Advancing Pharmaceutical Intermediate Production with Recyclable Ionic Liquid Catalysis
Advancing Pharmaceutical Intermediate Production with Recyclable Ionic Liquid Catalysis
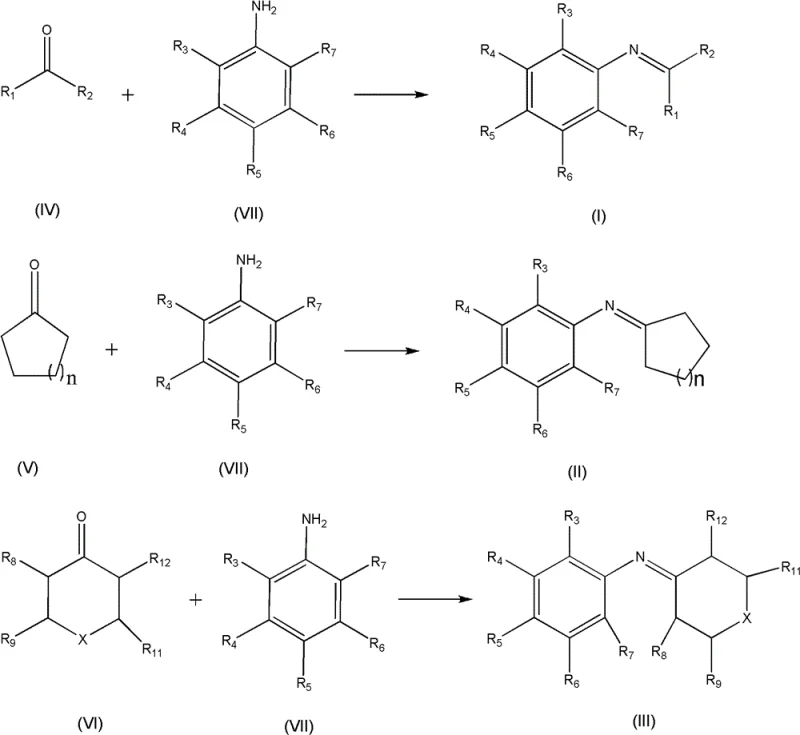
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for sustainable and efficient synthetic methodologies. Patent CN102134197B introduces a groundbreaking green synthesis method for Schiff base compounds, utilizing carboxylic acid-type acidic ionic liquids as catalysts. This technology addresses critical pain points in the production of pharmaceutical and agrochemical intermediates by replacing corrosive traditional acids with a reusable, environmentally benign catalytic system. The process involves the condensation of ketones and aromatic amines, achieving high product yields while simplifying downstream processing. For R&D directors and procurement managers seeking reliable Schiff base suppliers, this innovation represents a pivotal shift towards cost-effective and scalable manufacturing protocols that align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of Schiff bases via the dehydration condensation of aldehydes or ketones with aromatic amines has relied heavily on strong mineral acids, Lewis acids like zinc chloride, or solid acid catalysts. These conventional methods suffer from inherent drawbacks that complicate industrial scale-up and increase operational expenditures. The use of corrosive acids necessitates specialized reactor materials to prevent equipment degradation, leading to higher capital investment and maintenance costs. Furthermore, these catalysts are often difficult to separate from the reaction mixture, requiring extensive neutralization and washing steps that generate substantial volumes of aqueous waste. The low reactivity of ketones compared to aldehydes often demands harsh conditions such as high-temperature reflux with continuous water removal, yet even under these strenuous conditions, yields can remain suboptimal, impacting overall process efficiency and raw material utilization.
The Novel Approach
The novel approach detailed in the patent leverages the unique properties of functionalized acidic ionic liquids to overcome these historical barriers. By employing carboxylic acid-type acidic ionic liquids, the reaction proceeds under milder conditions with significantly enhanced kinetics. These ionic liquids act as both the solvent and the catalyst in some embodiments, or as a highly efficient catalyst in organic solvents, facilitating the removal of water and driving the equilibrium towards product formation. The liquid nature of the catalyst allows for homogeneous mixing, ensuring consistent reaction rates throughout the batch. Most critically, the ionic liquid remains immiscible with certain organic extraction solvents or settles at the bottom of the reactor, enabling simple decantation of the product solution. This physical property eliminates the need for complex filtration or neutralization workflows, drastically reducing the environmental footprint and operational complexity associated with traditional acid-catalyzed condensations.
Mechanistic Insights into Carboxylic Acid Ionic Liquid Catalysis
The efficacy of this synthesis route lies in the dual functionality of the ionic liquid catalyst, which combines the acidity of a carboxylic acid group with the tunable physicochemical properties of an imidazolium cation. The mechanism involves the protonation of the carbonyl oxygen of the ketone substrate by the acidic proton of the ionic liquid, thereby increasing the electrophilicity of the carbonyl carbon. This activation lowers the energy barrier for the nucleophilic attack by the amino group of the aromatic amine. Following the formation of the tetrahedral intermediate, the ionic liquid facilitates the elimination of a water molecule, regenerating the catalyst and yielding the stable imine or Schiff base product. The structural versatility of the ionic liquid, as depicted in the catalyst structure below, allows for fine-tuning of acidity and solubility profiles to match specific substrate requirements, ensuring broad applicability across diverse chemical spaces.
