Advanced Iron-Catalyzed Synthesis of N-Butylphthalide for Commercial Scale-Up
Advanced Iron-Catalyzed Synthesis of N-Butylphthalide for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and economically viable synthetic routes for critical neuroprotective agents such as n-butylphthalide (NBP). A recent technological breakthrough documented in patent CN116253705A introduces a novel methodology that fundamentally shifts the production paradigm from expensive precious metal catalysis to abundant transition metal systems. This innovation is particularly significant for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials without the volatility associated with palladium or platinum markets. The disclosed method utilizes a sophisticated catalytic system comprising ferric nitrate nonahydrate, 2,2,6,6-tetramethylpiperidine oxide (TEMPO), and potassium chloride to effect an oxidative cyclization under an oxygen atmosphere. By leveraging molecular oxygen as the terminal oxidant, this process not only aligns with green chemistry principles but also drastically simplifies the downstream processing requirements. For R&D directors and procurement strategists, this patent represents a tangible opportunity to optimize the cost structure of API manufacturing while maintaining stringent quality standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of n-butylphthalide has relied heavily on methodologies that present significant bottlenecks for large-scale commercialization and cost reduction in pharmaceutical intermediate manufacturing. Traditional routes often employ phthalic anhydride or phthalic acid derivatives which require high-temperature reactions followed by catalytic hydrogenation using palladium on carbon (Pd/C). These legacy processes are inherently capital intensive due to the requirement for high-pressure hydrogenation equipment and the substantial cost of precious metal catalysts. Furthermore, the use of stoichiometric oxidants or harsh acidic conditions in alternative pathways, such as those involving titanium superoxide or tert-butyl peroxide, introduces severe safety hazards and generates substantial chemical waste. The necessity to remove trace heavy metals from the final product to meet regulatory compliance adds additional purification steps, extending the production cycle and increasing the overall lead time for high-purity pharmaceutical intermediates. Consequently, these factors collectively inflate the cost of goods sold (COGS) and create supply chain vulnerabilities dependent on the availability of specific noble metals.
The Novel Approach
In stark contrast to these conventional limitations, the novel approach detailed in the patent data utilizes a mild, iron-catalyzed oxidative cyclization that operates efficiently at near-ambient temperatures. This methodology replaces the expensive palladium catalysts with inexpensive and environmentally benign iron salts, specifically ferric nitrate nonahydrate, which are readily available in bulk quantities globally. The reaction proceeds through a diol intermediate, 1-[2-(hydroxymethyl)phenyl]-1-pentanol, which is smoothly converted to the target lactone using molecular oxygen as the sole oxidant. This shift eliminates the need for high-pressure hydrogenation reactors and reduces the risk profile associated with energetic oxidants. The operational simplicity of this new route allows for easier commercial scale-up of complex pharmaceutical intermediates, as the reaction conditions (25°C to 60°C) are easily manageable in standard glass-lined or stainless steel reactors. By removing the dependency on precious metals and hazardous reagents, this approach offers a sustainable pathway that enhances supply chain reliability and significantly reduces the environmental footprint of the manufacturing process.
Mechanistic Insights into Fe(NO3)3/TEMPO-Catalyzed Oxidative Cyclization
The core of this technological advancement lies in the synergistic interaction between the iron catalyst and the nitroxyl radical mediator within the oxidative cycle. The mechanism involves the activation of molecular oxygen by the iron species, which subsequently regenerates the active oxoammonium species from the TEMPO radical. This active species then selectively oxidizes the benzylic alcohol moiety of the intermediate diol to an aldehyde in situ. Once the aldehyde is formed, it undergoes a spontaneous hemiacetal formation with the adjacent hydroxymethyl group, followed by further oxidation to yield the stable lactone ring of n-butylphthalide. The presence of potassium chloride in the catalytic system plays a crucial role in modulating the electronic environment of the iron center, enhancing the turnover frequency and ensuring high conversion rates. This mechanistic pathway is highly selective, minimizing the formation of over-oxidized byproducts such as carboxylic acids or ring-opened degradation products that often plague harsher oxidation methods. Understanding this catalytic cycle is essential for process chemists aiming to replicate these results, as it highlights the importance of maintaining precise oxygen flow rates and catalyst loading to maximize efficiency.

Furthermore, the impurity control mechanism inherent in this mild oxidative system provides a distinct advantage for producing high-purity OLED material or pharmaceutical grades. The reaction temperature, typically maintained between 25°C and 60°C, is sufficiently low to prevent thermal degradation of the sensitive lactone ring or polymerization of the intermediate species. In conventional high-temperature processes, thermal stress often leads to the formation of complex impurity profiles that are difficult to separate via crystallization or chromatography. By operating under these温和 conditions, the new method ensures a cleaner reaction profile, which directly translates to reduced purification costs and higher overall yields. The use of toluene as the preferred solvent further aids in the solubility of the organic intermediates while facilitating the easy removal of water generated during the oxidation process. For quality control teams, this means that the resulting crude product requires less aggressive purification protocols, thereby preserving the integrity of the final active pharmaceutical ingredient and ensuring consistent batch-to-batch reproducibility.
