Advanced Base-Catalyzed Synthesis of 3-Hydroxy-2-Oxindole Intermediates for Commercial Scale-Up
Advanced Base-Catalyzed Synthesis of 3-Hydroxy-2-Oxindole Intermediates for Commercial Scale-Up
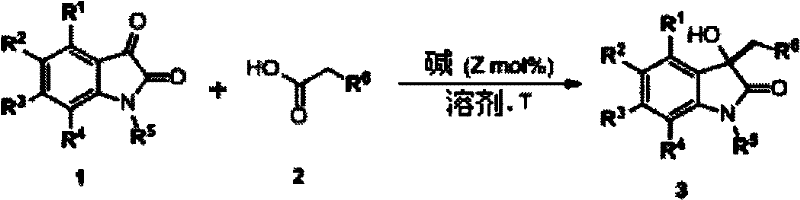
The pharmaceutical industry constantly seeks robust, scalable methodologies for constructing privileged scaffolds, and the 3-hydroxy-2-oxindole core remains a cornerstone in medicinal chemistry due to its presence in numerous bioactive natural products. Patent CN102659662A introduces a transformative synthetic approach that utilizes a simple base-catalyzed condensation between isatin and alpha-substituted acetic acids to generate these valuable intermediates. This technology represents a significant leap forward in process chemistry, moving away from complex metal-mediated transformations toward simpler, more sustainable organocatalytic processes. By leveraging readily available starting materials and mild reaction conditions, this method addresses critical pain points in the supply chain for high-purity pharmaceutical intermediates. The versatility of this route allows for the introduction of diverse functional groups at the C3 position, enabling the rapid assembly of compound libraries for drug discovery programs. Furthermore, the operational simplicity of the protocol makes it an ideal candidate for reliable pharmaceutical intermediate supplier networks aiming to reduce manufacturing complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3-hydroxy-2-oxindole skeleton has relied heavily on methods that pose significant challenges for large-scale industrial application. Traditional approaches often involve the use of stoichiometric amounts of organometallic reagents or transition metal catalysts, which introduce severe complications regarding heavy metal residue removal and regulatory compliance. Many prior art methods require cryogenic temperatures or strictly anhydrous conditions, driving up energy costs and necessitating specialized equipment that limits scalability. Additionally, the substrate scope in older methodologies is frequently narrow, failing to tolerate sensitive functional groups such as nitro or halo substituents without side reactions. These limitations result in lower overall yields and increased waste generation, creating bottlenecks in the production of key drug candidates. The reliance on expensive or difficult-to-synthesize nucleophiles further exacerbates the cost burden, making these conventional routes economically unviable for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes a direct, base-catalyzed addition-decarboxylation sequence that operates under remarkably mild conditions. By employing simple organic or inorganic bases such as triethylamine, DBU, or even sodium acetate, the reaction proceeds efficiently in common solvents like DMF or THF at temperatures ranging from room temperature to 70°C. This novel approach eliminates the need for toxic heavy metals, thereby simplifying the purification process and ensuring the final product meets stringent purity specifications required for API production. The reaction demonstrates exceptional functional group tolerance, successfully accommodating electron-withdrawing and electron-donating groups on the isatin ring without compromising yield. Moreover, the use of cheap, commodity chemicals like cyanoacetic acid as the nucleophile source drastically lowers the raw material costs. This shift towards organocatalysis not only enhances safety profiles by avoiding pyrophoric reagents but also aligns with green chemistry principles, making it a superior choice for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Base-Catalyzed Decarboxylative Addition
The core of this synthetic innovation lies in a clever mechanistic pathway involving 1,2-addition followed by spontaneous decarboxylation. Initially, the base catalyst deprotonates the alpha-substituted acetic acid to generate a reactive enolate species in situ. This nucleophilic enolate then attacks the electrophilic C3 carbonyl carbon of the isatin substrate, forming a tetrahedral intermediate. Unlike traditional aldol reactions that stop at the beta-hydroxy carbonyl stage, this system undergoes a subsequent intramolecular proton transfer and decarboxylation step. The loss of carbon dioxide drives the equilibrium forward, resulting in the formation of the stable 3-substituted-3-hydroxy-2-oxindole product. This decarboxylative drive is crucial as it renders the reaction essentially irreversible, pushing conversions to near completion even with catalytic amounts of base. The mechanism avoids the formation of stable byproducts that are common in non-decarboxylative pathways, thus enhancing the overall atom economy of the process.
Furthermore, the mechanistic flexibility allows for the introduction of chirality through the use of chiral base catalysts. The patent highlights the use of cinchona alkaloid derivatives and other chiral amines which can induce asymmetry during the nucleophilic attack phase. When chiral bases are employed, the enolate approaches the isatin carbonyl from a specific face, leading to the formation of enantiomerically enriched products with high ee values. This capability is vital for the synthesis of chiral drugs where specific stereochemistry dictates biological activity. The ability to tune the steric and electronic properties of the base catalyst provides a powerful handle for optimizing both reaction rate and stereoselectivity. Understanding this mechanism allows process chemists to fine-tune reaction parameters such as solvent polarity and temperature to maximize efficiency while minimizing impurity profiles, ensuring a robust and reproducible manufacturing process.
