Advanced Catalytic Oxidation for High-Purity Vanillin Production and Commercial Scale-Up
Advanced Catalytic Oxidation for High-Purity Vanillin Production and Commercial Scale-Up
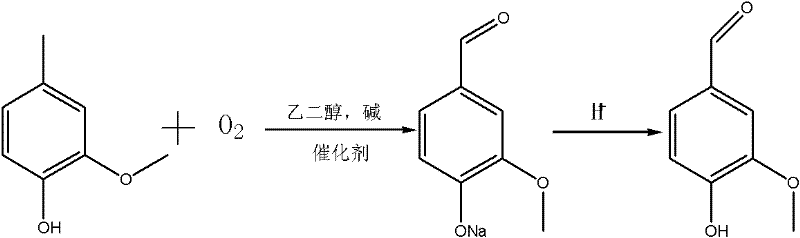
The global demand for vanillin, a critical ingredient in the food, cosmetic, and pharmaceutical industries, continues to drive innovation in synthetic methodologies that balance efficiency with environmental sustainability. Patent CN102381950A introduces a groundbreaking catalytic oxidation method that transforms 4-methylguaiacol into vanillin using novel salt-group modified Schiff base metal chelates. This technology represents a significant departure from conventional processes that rely on simple transition metal salts and hazardous solvents, offering a pathway to higher purity and reduced operational risks. By utilizing ethylene glycol as a solvent system and employing a recoverable catalyst, this approach addresses key pain points in cost reduction in flavor and fragrance manufacturing while ensuring robust supply chain continuity. For R&D directors and procurement specialists, understanding the mechanistic advantages of this route is essential for evaluating next-generation suppliers capable of delivering consistent, high-quality intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of vanillin via the oxidation of 4-methylguaiacol has been plagued by significant technical and safety challenges that hinder optimal production efficiency. Traditional protocols frequently employ simple transition metal salts, such as cobalt or copper salts, dissolved in strong alkaline methanol solutions to facilitate the oxidation reaction. While these methods can achieve conversion rates around 90%, they suffer from severe downstream processing difficulties, particularly the arduous task of separating the homogeneous metal catalyst from the final product. Furthermore, the reliance on methanol introduces substantial safety liabilities due to its low flash point, high toxicity, and wide explosive limits, necessitating expensive specialized equipment to mitigate fire and health hazards. The inability to effectively recycle these metal salts not only inflates raw material costs but also contributes to heavy metal waste streams, complicating environmental compliance and increasing the burden of three-waste treatment facilities.
The Novel Approach
In stark contrast, the methodology described in CN102381950A leverages a sophisticated catalyst design that fundamentally alters the reaction landscape to favor safety, selectivity, and recyclability. By replacing methanol with ethylene glycol or a mixture of ethylene glycol and water, the process eliminates the volatility risks associated with traditional solvents, creating a inherently safer reaction environment suitable for large-scale operations. The core innovation lies in the use of salt-group modified Schiff base metal chelates, which exhibit unique solubility properties that allow them to remain active during the reaction yet precipitate or separate easily upon the addition of water post-reaction. This heterogenized behavior enables the catalyst to be recovered via simple centrifugal filtration and reused multiple times without significant loss of activity, directly addressing the separation bottlenecks of prior art. Consequently, this novel approach achieves a complete 100% conversion of the starting material with yields consistently between 85% and 95%, demonstrating superior performance metrics for a reliable vanillin supplier.