How to Synthesize N-Butylphthalide Efficiently
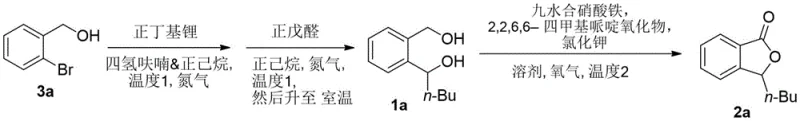
The synthesis of n-butylphthalide via this novel route is divided into two distinct operational stages that require careful control of stoichiometry and atmospheric conditions to ensure optimal outcomes. The first stage involves the generation of the key diol intermediate through a lithiation reaction, which must be conducted under strict anhydrous conditions to prevent quenching of the organolithium species. The second stage is the catalytic oxidation, where the introduction of oxygen and the precise ratio of the iron/TEMPO system are critical for driving the reaction to completion. While the patent provides specific experimental examples, scaling this process requires a thorough understanding of heat transfer and gas-liquid mass transfer dynamics, particularly during the oxygenation step. The detailed standardized synthesis steps见下方的指南 outline the precise addition rates, temperature ramps, and workup procedures necessary to achieve the reported yields safely and effectively.
- Perform lithiation of o-bromobenzyl alcohol with n-butyllithium at -25°C to -15°C, followed by addition of n-valeraldehyde to form the diol intermediate.
- Oxidize the intermediate using a catalytic system of ferric nitrate, TEMPO, and potassium chloride under an oxygen atmosphere in toluene.
- Purify the final n-butylphthalide product via flash column chromatography to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this iron-catalyzed synthesis route offers substantial cost savings and operational efficiencies that directly impact the bottom line of chemical manufacturing projects. The primary economic driver is the elimination of precious metal catalysts, which removes the volatility of palladium pricing from the cost model and eradicates the need for expensive metal recovery or scavenging units. Additionally, the use of molecular oxygen as an oxidant is vastly superior in terms of atom economy and cost compared to stoichiometric oxidants like peroxides or hypervalent iodine reagents. This transition not only lowers the direct material costs but also simplifies the waste treatment infrastructure, as the byproducts are primarily water and benign iron salts. For supply chain managers, the reliance on commodity chemicals such as iron nitrate and toluene ensures a stable supply base that is not subject to the geopolitical constraints often associated with rare earth or precious metal sourcing. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Cost Reduction in Manufacturing: The substitution of palladium catalysts with iron salts fundamentally alters the cost structure by removing high-value metal inputs and the associated purification costs required to meet residual metal specifications. This change allows for a drastic simplification of the downstream processing workflow, as there is no need for specialized filtration media or resin columns to trap heavy metals. Consequently, the overall consumption of solvents and auxiliary materials is reduced, leading to a leaner and more cost-effective manufacturing process. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to lower utility costs per kilogram of product produced. These cumulative efficiencies result in a significantly more competitive pricing model for the final n-butylphthalide product without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including o-bromobenzyl alcohol, n-valeraldehyde, and ferric nitrate, are commodity chemicals with robust global supply chains. This availability mitigates the risk of production stoppages due to raw material shortages, a common issue with specialized reagents or catalysts. The operational simplicity of the process also means that it can be easily transferred between different manufacturing sites or scaled up in existing facilities without requiring major capital investment in new equipment. This flexibility ensures that supply continuity can be maintained even in the face of regional disruptions or logistical challenges. For procurement managers, this translates to a lower risk profile and greater confidence in the ability to meet long-term supply agreements with key stakeholders.
- Scalability and Environmental Compliance: The use of oxygen as a green oxidant aligns perfectly with increasingly stringent environmental regulations regarding waste disposal and emissions. Unlike processes that generate heavy metal waste or toxic organic byproducts, this method produces minimal hazardous waste, simplifying the environmental compliance burden. The reaction's scalability is enhanced by the absence of exothermic hazards associated with strong oxidants, allowing for safer operation in large-scale reactors. This safety profile facilitates faster regulatory approvals and reduces the time required for process safety audits. Ultimately, the combination of environmental sustainability and operational safety makes this route highly attractive for long-term commercial production, ensuring that the manufacturing process remains viable and compliant for years to come.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed synthesis route for n-butylphthalide. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this new methodology for their specific production needs. The responses cover aspects ranging from catalyst performance to safety considerations, ensuring a comprehensive overview of the technology's value proposition.
Q: What are the advantages of using iron catalysts over palladium for NBP synthesis?
A: Iron catalysts significantly reduce raw material costs compared to precious metals like palladium and eliminate the need for expensive metal scavenging steps, simplifying the purification process.
Q: Is the oxidative cyclization step safe for industrial scale-up?
A: Yes, the reaction utilizes molecular oxygen as the terminal oxidant under mild temperatures (25°C to 60°C), avoiding the hazards associated with high-pressure hydrogenation or stoichiometric toxic oxidants.
Q: What is the expected purity profile for this synthetic route?
A: The mild reaction conditions minimize side reactions and decomposition, allowing for the production of high-purity n-butylphthalide suitable for pharmaceutical applications after standard chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Butylphthalide Supplier
The technological potential of this iron-catalyzed route underscores the importance of partnering with a CDMO expert who possesses the capability to translate complex laboratory patents into robust industrial processes. NINGBO INNO PHARMCHEM stands at the forefront of this transformation, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing reaction parameters such as catalyst loading and oxygen flow rates to ensure that the stringent purity specifications required for pharmaceutical applications are consistently met. With our rigorous QC labs and state-of-the-art manufacturing facilities, we are uniquely positioned to deliver high-purity pharmaceutical intermediates that adhere to the highest global quality standards. We understand the critical nature of supply chain continuity and are committed to providing a reliable supply of n-butylphthalide that supports your drug development and commercialization timelines.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain strategy. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this method for your projects. Our team is ready to provide the technical support and commercial flexibility needed to optimize your production costs and enhance your market competitiveness. Let us collaborate to engineer a more efficient and sustainable future for your chemical manufacturing needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