How to Synthesize 3-Cyanomethyl-3-hydroxyoxindole Efficiently
The practical implementation of this chemistry is straightforward and requires minimal specialized training, making it accessible for most synthetic laboratories. The general procedure involves dissolving the isatin substrate in a polar aprotic solvent, followed by the sequential addition of the alpha-substituted acetic acid and the chosen base catalyst. For standard substrates, heating the mixture to 70°C accelerates the reaction kinetics, although many examples proceed well at room temperature given sufficient time. Reaction progress is easily monitored by TLC, and upon completion, a simple aqueous workup involving extraction with ethyl acetate suffices to isolate the crude product. Final purification is typically achieved via flash column chromatography, yielding the target molecule as a high-purity solid. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures tailored to different substrates.
- Dissolve isatin substrate in a polar aprotic solvent such as DMF or THF at room temperature.
- Add alpha-substituted acetic acid (e.g., cyanoacetic acid) and a catalytic amount of organic or inorganic base.
- Heat the mixture to 70°C or maintain at room temperature until completion, then purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial strategic advantages by decoupling production from volatile supply chains associated with precious metal catalysts. The reliance on commodity chemicals like isatin and cyanoacetic acid ensures a stable and predictable supply of raw materials, mitigating the risk of production delays caused by sourcing bottlenecks. Since the reagents are widely available from multiple global suppliers, procurement managers can leverage competitive bidding to optimize costs further. The elimination of expensive ligands and metal salts removes a significant line item from the bill of materials, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. Additionally, the mild reaction conditions reduce energy consumption compared to processes requiring cryogenic cooling or high-pressure reactors, lowering the overall utility costs associated with production.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of costly transition metal catalysts with inexpensive organic bases. Traditional methods often require palladium, rhodium, or copper complexes along with specialized ligands, which represent a significant portion of the raw material cost. By replacing these with amines or carbonates, the direct material cost is drastically simplified. Furthermore, the absence of heavy metals simplifies the downstream processing; there is no need for expensive scavenging resins or complex filtration steps to meet residual metal limits. This reduction in processing steps translates to lower labor costs and higher throughput in the manufacturing facility. The high yields reported across various substrates mean less raw material is wasted, improving the overall mass balance and reducing the cost per kilogram of the final active ingredient.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved because the key reagents are bulk commodities with established global production capacities. Isatin and its derivatives are produced on a multi-ton scale for the dye and pharmaceutical industries, ensuring that supply disruptions are unlikely. Similarly, bases like triethylamine and solvents like DMF are standard inventory items for any chemical distributor. This universality means that switching suppliers in the event of a shortage is seamless, unlike specialized chiral ligands which may have single-source dependencies. The robustness of the reaction to moisture and oxygen, as indicated by the lack of strict inert atmosphere requirements in many examples, further reduces logistical complexities. Materials do not require special shipping conditions such as dry ice or nitrogen blanketing, simplifying transportation and storage logistics.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the exothermic nature of the reaction being manageable under the described conditions. The use of common solvents allows for easy recovery and recycling, aligning with environmental sustainability goals. The absence of toxic heavy metal waste streams simplifies wastewater treatment and disposal, reducing the environmental compliance burden on the manufacturing site. This 'green' profile is increasingly important for meeting the rigorous audit standards of major pharmaceutical companies. The process generates minimal hazardous waste, primarily consisting of aqueous salt solutions and organic solvents that can be treated using standard protocols. This ease of waste management accelerates the regulatory approval process for new drug applications, as the impurity profile is cleaner and easier to characterize.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this base-catalyzed synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy for process development teams. Understanding these nuances is critical for evaluating the feasibility of this route for specific target molecules. The data supports the versatility of the method across a wide range of electronic and steric environments.
Q: What are the primary advantages of this base-catalyzed method over traditional metal-catalyzed routes?
A: This method eliminates the need for expensive transition metal catalysts and harsh reaction conditions, utilizing cheap, commercially available bases like triethylamine or DBU, which significantly simplifies downstream purification and reduces heavy metal contamination risks.
Q: Can this synthetic route be adapted for enantioselective synthesis?
A: Yes, the patent demonstrates that chiral bases such as quinine derivatives or cinchona alkaloids can be employed to achieve high enantiomeric excess (ee), making it suitable for synthesizing chiral drug intermediates.
Q: What is the substrate scope for the isatin component in this reaction?
A: The process exhibits broad substrate tolerance, successfully accommodating various substituents on the isatin ring including halogens (F, Cl, Br), nitro groups, alkoxy groups, and alkyl chains, maintaining high yields across diverse electronic environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-2-Oxindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the lab bench to the manufacturing plant. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, utilizing rigorous QC labs to verify every batch against identity and potency standards. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing you with a secure supply chain partner you can trust. By adopting advanced technologies like the base-catalyzed decarboxylative addition described here, we help our clients optimize their manufacturing costs without compromising on quality.
We invite you to contact our technical procurement team to discuss how we can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for an existing route or need to explore the feasibility of this novel synthesis for a new target, we are ready to assist. Please reach out to request specific COA data and route feasibility assessments tailored to your molecule. Our goal is to be your long-term partner in bringing life-saving medicines to market faster and more economically.
