to substantial operational savings. This economic efficiency allows for more competitive pricing strategies when sourcing these complex fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as 2-alkynyl-2-enones and simple silicate radical precursors ensures a stable and continuous supply of inputs. Unlike specialized reagents that may have long lead times or single-source dependencies, the components of this reaction are commoditized and easily sourced from multiple global suppliers. The robustness of the reaction conditions also means that production is less likely to be interrupted by equipment failures related to extreme thermal stress, thereby guaranteeing consistent delivery schedules for downstream customers requiring reliable pharmaceutical intermediate suppliers.
- Scalability and Environmental Compliance: Scaling this photochemical process is facilitated by the use of flow chemistry technologies or scalable LED arrays, which provide uniform light penetration even in larger volumes. The use of DMSO, a polar aprotic solvent with a high boiling point, simplifies containment and reduces volatile organic compound (VOC) emissions compared to low-boiling etheral solvents. Furthermore, the mild reaction profile minimizes the generation of hazardous waste streams, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. This ease of scale-up supports the commercial scale-up of complex pharmaceutical intermediates from gram-scale discovery to multi-ton production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on substrate compatibility, reaction optimization, and potential industrial applications. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this photo/copper co-catalyzed method over traditional gold catalysis?
A: Unlike traditional gold-catalyzed methods that often require expensive metals and are limited to electron-rich nucleophiles, this novel approach utilizes inexpensive copper salts and visible light. It significantly expands substrate scope by enabling the use of carbon-centered radicals as nucleophiles, allowing for the synthesis of diverse polysubstituted furans under mild, room-temperature conditions.
Q: What specific radical precursors are compatible with this synthesis protocol?
A: The patent demonstrates high compatibility with alkyl bis(catechol)silicate-18-crown-6-potassium complexes and 4-alkyl substituted Hantzsch esters. These precursors effectively generate the necessary carbon-centered radicals under the photoredox conditions, facilitating the cyclization with 2-alkynyl-2-enones to form the target furan structures.
Q: Is this process suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the process operates at room temperature using standard blue LED arrays and common solvents like DMSO, which simplifies thermal management and safety protocols compared to high-temperature thermal cyclizations. The use of earth-abundant copper co-catalysts instead of precious metals like gold further enhances its economic viability for scaling up complex pharmaceutical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Furans Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photochemical methodologies in modern drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative academic discoveries like this light/copper co-catalyzed furan synthesis are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with specialized photoreactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-quality polysubstituted furans that empower your R&D teams to accelerate their pipeline programs with confidence.
We invite you to engage with our technical procurement team to discuss how this novel synthetic route can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your project needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted provider of cutting-edge chemical solutions tailored to your unique requirements.
