Advanced Nano-Palladium Catalysis for Scalable Biphenyl Pharmaceutical Intermediate Production
Introduction to Next-Generation Biphenyl Synthesis
The landscape of organic synthesis for pharmaceutical intermediates is undergoing a significant transformation driven by the need for more robust and cost-effective catalytic systems. Patent CN102351620A introduces a groundbreaking methodology for the preparation of biphenyl compounds utilizing a novel nano-palladium catalyst. This technology addresses long-standing challenges in Suzuki-Miyaura coupling reactions, particularly regarding catalyst stability and substrate scope. By leveraging a nanofibrous palladium structure, the invention enables the efficient coupling of aryl boronic acids with both brominated and, crucially, chlorinated aromatic hydrocarbons. This represents a substantial leap forward for industrial applications where operational simplicity and raw material costs are paramount concerns for procurement and R&D teams alike.

The core innovation lies in the catalyst's unique physicochemical properties, which allow it to remain active under ambient conditions without the degradation often seen with traditional homogeneous palladium complexes. For a reliable pharmaceutical intermediate supplier, adopting such a technology means transitioning away from sensitive reagents that require specialized handling infrastructure. The patent details a process that not only simplifies the reaction setup by removing the need for ligands but also expands the viable substrate range to include less reactive chlorides, thereby opening up cheaper starting material options for large-scale manufacturing of complex biaryl scaffolds essential in modern drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional Suzuki coupling protocols have historically relied heavily on palladium sources paired with expensive and air-sensitive phosphine ligands, such as tetrakis(triphenylphosphine)palladium or various bidentate phosphines. These conventional systems impose severe logistical burdens on chemical manufacturing facilities, necessitating strict anhydrous and oxygen-free environments to prevent catalyst deactivation. Furthermore, standard palladium catalysts often exhibit poor activity towards chlorinated arenes due to the strong carbon-chlorine bond, forcing manufacturers to utilize more expensive brominated or iodinated precursors. This reliance on activated substrates significantly inflates the bill of materials, while the sensitivity of the catalysts increases operational risks and complicates scale-up procedures in commercial production environments.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a pre-formed nano-palladium catalyst that exhibits exceptional stability against both water and air. This robustness eliminates the stringent requirement for inert atmosphere processing, allowing reactions to proceed under much more forgiving conditions. The catalyst demonstrates high turnover frequencies even with unactivated chlorinated substrates when appropriate promoters like iodine are employed in polyethylene glycol solvents. By removing the dependency on phosphine ligands, the process not only reduces the complexity of the reaction mixture but also mitigates the risk of phosphine oxide impurities which can be difficult to remove during downstream purification. This ligand-free, air-stable paradigm shifts the economic and operational feasibility of producing high-purity biphenyl derivatives for the fine chemical industry.
Mechanistic Insights into Nano-Palladium Catalyzed Coupling
The efficacy of this system stems from the specific morphology of the catalyst, described in the patent as nanofibrous palladium particles entrapped within an aluminum hydroxide matrix. This structural arrangement provides a high surface-area-to-volume ratio, maximizing the number of active sites available for the oxidative addition of the aryl halide. Unlike molecular palladium complexes that may aggregate into inactive palladium black during the reaction cycle, these nano-clusters maintain their dispersion and catalytic integrity throughout the process. The mechanism likely involves the adsorption of the aryl halide onto the nanoparticle surface, followed by transmetallation with the aryl boronic acid species activated by the base. The unique electronic environment of the nano-palladium facilitates the cleavage of the robust carbon-chlorine bond, a step that is typically the rate-determining bottleneck in conventional homogeneous catalysis.
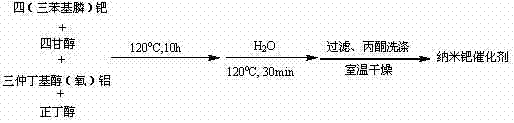
Regarding impurity control, the heterogeneous nature of the nano-catalyst offers distinct advantages over homogeneous counterparts. The solid support helps prevent the leaching of palladium into the solution phase, which simplifies the removal of heavy metal residues from the final product—a critical specification for pharmaceutical intermediates. Additionally, the absence of free phosphine ligands eliminates a major source of side reactions and contamination. The patent data indicates that homocoupling of the boronic acid is minimized under the optimized conditions, particularly when using the PEG-400 solvent system for chlorinated substrates. This selectivity ensures that the desired cross-coupled biphenyl product is formed with high fidelity, reducing the burden on purification units and improving the overall mass balance of the synthetic route.
