Scalable Base-Catalyzed Synthesis of 3-Hydroxy-2-Oxindole Pharmaceutical Intermediates
Scalable Base-Catalyzed Synthesis of 3-Hydroxy-2-Oxindole Pharmaceutical Intermediates
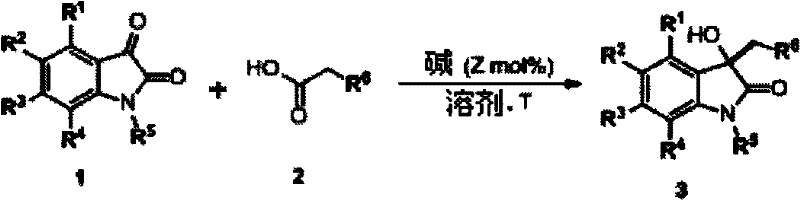
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for constructing complex heterocyclic scaffolds that serve as the backbone for bioactive molecules. A significant advancement in this domain is detailed in patent CN102659662A, which discloses a highly efficient method for synthesizing 3-R-3-hydroxy-2-oxindole compounds. This core structure is prevalent in numerous natural products and drug candidates exhibiting potent biological activities, including antioxidant, anticancer, and neuroprotective properties. The disclosed methodology utilizes readily available isatin and alpha-substituted acetic acids as starting materials, reacting them under the influence of a base catalyst to achieve the target skeleton. This approach represents a paradigm shift from traditional metal-dependent syntheses, offering a pathway that is not only chemically elegant but also commercially viable for the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3-hydroxy-2-oxindole framework has relied heavily on methods that present significant challenges for industrial application. Prior art techniques often involve the use of expensive transition metal catalysts or require harsh reaction conditions that can degrade sensitive functional groups. Many existing protocols suffer from limited substrate scope, meaning they are only effective for specific, narrowly defined substrates rather than a broad range of derivatives. Furthermore, metal-catalyzed routes introduce the risk of heavy metal contamination, necessitating rigorous and costly purification steps to meet stringent regulatory standards for active pharmaceutical ingredients. These factors collectively contribute to higher production costs, longer lead times, and increased environmental burden, limiting the practical utility of these conventional methods in large-scale manufacturing settings.
The Novel Approach
In contrast, the novel base-catalyzed approach described in the patent data offers a streamlined and versatile solution to these longstanding problems. By employing simple organic or inorganic bases such as triethylamine, DBU, or sodium acetate, the reaction proceeds efficiently without the need for precious metals. The process operates under mild conditions, typically ranging from room temperature to 70°C, which significantly reduces energy consumption and enhances safety profiles. This method demonstrates exceptional universality, accommodating a wide variety of substituents on the isatin ring, including halogens, nitro groups, and alkyl chains, thereby enabling the rapid generation of diverse compound libraries. The simplicity of the post-treatment process, involving standard extraction and chromatography, further underscores its suitability for cost-effective and sustainable chemical production.
Mechanistic Insights into Base-Catalyzed Decarboxylative Addition
The success of this synthetic strategy lies in its elegant mechanistic pathway, which facilitates the formation of the quaternary carbon center at the C3 position of the oxindole ring. The reaction initiates when the base catalyst abstracts an acidic proton from the alpha-substituted acetic acid, generating a reactive enol or enolate intermediate. This nucleophilic species then undergoes a 1,2-addition to the electrophilic ketone carbonyl at the C3 position of the isatin molecule. Following this addition, an intramolecular hydrogen transfer occurs, leading to an unstable intermediate that subsequently undergoes decarboxylation. This decarboxylation step is crucial as it drives the reaction forward and installs the desired functional group while releasing carbon dioxide, ultimately yielding the stable 3-hydroxy-2-oxindole product after protonation.
From a quality control perspective, this mechanism offers distinct advantages regarding impurity profiles and product purity. Because the reaction avoids transition metals, the final product is free from metal residues that often complicate downstream processing and regulatory approval. The mild reaction conditions minimize the formation of side products associated with thermal degradation or over-reaction, resulting in cleaner crude mixtures. Additionally, the tolerance of various functional groups allows for the direct synthesis of complex intermediates without the need for extensive protecting group strategies. This inherent selectivity and cleanliness of the reaction pathway ensure that the resulting high-purity oxindole derivatives meet the rigorous specifications required for use in drug discovery and development pipelines.
