Optimizing Citalopram Purity: A Technical Analysis of Novel Cyanide Exchange and Purification Strategies
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients (APIs) and their key intermediates, particularly for widely prescribed antidepressants like citalopram. Patent CN1282648C introduces a transformative approach to the preparation and purification of citalopram, specifically addressing the persistent challenge of removing desmethyl-citalopram derivatives that arise during the critical cyanide exchange reaction. This technical insight report analyzes the novel methodology disclosed in the patent, which combines a robust cyanide substitution strategy with a clever downstream purification technique involving selective acylation. By converting troublesome secondary amine impurities into easily separable amides, this process offers a viable pathway for manufacturers seeking to enhance product quality while streamlining production workflows. As a leading entity in the fine chemical sector, understanding these mechanistic nuances is essential for R&D directors and procurement specialists aiming to secure a reliable pharmaceutical intermediates supplier capable of delivering consistent, high-quality materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of citalopram has relied heavily on the conversion of 5-halo-phthalan derivatives into the corresponding nitriles, a transformation often achieved through Rosenmund-von Braun type reactions or modern palladium-catalyzed cyanations. However, a significant drawback inherent to these conventional pathways, as noted in prior art such as DE2657013 and various WO publications, is the concurrent formation of desmethyl-citalopram impurities. These impurities possess a secondary amine functionality rather than the desired tertiary dimethylamine group, resulting from demethylation side reactions or the use of imperfect precursors. The structural similarity between the target molecule and the desmethyl byproduct makes separation exceptionally difficult using standard crystallization or washing techniques. Consequently, manufacturers often face extensive and expensive purification procedures, including multiple recrystallizations or preparative chromatography, which drastically reduce overall yield and increase the cost of goods sold. Furthermore, the persistence of these genotoxic or pharmacologically active impurities poses a regulatory risk, necessitating a more robust solution for impurity control.
The Novel Approach
The methodology outlined in CN1282648C presents a strategic departure from traditional purification reliance by introducing a chemical derivatization step specifically designed to alter the physicochemical properties of the impurity. Instead of attempting to separate the free base of the desmethyl impurity directly from citalopram, the process treats the crude reaction mixture with an amide-forming agent, such as acetic anhydride or acetyl chloride. This reagent selectively reacts with the secondary amine of the desmethyl-citalopram to form a neutral amide derivative. Crucially, this chemical modification renders the impurity non-basic, meaning it no longer responds to acid-base extraction in the same manner as the tertiary amine citalopram. This fundamental shift in chemical behavior allows for a clean separation where the desired product can be extracted into an aqueous acidic phase while the derivatized impurity remains in the organic waste phase. This approach not only simplifies the workflow but also significantly enhances the final purity profile without the need for complex chromatographic interventions.