Mechanistic Insights into Schiff Base Metal Chelate Catalysis
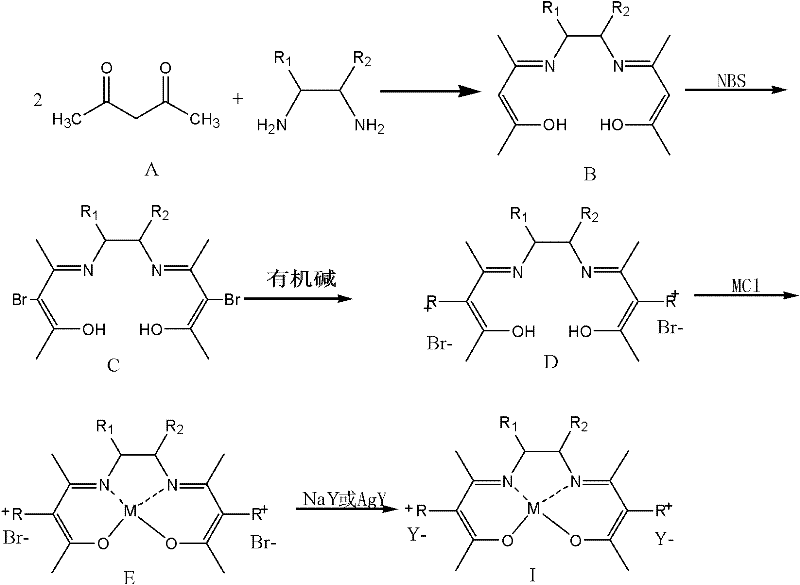
The exceptional performance of this oxidation process is rooted in the precise molecular architecture of the salt-group modified Schiff base metal chelates, which function as highly efficient oxygen carriers. Unlike simple metal ions that may aggregate or deactivate rapidly, the Schiff base ligand provides a rigid, stable coordination environment that protects the central metal ion (such as Fe, Co, Cu, or Mn) while facilitating electron transfer during the oxidation cycle. The modification with ionic liquid-like salt groups further enhances this stability and imparts the crucial solubility switch that permits catalyst recovery; these groups render the complex insoluble in the aqueous workup phase while maintaining compatibility with the organic reaction medium. This structural integrity ensures that the catalyst maintains high activity over extended periods, minimizing the formation of unwanted by-products and preserving the aromatic profile of the vanillin. For technical teams, this means the impurity profile is tightly controlled, reducing the need for aggressive purification steps that often degrade yield.
Furthermore, the mechanism involves a selective activation of molecular oxygen or air under atmospheric pressure, which is a greener alternative to stoichiometric oxidants that generate massive amounts of inorganic waste. The interaction between the metal center and the oxygen molecule is optimized by the electronic properties of the ligand substituents (R1, R2, and the ionic group R), allowing for a controlled radical or non-radical oxidation pathway that targets the methyl group of 4-methylguaiacol specifically. This high selectivity (>85%) prevents over-oxidation to carboxylic acids or ring degradation, which are common failure modes in less sophisticated catalytic systems. The ability to tune the metal center and the anionic counter-ion (Y-) provides a versatile platform for optimizing reaction kinetics for specific substrate batches, ensuring that the process remains robust against variations in raw material quality. Such mechanistic control is vital for achieving the high-purity vanillin specifications required by discerning global markets.
How to Synthesize Vanillin Efficiently
The synthesis of vanillin using this advanced catalytic system involves a streamlined sequence that integrates catalyst preparation with the oxidation step to maximize overall process efficiency. The initial phase requires the construction of the Schiff base ligand through the condensation of acetylacetone with various diamines, followed by bromination and quaternization with organic bases to install the ionic functionality. Once the metal chelate is formed and the anion exchanged to the desired species, it is introduced into the oxidation reactor along with 4-methylguaiacol, a base like sodium hydroxide, and the ethylene glycol solvent. The reaction proceeds under mild thermal conditions (60-120°C) with a continuous flow of oxygen or air, eliminating the need for high-pressure autoclaves typically associated with gas-liquid oxidations. Detailed standardized synthesis steps and specific parameter optimizations for different catalyst variants are outlined in the comprehensive guide below.
- Prepare the salt-group modified Schiff base metal chelate catalyst by reacting acetylacetone with diamines, followed by bromination, ionic liquid support, metal chelation, and anion exchange.
- Conduct the oxidation reaction by mixing 4-methylguaiacol, base (NaOH/KOH), ethylene glycol solvent, and the catalyst, then introducing oxygen or air at 60-120°C for 6-25 hours.
