Scalable Metal-Free Synthesis of Polysubstituted Furans for Advanced Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is constantly evolving, driven by the urgent need for more sustainable and cost-effective synthetic routes, particularly for high-value scaffolds like polysubstituted furans. Patent CN112341409B introduces a groundbreaking preparation method that addresses long-standing challenges in the synthesis of 2,3-disubstituted and 2,3,5-trisubstituted furan compounds. This technology leverages a transition-metal-free strategy, utilizing carbonyl compounds bearing alpha-position electron-withdrawing groups and dichloroolefins as key starting materials. By operating under alkaline conditions, this process achieves high efficiency and broad substrate applicability without the reliance on precious metal catalysts. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal shift towards greener, more economical manufacturing protocols that align with modern regulatory standards for pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of polysubstituted furan cores has relied heavily on methodologies that involve transition metal catalysis, such as silver-catalyzed reactions between phenylacetylenes and 1,3-dicarbonyl compounds. While effective in academic settings, these conventional approaches present significant hurdles for industrial scale-up. The primary limitation lies in the inherent cost and complexity associated with transition metals; catalysts like silver or palladium are not only expensive but also introduce severe purification challenges. Removing trace metal residues to meet the stringent ppm-level specifications required by the pharmaceutical industry often necessitates additional downstream processing steps, such as specialized scavenging or repeated chromatography. Furthermore, many traditional methods suffer from limited regioselectivity and narrow substrate scope, restricting their utility when diverse functional groups are required on the furan ring. These factors collectively inflate the cost of goods sold (COGS) and extend lead times, making conventional routes less attractive for large-scale commercial production of complex organic intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN112341409B offers a streamlined, metal-free alternative that fundamentally simplifies the synthetic workflow. By employing a base-mediated cyclization between alpha-functionalized carbonyls and dichloroalkenes, this novel approach bypasses the need for any transition metal catalyst entirely. The reaction proceeds efficiently in polar aprotic solvents such as DMSO or DMF at moderate temperatures ranging from 80°C to 120°C. This shift in chemistry eliminates the risk of heavy metal contamination at the source, thereby drastically simplifying the purification process and ensuring higher final product purity. The robustness of this method is evidenced by its tolerance for a wide array of substituents, including phenyl, alkyl, alkoxy, and various electron-withdrawing groups, allowing for the rapid generation of diverse furan libraries. This operational simplicity, combined with the use of cheap and readily available raw materials, positions this technology as a superior choice for cost reduction in pharmaceutical intermediates manufacturing.
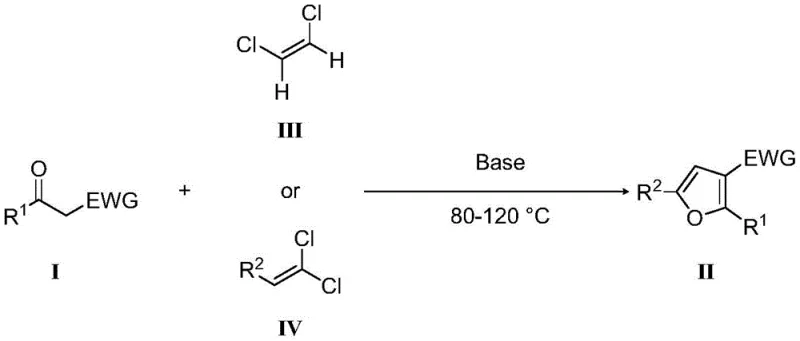
Mechanistic Insights into Base-Mediated Cyclization
The mechanistic pathway of this transformation is rooted in the nucleophilic properties of the enolate generated from the carbonyl precursor. Under the influence of a strong base such as sodium hydride or cesium carbonate, the acidic alpha-proton of the carbonyl compound (Formula I) is abstracted to form a reactive enolate species. This nucleophile then attacks the electron-deficient carbon of the dichloroalkene (Formula III or IV), initiating a cascade of substitution and elimination events. The presence of chlorine atoms on the olefin serves as excellent leaving groups, facilitating the formation of the new carbon-carbon bonds required to close the furan ring. Unlike metal-catalyzed cycles that depend on oxidative addition and reductive elimination steps, this base-mediated mechanism relies purely on ionic interactions and thermodynamic driving forces provided by the formation of the aromatic furan system. This distinct mechanistic profile ensures that the reaction is less sensitive to oxygen and moisture compared to organometallic processes, enhancing its practicality for large-scale operations where strict inert atmosphere control can be costly and difficult to maintain.
