Scalable Synthesis of 3-Hydroxy-2-Oxindole Intermediates for Global Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust pathways for constructing complex heterocyclic scaffolds, particularly the 3-hydroxy-2-oxindole core found in numerous bioactive natural products. Patent CN102659662A introduces a transformative synthetic methodology that utilizes inexpensive isatin and alpha-substituted acetic acid precursors under mild base catalysis. This approach addresses critical bottlenecks in traditional manufacturing by offering a versatile route that tolerates diverse functional groups while maintaining exceptional reaction efficiency. The technology represents a significant leap forward for producing high-purity pharmaceutical intermediates, ensuring that supply chains remain resilient against raw material volatility. By leveraging common industrial reagents and avoiding sensitive metal catalysts, this process aligns perfectly with modern green chemistry principles and cost-effective manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, constructing the 3-hydroxy-2-oxindole skeleton relied heavily on metal-catalyzed additions or multi-step sequences involving hazardous reagents and stringent anhydrous conditions. Traditional aldol reactions often suffered from limited substrate scope, requiring specialized ketones or aldehydes that are difficult to source commercially at scale. Furthermore, the reliance on transition metals introduced significant downstream purification challenges, necessitating expensive heavy metal removal steps to meet regulatory standards for pharmaceutical ingredients. These conventional pathways frequently exhibited poor atom economy and generated substantial chemical waste, thereby inflating production costs and complicating environmental compliance. The sensitivity of many prior art methods to moisture and oxygen also posed severe risks during commercial scale-up, leading to inconsistent batch quality and extended lead times.
The Novel Approach
In stark contrast, the disclosed invention employs a direct condensation strategy driven by accessible organic or inorganic bases, drastically simplifying the operational complexity of the synthesis. 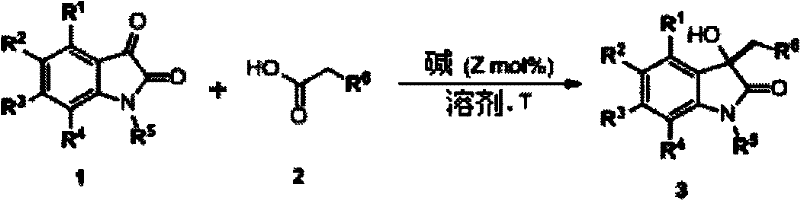 The new protocol operates effectively in common solvents like DMF or THF at moderate temperatures ranging from room temperature to 100°C, eliminating the need for cryogenic conditions or high-pressure equipment. This method demonstrates remarkable universality, accommodating a wide array of substituents on the isatin ring without compromising yield or selectivity. By utilizing cheap and stable starting materials, the process inherently lowers the barrier to entry for large-scale production while ensuring consistent supply continuity. The simplicity of the work-up procedure, involving standard aqueous extraction and crystallization, further enhances its appeal for industrial adoption by reducing processing time and solvent consumption.
The new protocol operates effectively in common solvents like DMF or THF at moderate temperatures ranging from room temperature to 100°C, eliminating the need for cryogenic conditions or high-pressure equipment. This method demonstrates remarkable universality, accommodating a wide array of substituents on the isatin ring without compromising yield or selectivity. By utilizing cheap and stable starting materials, the process inherently lowers the barrier to entry for large-scale production while ensuring consistent supply continuity. The simplicity of the work-up procedure, involving standard aqueous extraction and crystallization, further enhances its appeal for industrial adoption by reducing processing time and solvent consumption.
Mechanistic Insights into Base-Catalyzed Cyclization
The core of this technological advancement lies in the efficient activation of the alpha-substituted acetic acid by the base catalyst to form a reactive enol intermediate. 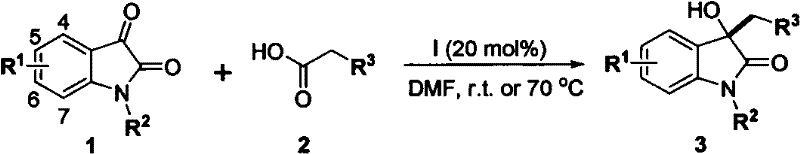 This nucleophilic species subsequently undergoes a precise 1,2-addition to the C3 carbonyl of the isatin molecule, forming a critical tetrahedral intermediate that dictates the stereochemistry of the final product. The mechanism proceeds through a smooth intramolecular hydrogen transfer followed by decarboxylation, which drives the reaction equilibrium towards the desired 3-hydroxy-2-oxindole structure. This elegant cascade avoids the formation of stable by-products that typically plague metal-catalyzed routes, ensuring a cleaner reaction profile. The ability to tune the electronic properties of the base catalyst allows chemists to optimize reaction kinetics for specific substrates, providing a high degree of control over the synthetic outcome.
