Revolutionizing Aromatic Amine Synthesis via Direct Nitrogen Fixation for Commercial Scale-up
Revolutionizing Aromatic Amine Synthesis via Direct Nitrogen Fixation for Commercial Scale-up
The chemical industry is currently witnessing a paradigm shift in how we approach the synthesis of high-value nitrogen-containing scaffolds, driven by the urgent need for more sustainable and cost-effective feedstocks. Patent CN112759520B introduces a groundbreaking methodology that bypasses traditional ammonia-dependent routes, instead utilizing molecular nitrogen (N2) directly as the nitrogen source for constructing organic aromatic nitrogen-containing compounds. This innovation is particularly transformative for the production of aromatic amines and carbazoles, which serve as critical building blocks in the pharmaceutical and organic light-emitting diode (OLED) sectors. By leveraging a unique lithium-mediated reduction followed by transition metal catalysis, this technology offers a reliable aromatic amine supplier pathway that drastically reduces dependency on the energy-intensive Haber-Bosch process derivatives. The strategic implication for R&D directors and procurement managers is profound, as it opens a new avenue for securing high-purity intermediates with a significantly reduced carbon footprint and enhanced supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of aromatic amines and related heterocycles has relied heavily on ammonia or pre-functionalized amine sources, which present substantial logistical and economic challenges. The storage and handling of anhydrous ammonia require specialized high-pressure infrastructure and rigorous safety protocols due to its toxicity and volatility, creating bottlenecks in cost reduction in electronic chemical manufacturing. Furthermore, conventional methods such as the o-aminobiphenyl ring-closure dehydrogenation or aluminum trichloride catalysis often suffer from harsh reaction conditions, poor atom economy, and limited substrate scope. These legacy processes frequently generate significant amounts of hazardous waste and struggle to accommodate sensitive functional groups, necessitating complex protecting group strategies that inflate production costs and extend lead times. For supply chain heads, the reliance on these mature but inefficient technologies means dealing with fluctuating raw material prices and rigid manufacturing constraints that hinder rapid scale-up.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes atmospheric nitrogen, the most abundant element in the earth's atmosphere, as a direct feedstock, effectively decoupling production from the volatile ammonia market. This novel approach employs a tandem sequence where nitrogen is first activated by lithium powder to form lithium nitride, which then undergoes a palladium-catalyzed cross-coupling with aryl halides in a single pot. This one-pot design eliminates the need to isolate the reactive lithium nitride intermediate, thereby enhancing operational safety and process efficiency. The system demonstrates remarkable versatility, accommodating a wide range of aryl bromides and iodides with diverse electronic and steric properties. By shifting the nitrogen source from a processed chemical to a ubiquitous gas, manufacturers can achieve substantial cost savings and improve the sustainability profile of their synthetic routes, making it an ideal solution for the commercial scale-up of complex organic intermediates.
Mechanistic Insights into Lithium-Mediated Nitrogen Fixation and Pd-Catalysis
The core of this technological breakthrough lies in the synergistic combination of strong reductive activation and sophisticated transition metal catalysis. The process initiates with the reduction of inert dinitrogen (N≡N) by lithium metal at elevated temperatures, typically around 150°C, to generate lithium nitride (Li3N). This step is critical as it breaks the formidable triple bond of nitrogen, converting it into a nucleophilic nitrogen source capable of participating in subsequent bond-forming events. Following the formation of the nitride species, the reaction transitions into a palladium-catalyzed coupling regime. A zero-valent palladium catalyst, such as tris(dibenzylideneacetone)dipalladium, coordinates with the aryl halide substrate to form an aryl-palladium complex. The presence of bulky, electron-rich phosphine ligands, such as 2-dicyclohexylphosphino-2',6'-diisopropoxy-1,1'-biphenyl, is essential for stabilizing the active catalytic species and facilitating the oxidative addition step, even with sterically hindered substrates.
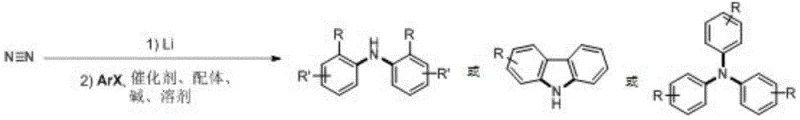
Once the aryl-palladium species is formed, it interacts with the in situ generated lithium nitride. The phase transfer catalyst, typically a tetraalkylammonium salt like tetrabutylammonium bromide, plays a pivotal role in solubilizing the ionic lithium nitride in the organic solvent, ensuring efficient contact with the organometallic catalyst. The subsequent reductive elimination releases the desired aromatic amine product and regenerates the palladium catalyst for the next cycle. This mechanistic pathway allows for the construction of C-N bonds with high precision and yield, as evidenced by the successful synthesis of diverse targets like triphenylamine and carbazole derivatives. The robustness of this catalytic cycle ensures that impurities are minimized, providing R&D teams with a clean reaction profile that simplifies downstream purification and quality control processes.
