Advanced Isoxazoline Manufacturing via Metal-Free Lewis Acid Catalysis for Global Supply Chains
The pharmaceutical and agrochemical industries continuously seek robust synthetic routes for heterocyclic scaffolds like isoxazolines, which serve as critical building blocks in drug discovery. Patent CN112028848A introduces a groundbreaking method for preparing isoxazoline derivatives through a three-component reaction involving olefins, diazo compounds, and tert-butyl nitrite. Unlike traditional approaches that often rely on harsh conditions or precious metal catalysts, this innovation employs a Lewis acid catalyst to drive the cycloaddition efficiently at mild temperatures ranging from 25 to 50°C. This technological leap not only enhances reaction yields but also significantly simplifies the operational complexity associated with synthesizing these valuable intermediates. By enabling the reaction to proceed in air without the need for inert atmosphere protection, the process offers a safer and more economically viable pathway for manufacturers aiming to scale production. 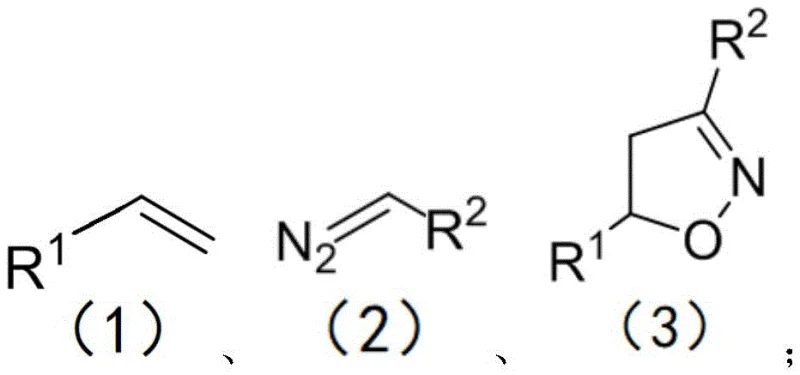
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of isoxazoline rings has been plagued by significant technical hurdles that hinder efficient commercial manufacturing. Conventional methodologies, such as those reported by research groups utilizing copper nitrate or other transition metals, often necessitate strict inert atmosphere conditions like nitrogen protection to achieve acceptable yields. These requirements impose substantial burdens on production facilities, demanding specialized equipment and rigorous safety protocols that escalate operational costs. Furthermore, the reliance on stoichiometric amounts of transition metals introduces complex downstream purification challenges, as removing trace metal residues to meet pharmaceutical standards can be both time-consuming and expensive. Additionally, many existing routes require the pre-preparation of unstable intermediates or employ harsh reaction conditions that limit substrate scope and pose safety risks during scale-up operations.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this landscape by leveraging a Lewis acid-catalyzed three-component coupling that operates under remarkably mild conditions. By utilizing catalysts such as boron trifluoride etherate, the reaction avoids the use of transition metals entirely, thereby eliminating the risk of heavy metal contamination in the final product. This method allows the synthesis to be conducted in air at temperatures as low as room temperature, drastically reducing energy consumption and equipment requirements. The direct use of commercially available olefins and diazo compounds without extensive pre-functionalization streamlines the supply chain and reduces raw material costs. Consequently, this approach provides a versatile platform that accommodates a wide range of substrates while maintaining high efficiency and selectivity, making it an ideal candidate for modern green chemistry applications in fine chemical manufacturing.
Mechanistic Insights into Lewis Acid-Catalyzed Cycloaddition
The core of this synthetic breakthrough lies in the precise activation of reactants by the Lewis acid catalyst, which facilitates the generation of reactive nitrile oxide intermediates in situ. The mechanism involves the interaction of the diazo compound with tert-butyl nitrite under the influence of the Lewis acid, leading to the formation of a key nitrile oxide species without the need for external oxidants or metal promoters. This intermediate then undergoes a concerted 1,3-dipolar cycloaddition with the olefin substrate to construct the isoxazoline ring system with high regioselectivity. The mild nature of the Lewis acid ensures that sensitive functional groups on the substrates remain intact, preserving the chemical integrity required for subsequent derivatization steps in drug synthesis. 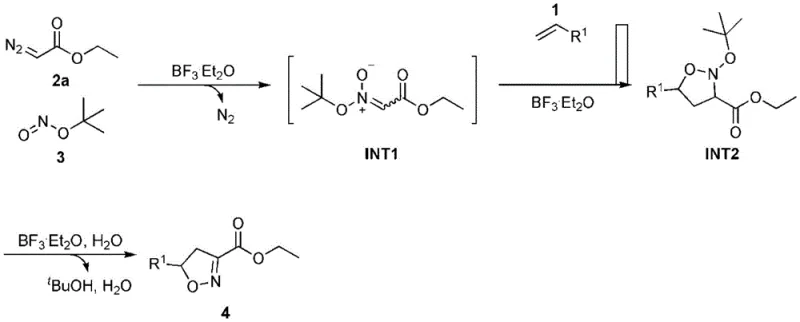
From an impurity control perspective, the absence of transition metals fundamentally alters the impurity profile of the resulting isoxazoline products. Traditional metal-catalyzed routes often generate metal-complexed byproducts or require aggressive chelating agents for purification, which can introduce new contaminants. In contrast, this Lewis acid-mediated process generates primarily organic byproducts that are easier to separate through standard extraction and chromatography techniques. The high atom economy of the three-component reaction minimizes waste generation, aligning with stringent environmental regulations and sustainability goals. Furthermore, the stability of the reaction system in air reduces the formation of oxidation-related impurities that might occur under more forcing conditions, ensuring a cleaner crude product that requires less intensive purification efforts before reaching commercial specifications.