How to Synthesize Biphenyl Compounds Efficiently
Implementing this synthesis route requires precise adherence to the optimized parameters outlined in the patent to ensure maximum yield and purity. The process begins with the in-situ or ex-situ generation of the nano-palladium catalyst, followed by the careful selection of solvent systems tailored to the specific halide reactivity. For brominated substrates, a mixed solvent of DMF and water proves highly effective at moderate temperatures, whereas chlorinated substrates demand higher thermal energy and the use of PEG-400 with iodine promotion. Understanding these nuances is vital for process chemists aiming to replicate the high yields reported in the literature. The following guide outlines the standardized operational steps derived from the patent examples to facilitate technology transfer.
- Prepare the nano-palladium catalyst by reacting tetrakis(triphenylphosphine)palladium with tetraethylene glycol and aluminum sec-butoxide at 120°C.
- Combine aryl boronic acid and halogenated arene (chloro or bromo) with the catalyst and base in a solvent system like DMF/Water or PEG-400.
- Heat the mixture to 40-140°C depending on substrate reactivity, optionally adding iodine for chlorinated substrates, until completion.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers transformative benefits for supply chain resilience and cost management in the production of fine chemical intermediates. The primary driver for cost reduction is the elimination of expensive phosphine ligands and the ability to utilize cheaper chlorinated starting materials instead of premium brominated or iodinated analogs. By removing the need for specialized inert gas lines and glovebox operations, capital expenditure for reactor setup is significantly lowered, and operational throughput is increased due to simplified charging and discharging procedures. These factors combine to create a manufacturing process that is not only economically superior but also inherently safer and more scalable for multi-ton production campaigns.
- Cost Reduction in Manufacturing: The ligand-free nature of this catalytic system directly translates to substantial raw material savings, as high-cost phosphines are completely removed from the bill of materials. Furthermore, the successful activation of chlorinated arenes allows procurement teams to source significantly less expensive feedstocks compared to their brominated counterparts, driving down the variable cost per kilogram of the final API intermediate. The simplified workup procedure, which avoids complex ligand stripping steps, also reduces solvent consumption and waste disposal costs, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: The air and water stability of the nano-palladium catalyst drastically reduces the risk of batch failures caused by environmental exposure during handling. This robustness ensures consistent reaction performance regardless of minor fluctuations in plant humidity or operator technique, leading to more predictable production schedules. Additionally, the use of common, non-hazardous solvents like PEG-400 and water mixtures mitigates supply chain risks associated with specialized or regulated solvents, ensuring continuous availability of critical reagents for uninterrupted manufacturing cycles.
- Scalability and Environmental Compliance: The heterogeneous nature of the catalyst facilitates easier separation and potential recycling, aligning with green chemistry principles and reducing the environmental footprint of the process. The ability to run reactions in aqueous or semi-aqueous media minimizes the generation of volatile organic compound (VOC) emissions, simplifying compliance with increasingly stringent environmental regulations. This scalability is further supported by the mild reaction conditions for bromides and the manageable high-temperature conditions for chlorides, making the transition from laboratory benchtop to commercial pilot plants seamless and efficient.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this nano-palladium catalyzed Suzuki coupling technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these specifics is crucial for assessing the feasibility of integrating this method into existing production workflows for biphenyl-based pharmaceutical intermediates.
Q: Does this nano-palladium catalyst require inert atmosphere conditions?
A: No, a key advantage of this patented nano-palladium catalyst is its stability towards water and air, eliminating the need for rigorous anhydrous or oxygen-free processing typically required for traditional palladium sources.
Q: Can this method activate low-reactivity chlorinated aromatics?
A: Yes, unlike many conventional catalysts that struggle with chlorides, this nano-palladium system effectively catalyzes the coupling of chlorinated arenes, especially when promoted with additives like iodine in PEG-400 solvent.
Q: Is the addition of expensive phosphine ligands necessary?
A: No, the process is explicitly ligand-free. The nano-structure of the palladium provides sufficient catalytic activity without the need for costly phosphine ligands such as Pd(PPh3)4 or dppf, significantly reducing raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biphenyl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced catalytic technologies to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this nano-palladium coupling are successfully translated into robust industrial processes. We are committed to delivering high-purity biphenyl compounds that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards for residual metals and organic impurities.
We invite you to collaborate with us to leverage these cost-saving synthetic strategies for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your target molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise in nano-catalysis can optimize your supply chain and reduce your overall manufacturing costs effectively.
