Revolutionizing Quinazolinone Production: Scalable Pd-Catalyzed Synthesis for High-Purity Pharmaceutical Intermediates
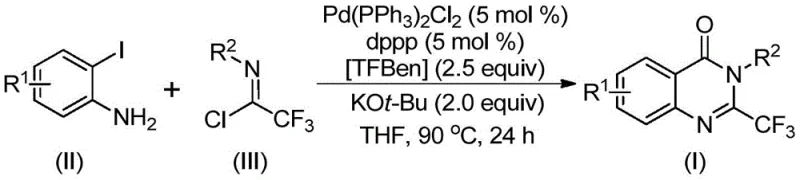
The patent CN112125856A introduces a groundbreaking synthetic methodology for producing 2-trifluoromethyl-substituted quinazolinone derivatives, a critical structural motif in numerous pharmaceutical compounds including anticonvulsants and antitumor agents. This innovative process addresses long-standing limitations in heterocyclic chemistry by employing a palladium-catalyzed carbonylation strategy that utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide surrogate, thereby eliminating the need for handling toxic gaseous CO. The methodology demonstrates exceptional versatility across diverse substrate classes while maintaining operational simplicity and high yields, representing a significant advancement in the manufacturing of fluorinated heterocyclic compounds essential for modern drug development pipelines. This technical breakthrough directly responds to industry demands for safer, more sustainable synthetic routes that maintain stringent quality requirements without compromising efficiency or scalability in pharmaceutical intermediate production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for 2-trifluoromethyl quinazolinones suffer from multiple critical deficiencies that hinder commercial adoption, including harsh reaction conditions requiring high-pressure carbon monoxide systems that pose significant safety hazards and necessitate specialized infrastructure. Existing methodologies such as cyclization of anthranilamide with trifluoroacetic anhydride or T3P-promoted tandem reactions often demand expensive pre-activated substrates and exhibit narrow substrate scope with limited functional group tolerance, resulting in inconsistent yields across different molecular architectures. Furthermore, these processes frequently generate complex impurity profiles that complicate purification and compromise final product quality, while the requirement for toxic gas handling creates substantial regulatory and operational burdens that increase both capital expenditure and production costs. The cumulative effect of these limitations has restricted the commercial viability of quinazolinone-based pharmaceutical intermediates despite their significant therapeutic potential.
The Novel Approach
The patented methodology overcomes these challenges through an elegant palladium-catalyzed carbonylation process that employs TFBen as a solid carbon monoxide equivalent, completely eliminating the need for hazardous gas handling while maintaining high reaction efficiency. This innovative approach utilizes readily available starting materials—o-iodoaniline derivatives and trifluoroethyl imidoyl chlorides—that are both cost-effective and structurally versatile, enabling the synthesis of diverse quinazolinone derivatives through simple substrate modification. The reaction proceeds under mild conditions (90°C in THF) with excellent functional group compatibility, accommodating halogens, alkyl groups, nitro substituents, and various aryl moieties without requiring pre-activation or specialized handling procedures. Crucially, the process delivers consistently high yields across a broad substrate range while generating minimal byproducts, significantly simplifying downstream purification and ensuring superior product quality suitable for pharmaceutical applications.
Mechanistic Insights into Pd-Catalyzed Carbonylation with TFBen
The catalytic cycle begins with potassium tert-butoxide-mediated intermolecular carbon-nitrogen bond coupling between o-iodoaniline and trifluoroethyl imidoyl chloride to form a trifluoroacetamidine intermediate. Subsequent oxidative addition of the palladium(0) catalyst into the carbon-iodine bond generates a key arylpalladium(II) species that undergoes CO insertion facilitated by thermal decomposition of TFBen at elevated temperatures. This critical step releases carbon monoxide in situ, which inserts into the carbon-palladium bond to form an acylpalladium intermediate that subsequently cyclizes through nucleophilic attack by the amine nitrogen. The resulting seven-membered palladacycle undergoes reductive elimination to release the final quinazolinone product while regenerating the active palladium catalyst for subsequent cycles. This mechanism elegantly avoids the need for external CO supply while maintaining precise control over the carbonylation step through the controlled release of CO from the solid surrogate.
Impurity control is achieved through multiple synergistic factors inherent to this catalytic system: the mild reaction conditions (90°C) prevent thermal decomposition pathways common in traditional high-temperature syntheses; the homogeneous nature of the palladium catalyst system minimizes side reactions associated with heterogeneous catalysts; and the precise stoichiometric control of TFBen ensures complete conversion without over-carbonylation byproducts. The use of potassium tert-butoxide as base promotes clean amide formation while suppressing competing hydrolysis pathways that could generate carboxylic acid impurities. Furthermore, the well-defined catalytic cycle prevents palladium black formation through continuous regeneration of active species, eliminating metal-derived impurities that typically complicate purification in alternative methodologies. This comprehensive impurity management strategy delivers products with exceptional purity profiles suitable for direct use in pharmaceutical synthesis without extensive additional purification steps.