How to Synthesize Isoxazoline Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent ratios and mixing protocols to maximize yield and purity. The process begins with the dissolution of the olefin and diazo compound in a suitable organic solvent such as acetonitrile, followed by the controlled addition of tert-butyl nitrite and the Lewis acid catalyst. Maintaining the reaction temperature within the optimal range of 25 to 50°C is crucial for balancing reaction rate and selectivity, while the exposure to air simplifies the setup compared to glovebox techniques. Detailed standardized synthesis steps see the guide below.
- Combine olefin, diazo compound, and tert-butyl nitrite in an organic solvent such as acetonitrile within a reaction vessel.
- Add a Lewis acid catalyst like boron trifluoride etherate and maintain the mixture at 25-50°C in air for 12-24 hours.
- Quench the reaction with saturated sodium chloride solution, extract with ethyl acetate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this technology presents a compelling value proposition by addressing key pain points related to cost, availability, and regulatory compliance. The elimination of expensive transition metal catalysts directly reduces the bill of materials, while the simplified workup procedure lowers labor and utility costs associated with purification. The ability to operate in air removes the need for costly inert gas infrastructure, allowing for more flexible manufacturing scheduling and reduced downtime. Moreover, the use of readily available starting materials mitigates supply chain risks associated with specialized reagents, ensuring consistent production continuity even during market fluctuations.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts eliminates the need for expensive metal scavengers and complex filtration steps, leading to substantial cost savings in downstream processing. By avoiding precious metals, the process reduces the overall raw material expenditure and minimizes the financial burden of waste disposal associated with hazardous metal residues. The mild reaction conditions also translate to lower energy consumption for heating or cooling, further enhancing the economic efficiency of the manufacturing process. These cumulative factors contribute to a more competitive cost structure for isoxazoline intermediates in the global market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as olefins and tert-butyl nitrite ensures a stable supply of raw materials, reducing dependency on niche suppliers with limited capacity. The robustness of the reaction in air means that production is less susceptible to disruptions caused by gas supply issues or equipment failures related to inert atmosphere systems. This reliability allows for more accurate forecasting and inventory management, enabling companies to meet delivery commitments with greater confidence. Additionally, the scalability of the process supports rapid ramp-up capabilities to respond to sudden increases in demand without significant capital investment.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, including high atom economy and reduced hazardous waste, facilitate easier compliance with increasingly strict environmental regulations. The absence of heavy metals simplifies the treatment of effluent streams, lowering the cost and complexity of wastewater management systems. The mild operating conditions enhance process safety, reducing the risk of thermal runaways and making the technology suitable for large-scale continuous flow manufacturing. These features position the process as a sustainable choice for long-term production strategies aligned with corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isoxazoline synthesis technology. These answers are derived directly from the patent specifications and experimental data to provide accurate guidance for potential adopters. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: Does this synthesis method require transition metal catalysts?
A: No, this patented method utilizes Lewis acid catalysts instead of expensive and potentially toxic transition metals, simplifying purification and reducing heavy metal residue risks.
Q: What are the typical reaction conditions for this isoxazoline preparation?
A: The reaction proceeds efficiently under mild conditions, typically between 25-50°C in air, eliminating the need for inert gas protection or extreme temperatures.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of readily available raw materials, mild operating conditions, and simple workup procedures makes this method highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and compliant synthetic routes for critical pharmaceutical intermediates like isoxazolines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest quality standards required by global regulatory bodies. Our commitment to technical excellence allows us to offer customized solutions that optimize both performance and cost for our partners.
We invite you to collaborate with us to leverage this advanced Lewis acid-catalyzed technology for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain objectives and drive value for your organization.