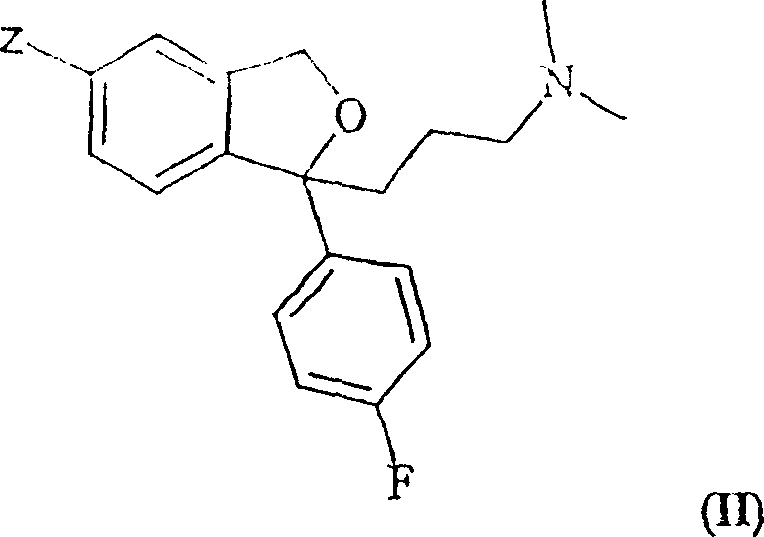
Mechanistic Insights into Cyanide Exchange and Selective Acylation
The core of this synthesis begins with the cyanide exchange reaction involving a compound of Formula II, where the leaving group Z can be iodine, bromine, chlorine, or a triflate-like group such as CF3-(CF2)n-SO2-O-. In a typical embodiment, the 5-bromo derivative is reacted with a cyanide source, such as copper(I) cyanide, in a high-boiling polar solvent like sulfolane at elevated temperatures around 150°C. This step generates the crude citalopram base, but inevitably co-produces the desmethyl impurity due to thermal degradation or precursor quality issues. The innovation lies in the subsequent treatment of this crude oil. By introducing an acylating agent, specifically acetic anhydride, the nucleophilic secondary nitrogen of the desmethyl impurity attacks the carbonyl carbon of the anhydride. This reaction proceeds efficiently even at moderate temperatures, such as 60°C, converting the impurity into an N-acetyl derivative. This derivatization is chemically orthogonal to the tertiary amine of the citalopram molecule, which remains unreacted under these conditions due to steric hindrance and lower nucleophilicity compared to the secondary amine.
Following the acylation step, the separation mechanism leverages the difference in basicity between the product and the derivatized impurity. The reaction mixture is subjected to an acid wash, typically using hydrochloric acid, which protonates the tertiary amine of the citalopram, rendering it water-soluble as a hydrochloride salt. Conversely, the newly formed amide impurity lacks basic character and remains dissolved in the organic solvent, such as toluene. By separating the aqueous phase, the manufacturer effectively discards the bulk of the organic-soluble impurities. The aqueous phase is then basified using ammonia or sodium hydroxide to regenerate the free base of the citalopram, which is subsequently extracted back into an organic solvent. This acid-base cycle acts as a powerful purification gate, ensuring that the final crystallized product meets stringent purity specifications, often exceeding 99.7% purity as confirmed by HPLC analysis. This mechanistic elegance ensures that cost reduction in pharmaceutical intermediates manufacturing is achieved through chemical intelligence rather than brute-force purification.
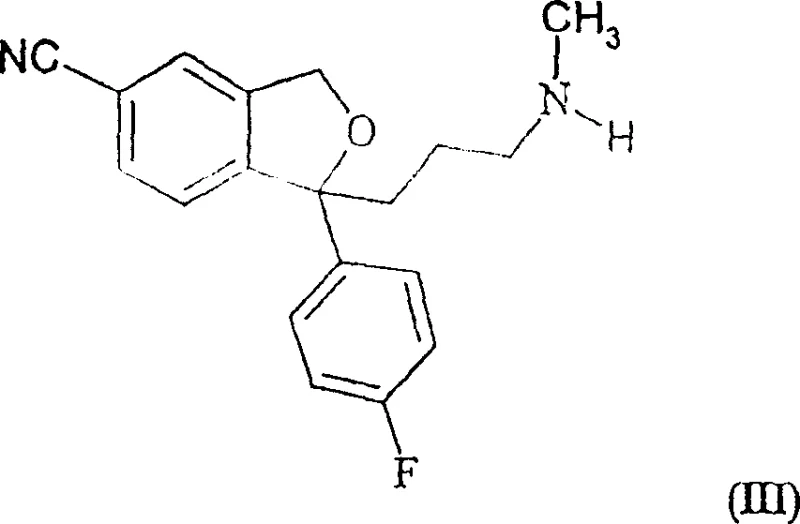
How to Synthesize Citalopram Efficiently
The synthesis of high-purity citalopram via this patented route involves a sequence of well-defined unit operations that balance reaction kinetics with separation efficiency. The process initiates with the cyanation of the phthalan precursor, followed by a workup to remove metal salts, and culminates in the selective acylation and extraction protocol described above. This methodology is particularly advantageous for commercial scale-up of complex pharmaceutical intermediates because it avoids the use of specialized chromatography resins and minimizes solvent consumption associated with repeated recrystallizations. The following guide outlines the critical operational phases required to implement this technology effectively in a GMP-compliant environment.