How to Synthesize TFQ Derivatives Efficiently
This patented synthetic route represents a significant advancement in the manufacturing of fluorinated heterocyclic compounds through its innovative use of solid carbon monoxide surrogates and optimized palladium catalysis. The methodology provides pharmaceutical manufacturers with a reliable pathway to produce high-purity quinazolinone derivatives under safe operating conditions while maintaining excellent yield and selectivity across diverse substrate classes. Detailed standardized synthesis procedures have been developed to ensure consistent product quality at commercial scale, with specific protocols tailored to different derivative types based on their structural characteristics. The following section outlines the essential operational parameters and quality control checkpoints required for successful implementation of this technology in GMP manufacturing environments.
- Prepare reaction mixture with precise molar ratios of o-iodoaniline, trifluoroethyl imidoyl chloride, and TFBen in anhydrous THF under inert atmosphere
- Introduce palladium catalyst system comprising Pd(PPh3)2Cl2 and dppp ligand with potassium tert-butoxide base at controlled stoichiometry
- Execute carbonylation reaction at 90°C for optimized duration with continuous monitoring to ensure complete conversion and minimal byproduct formation
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process delivers substantial strategic advantages for procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate sourcing. The elimination of hazardous carbon monoxide handling requirements significantly reduces facility qualification costs and regulatory compliance burdens, while the use of readily available starting materials enhances supply chain resilience through multiple sourcing options. The simplified reaction workup and purification procedures decrease production cycle times without compromising quality standards, enabling more responsive order fulfillment to meet dynamic market demands. These operational improvements collectively strengthen the commercial viability of quinazolinone-based pharmaceutical development programs by providing reliable access to high-quality intermediates through a sustainable manufacturing platform.
- Cost Reduction in Manufacturing: The substitution of toxic gaseous carbon monoxide with solid TFBen eliminates expensive gas handling infrastructure and safety protocols while reducing waste treatment costs associated with metal scavenging operations required in alternative methodologies. The simplified purification process through standard column chromatography avoids costly specialized separation techniques, and the use of commercially available starting materials at favorable price points contributes to overall cost optimization without requiring expensive reagents or specialized equipment.
- Enhanced Supply Chain Reliability: The broad substrate scope enables flexible production scheduling based on raw material availability, while the use of stable solid reagents instead of hazardous gases improves logistics reliability through simplified transportation and storage requirements. Multiple sourcing options exist for all key starting materials including o-iodoaniline derivatives and trifluoroethyl imidoyl chlorides, which are commercially available from numerous global suppliers with established quality systems. This redundancy significantly reduces supply chain vulnerability compared to processes dependent on single-source specialty chemicals.
- Scalability and Environmental Compliance: The homogeneous reaction system demonstrates excellent scalability from laboratory to commercial production without requiring process re-engineering, with consistent performance maintained across different batch sizes due to the absence of gas-liquid mass transfer limitations. The elimination of toxic gas usage substantially reduces environmental compliance burdens while generating fewer hazardous byproducts compared to traditional methods, aligning with increasingly stringent global environmental regulations. The simplified waste stream composition facilitates more efficient treatment processes that reduce overall environmental impact while maintaining high product quality standards.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations based on detailed analysis of the patented methodology and its implementation in pharmaceutical manufacturing environments. These insights reflect practical experience from process development studies and scale-up trials conducted under GMP conditions.
Q: How does this method eliminate safety risks associated with traditional carbonylation processes?
A: The patented process replaces toxic gaseous carbon monoxide with solid TFBen (1,3,5-tricarboxylate phenol ester) as a carbon monoxide surrogate, completely eliminating handling hazards while maintaining reaction efficiency. This innovation addresses critical safety concerns in conventional methods that require specialized equipment for CO management.
Q: What substrate scope advantages does this methodology offer for pharmaceutical development?
A: The process demonstrates exceptional substrate tolerance across diverse functional groups including halogens, alkyl, nitro, and aryl substituents at various positions. This broad applicability enables rapid synthesis of structurally varied quinazolinone derivatives essential for drug discovery pipelines without requiring pre-activated substrates.
Q: How does the elimination of transition metal removal steps impact commercial manufacturing costs?
A: By utilizing a homogeneous palladium catalyst system with simplified workup procedures (filtration followed by column chromatography), the process avoids expensive metal scavenging operations required in alternative methodologies. This streamlines purification and reduces overall production costs through reduced processing steps and solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TFQ Derivatives Supplier
Our company leverages this patented technology to deliver high-purity 2-trifluoromethyl quinazolinone derivatives with exceptional consistency and reliability for global pharmaceutical partners. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art manufacturing facilities and rigorous QC labs. Our technical team has successfully implemented this CO-free carbonylation methodology across multiple client programs, demonstrating consistent ability to meet demanding quality requirements while optimizing production economics through continuous process improvement initiatives.
We invite you to request a Customized Cost-Saving Analysis tailored to your specific compound requirements from our technical procurement team, who can provide detailed information on available capacity, quality documentation, and technical support services. Please contact us to obtain specific COA data and route feasibility assessments for your target molecules to evaluate how this innovative manufacturing platform can enhance your pharmaceutical development pipeline.