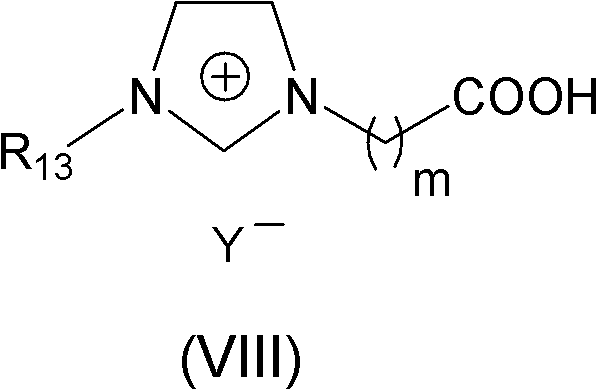
Impurity control is another critical aspect where this mechanistic approach excels. Traditional acid catalysts often promote side reactions such as polymerization or over-alkylation due to their uncontrolled acidity and difficulty in quenching. In contrast, the mild and controlled acidity of the carboxylic acid-functionalized ionic liquid minimizes these side pathways, resulting in a cleaner crude product profile. The absence of metal ions, which are common in Lewis acid catalysts, eliminates the risk of metal contamination in the final API intermediate, a crucial factor for pharmaceutical compliance. The ease of separation means that the product can be isolated simply by removing the solvent, leaving the catalyst behind for reuse, which inherently prevents catalyst-derived impurities from carrying over into the final product stream, thus simplifying the purification burden on downstream processing teams.
How to Synthesize Schiff Base Compounds Efficiently
The operational simplicity of this green synthesis method makes it highly attractive for rapid process development and scale-up. The standard protocol involves charging the reactor with the ionic liquid catalyst, the ketone, and the aromatic amine, followed by the addition of a solvent like toluene or cyclohexane if necessary. The mixture is then heated to reflux with a water separator to drive the reaction to completion. Detailed standardized synthesis steps, including specific molar ratios and temperature profiles optimized for different substrate classes, are outlined in the technical guide below to ensure reproducibility and safety during implementation.
- Charge a reaction vessel with the carboxylic acid-type acidic ionic liquid catalyst, the ketone substrate, and the aromatic amine.
- Add a reaction solvent such as cyclohexane or toluene, or use the raw material ketone as the solvent, and stir the mixture.
- Heat the mixture to reflux with water separation for 0.5 to 40 hours, then pour out the reaction solution to isolate the product and recover the catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid-catalyzed process offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the workflow, which translates directly into reduced operational costs and enhanced supply reliability. By eliminating the need for stoichiometric amounts of corrosive acids and the subsequent neutralization steps, the process reduces the consumption of auxiliary chemicals and the generation of hazardous waste. This streamlined workflow not only lowers the cost of goods sold but also mitigates regulatory risks associated with waste disposal, making the supply chain more resilient to environmental compliance audits.
- Cost Reduction in Manufacturing: The ability to recycle the ionic liquid catalyst multiple times without significant loss of activity represents a major economic driver. Unlike traditional catalysts that are consumed or require complex regeneration, this system allows the catalyst to remain in the reactor for consecutive batches. This reuse capability significantly lowers the effective cost per kilogram of the catalyst, contributing to substantial overall cost savings in pharmaceutical intermediate manufacturing. Additionally, the high yields reported in the patent examples reduce raw material waste, further optimizing the cost structure.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent output quality, which is vital for maintaining uninterrupted supply to downstream customers. The tolerance of the method to various substrates means that a single production line can be adapted to manufacture a wide range of Schiff base derivatives without extensive retooling. This flexibility allows manufacturers to respond quickly to market demands for specific intermediates, reducing lead times for high-purity pharmaceutical intermediates and strengthening the overall agility of the supply network.
- Scalability and Environmental Compliance: Scaling up this process is straightforward due to the liquid nature of the catalyst and the absence of exothermic hazards often associated with strong mineral acids. The low vapor pressure of the ionic liquid minimizes volatile organic compound (VOC) emissions, aligning with increasingly stringent environmental regulations. The simplified workup procedure reduces the load on wastewater treatment facilities, making the process inherently greener and easier to permit in jurisdictions with strict environmental controls, thereby securing long-term production continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the feasibility of adopting this method in your own production facilities or sourcing strategies.
Q: What are the advantages of using carboxylic acid ionic liquids over traditional acids?
A: Unlike traditional mineral acids or Lewis acids which cause equipment corrosion and generate large amounts of waste, carboxylic acid ionic liquids offer low vapor pressure, high thermal stability, and can be directly reused multiple times without significant loss of activity.
Q: Can the ionic liquid catalyst be recycled after the reaction?
A: Yes, the patent demonstrates that after pouring out the reaction solution, the ionic liquid remains at the bottom of the vessel and can be directly reused for subsequent batches, maintaining high yields over at least 5 cycles.
Q: What types of substrates are compatible with this synthesis method?
A: The method is highly versatile, accommodating various ketones including cyclic ketones like cyclohexanone and acyclic ketones, as well as aromatic amines with diverse substituents such as halogens, alkyl groups, and alkoxy groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Schiff Base Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green catalytic technologies in modernizing the production of complex organic intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the ionic liquid-catalyzed Schiff base synthesis can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required by the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to optimize your supply chain for Schiff base intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can deliver both quality and value to your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