This nucleophilic species subsequently undergoes a precise 1,2-addition to the C3 carbonyl of the isatin molecule, forming a critical tetrahedral intermediate that dictates the stereochemistry of the final product. The mechanism proceeds through a smooth intramolecular hydrogen transfer followed by decarboxylation, which drives the reaction equilibrium towards the desired 3-hydroxy-2-oxindole structure. This elegant cascade avoids the formation of stable by-products that typically plague metal-catalyzed routes, ensuring a cleaner reaction profile. The ability to tune the electronic properties of the base catalyst allows chemists to optimize reaction kinetics for specific substrates, providing a high degree of control over the synthetic outcome.
Impurity control is inherently managed through the mildness of the reaction conditions, which prevent the degradation of sensitive functional groups often present in complex drug intermediates. The absence of aggressive reagents minimizes side reactions such as polymerization or over-oxidation, resulting in a crude product profile that is easier to purify to pharmaceutical grades. This high level of chemical fidelity is crucial for R&D teams aiming to synthesize analog libraries for structure-activity relationship studies without being hindered by purification bottlenecks. Moreover, the compatibility with chiral bases opens avenues for asymmetric synthesis, enabling the production of single-enantiomer compounds required for modern drug safety profiles. Such mechanistic robustness ensures that the process remains reliable even when scaling from gram to ton quantities.
How to Synthesize 3-Hydroxy-2-Oxindole Efficiently
Implementing this synthesis route requires careful attention to solvent selection and catalyst loading to maximize yield and minimize reaction time. The standard protocol involves dissolving the isatin derivative in a polar solvent, followed by the sequential addition of the acetic acid component and the base catalyst under stirring. Operators should monitor the reaction progress via thin-layer chromatography to determine the optimal quenching point, ensuring complete conversion of the starting material. Post-reaction processing involves simple aqueous work-up and recrystallization, which can be easily automated in a pilot plant setting. This straightforward operational flow reduces the training burden on technical staff and minimizes the risk of human error during manufacturing campaigns.
- Dissolve isatin substrate in a polar aprotic solvent such as DMF or THF at room temperature.
- Add alpha-substituted acetic acid and a catalytic amount of organic or inorganic base.
- Heat the mixture to 70°C, monitor by TLC, and purify the resulting solid via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial strategic benefits by decoupling production from the volatile pricing of precious metal catalysts and specialized reagents. The reliance on commodity chemicals like isatin and triethylamine ensures that raw material costs remain stable and predictable over long-term supply contracts. Cost Reduction in Manufacturing: The elimination of expensive transition metals and the associated scavenging resins directly lowers the bill of materials, while the simplified purification process reduces solvent usage and energy consumption. By removing complex metal removal steps, manufacturers can achieve significant operational expenditure savings and shorten the overall production cycle time. This economic efficiency translates into more competitive pricing for downstream pharmaceutical clients seeking to optimize their own cost structures.
- Enhanced Supply Chain Reliability: The use of widely available industrial feedstocks mitigates the risk of supply disruptions caused by geopolitical issues or niche supplier dependencies. Since the reagents are common commercial items, procurement teams can easily qualify multiple vendors to ensure redundancy and continuity of supply. The robustness of the reaction conditions also means that production is less susceptible to delays caused by equipment failures or environmental control issues. This reliability is critical for maintaining just-in-time inventory levels and meeting strict delivery commitments to global partners.
- Scalability and Environmental Compliance: The mild thermal profile and low toxicity of the reagents facilitate seamless scale-up from laboratory benchtop to multi-ton commercial reactors without extensive re-engineering. The process generates minimal hazardous waste, aligning with increasingly stringent environmental regulations and reducing the costs associated with waste disposal and treatment. This eco-friendly profile enhances the corporate sustainability metrics of manufacturing partners, making it an attractive option for companies with green chemistry mandates. The combination of scalability and compliance ensures long-term viability for the production of these key pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology in industrial settings. These insights are derived directly from the patent data and practical experience in fine chemical manufacturing, providing clarity on feasibility and performance. Understanding these aspects helps stakeholders make informed decisions about integrating this route into their existing production portfolios.
Q: What are the primary advantages of this base-catalyzed method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts and harsh reaction conditions, significantly reducing raw material costs and simplifying post-reaction purification processes while maintaining high yields.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes readily available commercial reagents like isatin and operates under mild thermal conditions with low sensitivity to water and oxygen, making it highly robust for commercial scale-up.
Q: What level of stereochemical control can be achieved with this protocol?
A: By employing specific chiral base catalysts such as cinchona alkaloid derivatives, the method enables asymmetric synthesis, providing access to enantiomerically enriched 3-hydroxy-2-oxindole scaffolds essential for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-2-Oxindole Supplier
At NINGBO INNO PHARMCHEM, we leverage this advanced synthetic technology to deliver high-quality intermediates that meet the rigorous demands of the global pharmaceutical industry. Our facility boasts extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch adheres to the highest international standards for drug substance manufacturing. Our commitment to technical excellence allows us to navigate complex chemical landscapes efficiently, providing you with a dependable source for critical building blocks.
We invite you to collaborate with our technical procurement team to explore how this optimized synthesis route can benefit your specific project requirements. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume needs and timeline constraints. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and commercialization efforts. Partner with us to secure a sustainable and cost-effective supply chain for your next generation of therapeutic agents.