From an impurity control perspective, the absence of metal catalysts significantly reduces the complexity of the impurity profile. In metal-catalyzed reactions, side products often arise from homocoupling of the metal species or incomplete catalyst turnover, leading to difficult-to-remove organometallic byproducts. In this base-mediated system, the primary byproducts are inorganic salts derived from the base and the eliminated chloride ions, which are water-soluble and easily removed during the aqueous workup phase. This inherent cleanliness of the reaction mixture translates directly to higher isolated yields, with experimental data showing yields ranging from 64% to 86% across various substrates. For quality control teams, this means a more predictable and manageable purification process, reducing the burden on analytical resources and ensuring consistent batch-to-batch quality essential for regulatory compliance in drug substance manufacturing.
How to Synthesize Polysubstituted Furans Efficiently
The practical implementation of this synthesis route is designed for straightforward execution in standard laboratory and pilot plant equipment. The process begins with the charging of the alpha-substituted carbonyl compound and the selected inorganic base into a reaction vessel, followed by the addition of the polar aprotic solvent. Once the mixture is homogenized, the dichloroalkene reagent is introduced, and the system is heated to the optimal temperature window of 80-120°C. Reaction monitoring is typically conducted via thin-layer chromatography (TLC) to determine the endpoint, ensuring maximum conversion of starting materials. Upon completion, the reaction mixture undergoes a standard workup procedure involving aqueous quenching and extraction, followed by purification via column chromatography to afford the pure polysubstituted furan product. This operational simplicity makes the technology highly accessible for process chemists looking to optimize existing routes or develop new analogs.
- Combine a carbonyl compound with an alpha-position electron-withdrawing group and a base in an aprotic organic solvent within a reaction vessel.
- Add the dichloroalkene reactant to the mixture and heat the reaction system to a temperature between 80°C and 120°C.
- Monitor the reaction progress via TLC, and upon completion, purify the crude product using column chromatography to isolate the pure polysubstituted furan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis protocol offers tangible strategic benefits that extend beyond mere technical feasibility. The most significant advantage is the drastic simplification of the supply chain for raw materials. Transition metal catalysts often have volatile pricing and complex sourcing logistics, whereas the inorganic bases and dichloroalkenes used in this process are commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Furthermore, the elimination of expensive noble metals directly impacts the bill of materials, leading to substantial cost savings that can be passed down through the value chain. The reduced need for specialized metal scavenging resins and the shortened purification timeline also contribute to lower overall manufacturing costs and improved throughput.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts from the process equation results in immediate and significant cost optimization. Without the need for purchasing expensive silver or palladium complexes, the direct material cost is lowered considerably. Additionally, the downstream processing costs are reduced because there is no requirement for metal scavenging agents or extensive analytical testing for residual metals. This streamlined workflow allows for faster batch turnover and reduced labor hours per kilogram of product, enhancing the overall economic efficiency of producing high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like cesium carbonate, sodium hydride, and dichloroethylene ensures a stable and resilient supply chain. Unlike specialized catalysts that may have single-source suppliers or long lead times, these reagents are produced in vast quantities globally. This availability ensures that production schedules can be maintained consistently without the fear of bottlenecking due to reagent scarcity. For supply chain planners, this reliability translates to better inventory management and the ability to respond quickly to fluctuating market demands for furan-based active pharmaceutical ingredients.
- Scalability and Environmental Compliance: The benign nature of the reagents and the absence of heavy metals make this process inherently more environmentally friendly and easier to scale. Waste streams are simpler to treat, consisting mainly of inorganic salts rather than toxic metal sludge, which simplifies compliance with increasingly strict environmental regulations. The robustness of the reaction conditions allows for seamless scale-up from gram-scale laboratory synthesis to multi-ton commercial production without the need for specialized reactor linings or complex safety protocols associated with pyrophoric metal catalysts, facilitating rapid commercialization.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel furan synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for stakeholders evaluating this route for potential integration into their manufacturing portfolios. Understanding these nuances is critical for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this metal-free furan synthesis method?
A: The primary advantage is the complete elimination of transition metal catalysts, which removes the need for expensive metal scavengers and complex purification steps, thereby reducing production costs and preventing metal contamination in pharmaceutical products.
Q: What types of substrates are compatible with this reaction protocol?
A: The method exhibits strong substrate applicability, accommodating various carbonyl compounds with electron-withdrawing groups such as ketones, esters, and cyano groups, as well as diverse dichloroalkenes including 1,2-dichloroethylene and 1,1-dichlorostyrene derivatives.
Q: What are the typical reaction conditions required for this cyclization?
A: The reaction typically proceeds in polar aprotic solvents like DMSO or DMF at temperatures ranging from 80°C to 120°C, using inorganic bases such as cesium carbonate or sodium hydride to facilitate the cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Furan Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies in modernizing the production of complex heterocycles. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries like CN112341409B are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this base-mediated cyclization, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this cost-effective technology for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this metal-free route can optimize your budget. We encourage you to reach out today to request specific COA data and comprehensive route feasibility assessments, allowing us to demonstrate our commitment to delivering high-quality, reliable polysubstituted furan solutions that drive your business forward.