- Recover the catalyst via centrifugal filtration after adding water, then isolate the vanillin sodium salt through crystallization, acidification, and recrystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology translates into tangible strategic advantages that extend far beyond simple yield improvements. The shift from hazardous methanol to ethylene glycol drastically simplifies facility safety requirements and reduces insurance and compliance costs associated with handling volatile organic compounds. Moreover, the recoverability of the expensive metal catalyst represents a significant opportunity for cost reduction in flavor and fragrance manufacturing, as the effective catalyst loading per ton of product is minimized through multiple reuse cycles. This efficiency gain is compounded by the reduction in heavy metal waste disposal fees, aligning production practices with increasingly stringent global environmental regulations. By mitigating the risks of supply disruptions caused by safety incidents or regulatory shutdowns, this process enhances the overall resilience of the supply chain.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the elimination of costly downstream purification steps required to remove residual metal contaminants from the final product. Since the catalyst separates cleanly from the reaction mixture, the need for complex chelating agents or extensive washing protocols is removed, leading to substantial savings in utility consumption and processing time. Additionally, the high conversion rate ensures that raw material utilization is maximized, reducing the cost of goods sold by minimizing the loss of valuable 4-methylguaiacol to unreacted feedstock or degradation products. These cumulative efficiencies allow for a more competitive pricing structure without compromising on the quality or purity of the delivered vanillin.
- Enhanced Supply Chain Reliability: Operational stability is a critical factor for maintaining consistent supply to downstream customers, and this technology offers superior reliability compared to traditional batch processes. The use of atmospheric pressure and moderate temperatures reduces mechanical stress on reactor vessels and associated piping, lowering the frequency of maintenance downtime and unplanned outages. Furthermore, the robustness of the catalyst against deactivation means that production campaigns can run for longer durations without the need for frequent catalyst replenishment or system cleaning. This continuity is essential for reducing lead time for high-purity vanillin deliveries, ensuring that inventory levels remain stable even during periods of surging market demand.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the benign nature of the solvent system and the simplicity of the catalyst recovery mechanism. The absence of explosive solvents removes a major barrier to increasing reactor size, allowing for the commercial scale-up of complex flavor intermediates with minimal engineering modifications. From an environmental perspective, the reduction in toxic waste generation and the potential for solvent recycling contribute to a smaller carbon footprint, which is increasingly valued by end-consumer brands seeking sustainable sourcing. This alignment with green chemistry principles not only future-proofs the manufacturing asset but also enhances the brand value of the supplied ingredients in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic oxidation technology for vanillin production. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing supply chains or R&D pipelines. The answers reflect the specific advantages of the Schiff base catalyst system over legacy technologies.
Q: What are the primary advantages of using Schiff base metal chelates over traditional transition metal salts in vanillin synthesis?
A: Schiff base metal chelates modified with salt groups offer superior structural stability and higher catalytic activity compared to simple transition metal salts. Crucially, their insolubility in the reaction medium allows for easy separation and repeated reuse, significantly reducing catalyst consumption and metal contamination in the final product.
Q: How does the use of ethylene glycol improve the safety profile of the vanillin oxidation process?
A: Traditional methods often utilize methanol, which is highly toxic, flammable, and possesses a wide explosion limit, posing significant safety risks during large-scale oxidation. Replacing methanol with ethylene glycol or its aqueous mixture eliminates these volatility hazards, creating a much safer operating environment for industrial production facilities.
Q: What level of purity and yield can be expected from this catalytic oxidation method?
A: This patented process achieves a 4-methylguaiacol conversion rate of 100% with a product yield ranging between 85% and 95%. The resulting vanillin exhibits a purity of approximately 98.0% with a selectivity greater than 85%, meeting stringent quality standards for food and cosmetic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vanillin Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes like the one described in CN102381950A requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of vanillin meets the exacting standards required for food and pharmaceutical applications. Our commitment to process innovation allows us to offer solutions that are not only chemically superior but also economically viable for long-term partnerships.
We invite you to engage with our technical procurement team to discuss how this catalytic oxidation technology can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits of switching to this greener, more efficient production method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to serve as your trusted source for high-performance flavor and fragrance intermediates.