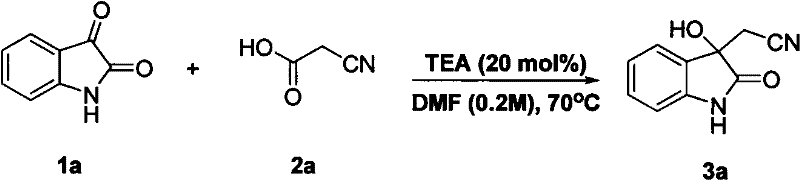
How to Synthesize 3-Cyanomethyl-3-hydroxyoxindole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and efficiency. The protocol typically involves dissolving the isatin substrate in a polar aprotic solvent such as DMF or THF, followed by the sequential addition of the alpha-substituted acetic acid and the chosen base catalyst. While the reaction can proceed at room temperature, heating to approximately 70°C often accelerates the kinetics, ensuring complete conversion within a few hours. Monitoring the reaction progress via thin-layer chromatography (TLC) is recommended to determine the optimal endpoint before proceeding to the workup phase, which involves aqueous extraction and purification. For a comprehensive guide on the standardized operating procedures and specific stoichiometric ratios, please refer to the detailed synthesis steps provided below.
- Dissolve isatin substrate in a polar aprotic solvent such as DMF or THF at room temperature.
- Add alpha-substituted acetic acid (e.g., cyanoacetic acid) and a base catalyst like triethylamine to the reaction mixture.
- Heat the reaction to 70°C or maintain at room temperature depending on substrate reactivity, monitor by TLC, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this base-catalyzed technology translates into tangible strategic benefits that enhance overall operational efficiency. The elimination of expensive noble metal catalysts directly impacts the bill of materials, leading to substantial cost savings without compromising on reaction performance. Furthermore, the use of commodity chemicals like isatin and cyanoacetic acid ensures a stable and reliable supply chain, mitigating the risks associated with sourcing specialized or scarce reagents. The robustness of the process also means that production schedules are less likely to be disrupted by complex handling requirements or sensitivity to environmental factors, thereby improving delivery reliability for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary economic driver for this technology is the complete removal of transition metal catalysts, which are often cost-prohibitive and require specialized disposal protocols. By utilizing inexpensive organic bases like triethylamine or inorganic salts, manufacturers can drastically reduce raw material costs while simplifying the waste management process. The mild reaction temperatures further contribute to energy efficiency, lowering the utility costs associated with heating and cooling large-scale reactors. Additionally, the high yields reported across various substrates minimize material loss, ensuring that the maximum amount of starting material is converted into valuable product, thus optimizing the overall cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available, industrial-grade starting materials significantly de-risks the supply chain for this key pharmaceutical scaffold. Isatin and its derivatives are produced globally in large quantities, ensuring that procurement teams can secure consistent volumes without facing shortages or price volatility. The insensitivity of the reaction system to water and oxygen simplifies storage and handling requirements, allowing for greater flexibility in logistics and inventory management. This stability ensures that production can continue uninterrupted, providing a dependable source of high-quality intermediates for downstream drug synthesis and reducing the lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with green chemistry principles, facilitating easier compliance with increasingly strict environmental regulations. The absence of heavy metals eliminates the need for complex scavenging steps and reduces the toxicity of the effluent, simplifying wastewater treatment and disposal. The straightforward workup procedure, involving standard extraction and crystallization, is easily adaptable to multi-kilogram and ton-scale production, supporting the commercial scale-up of complex heterocycles. This scalability ensures that the technology can grow with the demand of the drug candidate, from early-stage clinical trials to full commercial launch, without requiring significant process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the experimental data and scope analysis presented in the patent documentation, providing clarity on catalyst selection, substrate compatibility, and process optimization. Understanding these nuances is essential for R&D teams evaluating this technology for integration into their existing manufacturing workflows and for procurement specialists assessing its long-term viability.
Q: What are the advantages of this base-catalyzed method over metal-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts, reducing heavy metal contamination risks and simplifying purification, which is critical for pharmaceutical compliance.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the process uses commercially available raw materials like isatin and operates under mild conditions (room temperature to 70°C), making it highly suitable for large-scale manufacturing.
Q: What types of substituents are tolerated on the isatin ring?
A: The reaction demonstrates broad substrate scope, tolerating electron-withdrawing groups like halogens and nitro groups, as well as electron-donating groups like methoxy and alkyl chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-2-Oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to plant. We are committed to delivering high-purity 3-hydroxy-2-oxindole derivatives that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced base-catalyzed technologies, we provide a competitive edge in the synthesis of complex pharmaceutical intermediates.
We invite you to collaborate with us to explore how this innovative synthesis can optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your journey from discovery to market.