How to Synthesize Aromatic Amines Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and the maintenance of an inert atmosphere to prevent the oxidation of the lithium reagent. The process is designed to be operationally simple, consolidating what would traditionally be multiple steps into a streamlined one-pot procedure. Detailed standard operating procedures regarding stoichiometry, solvent selection, and workup protocols are critical for maximizing yield and reproducibility. For laboratory and pilot plant chemists, understanding the precise timing of the nitrogen reduction phase versus the coupling phase is key to success. The detailed standardized synthesis steps are outlined in the guide below to ensure consistent results across different batches.
- Reduce nitrogen gas using lithium powder at elevated temperatures (approx. 150°C) to generate lithium nitride in situ.
- Add aryl halide substrates, a zero-valent palladium catalyst, specialized phosphine ligands, alkali metal alkoxide base, and a phase transfer catalyst to the reaction mixture.
- Maintain the reaction temperature between 120°C and 160°C for 8 to 48 hours to complete the coupling and form the final aromatic amine or carbazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this nitrogen-fixation technology represents a significant opportunity to optimize the cost structure and reliability of raw material sourcing. By replacing expensive and logistically challenging ammonia or amine precursors with inexpensive nitrogen gas and lithium, the direct material costs associated with nitrogen incorporation are drastically reduced. This shift not only lowers the bill of materials but also mitigates the risks associated with the transportation and storage of hazardous compressed gases. The simplified one-pot workflow reduces the number of unit operations required, leading to lower energy consumption and reduced labor costs per kilogram of product. These efficiencies translate directly into improved margins and a more competitive pricing structure for high-value intermediates in the global market.
- Cost Reduction in Manufacturing: The elimination of pre-functionalized amine starting materials removes a major cost driver from the supply chain, as nitrogen gas is virtually free and lithium is a commodity metal. Furthermore, the high atom economy of the direct coupling reaction minimizes waste generation, reducing the costs associated with waste disposal and environmental compliance. The use of efficient catalytic systems ensures that precious metal loading is kept to a minimum while maintaining high turnover numbers, further contributing to overall process economics. This comprehensive approach to cost optimization allows manufacturers to offer high-purity organic intermediates at a more attractive price point without compromising on quality.
- Enhanced Supply Chain Reliability: Relying on atmospheric nitrogen as a feedstock insulates the production process from the supply volatility often seen with specialized amine reagents. Since nitrogen can be generated on-site or sourced easily, the risk of supply disruption due to vendor issues or transportation delays is significantly mitigated. The robust nature of the catalytic system also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different suppliers. This stability is crucial for maintaining continuous production schedules and meeting the stringent delivery commitments required by downstream pharmaceutical and electronics clients.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of highly toxic reagents make this process inherently safer and easier to scale from kilogram to multi-ton quantities. The reduced generation of hazardous byproducts aligns with increasingly strict environmental regulations, simplifying the permitting process for new manufacturing facilities. Additionally, the compatibility of the method with flow chemistry setups offers potential for further intensification and safety improvements in large-scale production. This scalability ensures that the technology can grow alongside market demand, providing a future-proof solution for the manufacturing of complex aromatic nitrogen compounds.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrogen fixation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to provide accurate guidance for potential adopters. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production lines. We encourage technical teams to review these points carefully to assess the fit for their specific application requirements.
Q: What are the primary advantages of using nitrogen gas over ammonia for synthesizing aromatic amines?
A: Using nitrogen gas eliminates the logistical hazards and storage costs associated with anhydrous ammonia. Furthermore, this direct fixation method described in patent CN112759520B operates under milder conditions compared to traditional high-pressure ammonia processes, significantly improving safety profiles and functional group tolerance.
Q: Is this catalytic system compatible with sensitive functional groups on the aryl halide substrate?
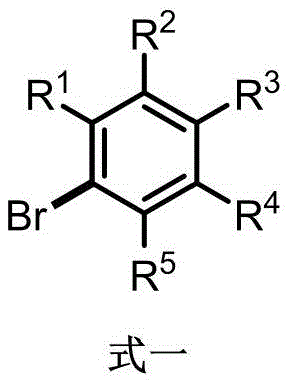
A: Yes, the protocol demonstrates excellent compatibility with various substituents including methyl, methoxy, trifluoromethoxy, and trimethylsilyl groups. The use of bulky phosphine ligands protects the catalytic center, allowing for the synthesis of complex intermediates without extensive protecting group strategies.
Q: Can this process be scaled for industrial production of OLED materials?
A: The one-pot nature of the reaction simplifies the workflow by avoiding the isolation of unstable lithium nitride intermediates. This streamlined approach, combined with the use of abundant nitrogen feedstock, makes it highly suitable for the commercial scale-up of complex organic intermediates used in optoelectronic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of direct nitrogen fixation technologies in reshaping the landscape of fine chemical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of aromatic amines or carbazoles meets the exacting standards of the pharmaceutical and electronic materials industries. We are committed to leveraging cutting-edge chemistry like the lithium-mediated nitrogen fixation described in CN112759520B to deliver superior value to our global clientele.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments for your target molecules, and let us demonstrate how our expertise can accelerate your project timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