- Perform cyanide exchange on the 5-halo-phthalan precursor (Formula II) using copper(I) cyanide or palladium-catalyzed conditions to generate crude citalopram.
- Treat the crude reaction mixture containing desmethyl-citalopram impurities with an acylating agent such as acetic anhydride to convert the secondary amine impurity into a neutral amide.
- Execute an acid-base extraction sequence where the basic citalopram is moved to the aqueous phase while the neutral amide impurity remains in the organic waste phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates into tangible operational efficiencies and risk mitigation. The primary advantage lies in the drastic simplification of the downstream processing train. By chemically altering the impurity profile to facilitate easy removal, the process eliminates the need for multiple, yield-eroding recrystallization steps that are characteristic of older methods. This streamlining leads to substantial cost savings in terms of solvent usage, energy consumption for heating and cooling cycles, and labor hours required for monitoring and handling. Furthermore, the robustness of the acid-base extraction method ensures high recovery rates of the valuable API intermediate, maximizing the return on raw material investment. From a supply continuity perspective, the reliance on commodity chemicals like acetic anhydride and standard solvents like toluene ensures that the supply chain is not vulnerable to shortages of exotic reagents.
- Cost Reduction in Manufacturing: The elimination of extensive chromatographic purification and the reduction in recrystallization cycles directly lower the manufacturing cost per kilogram. By converting a difficult-to-remove impurity into a waste product that partitions cleanly into the organic phase, the process minimizes product loss typically associated with aggressive purification attempts. This efficiency gain allows for a more competitive pricing structure for the final citalopram hydrobromide or hydrochloride salts, providing a distinct economic advantage in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The use of stable, commercially available reagents such as copper cyanide and acetic anhydride ensures that production schedules are not disrupted by reagent scarcity. Additionally, the process tolerance for varying levels of crude impurity means that upstream synthesis variations can be corrected downstream without rejecting entire batches. This flexibility enhances the overall reliability of the supply chain, ensuring that delivery commitments to downstream API manufacturers are met consistently without unexpected delays caused by failed quality control tests.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard reactor vessels and separation equipment found in most multipurpose chemical plants. The waste streams generated are primarily organic solvents and aqueous salt solutions, which are manageable through standard wastewater treatment protocols. By avoiding the generation of heavy metal-laden solid wastes often associated with alternative purification methods, the process aligns better with modern environmental compliance standards, reducing the regulatory burden and disposal costs associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and claims presented in the patent literature, offering clarity on the feasibility and performance of the method. Understanding these details is crucial for technical teams evaluating the integration of this process into their existing manufacturing portfolios.
Q: How does this process remove desmethyl-citalopram impurities?
A: The process utilizes a selective acylation step where acetic anhydride reacts with the secondary amine group of the desmethyl impurity to form a neutral amide. Unlike the tertiary amine of citalopram, this amide does not form a salt with acid, allowing it to be separated via acid-base extraction.
Q: What purity levels can be achieved with this method?
A: According to the patent data, this method allows for the production of citalopram salts with purity exceeding 99.7%, and most preferably exceeding 99.8%, effectively meeting stringent pharmacopeial standards.
Q: Is this method scalable for commercial manufacturing?
A: Yes, the method relies on standard unit operations such as heating, liquid-liquid extraction, and crystallization, which are highly scalable and do not require exotic chromatography or complex filtration equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citalopram Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and process robustness in the production of antidepressant intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of citalopram intermediate meets the highest international standards, including the >99.7% purity benchmarks enabled by advanced purification techniques like the one analyzed here. Our commitment to quality assurance ensures that our partners receive materials that facilitate smooth regulatory filings and consistent drug product performance.
We invite global pharmaceutical partners to collaborate with us to leverage these advanced manufacturing capabilities. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential clients to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain and reduce time-to-market for your final drug products. Let us be your trusted partner in delivering high-quality chemical solutions.
