Advanced Manufacturing of 2-Trifluoromethyl Quinazolinone Derivatives: Safe Scalable Synthesis for Pharmaceutical Applications
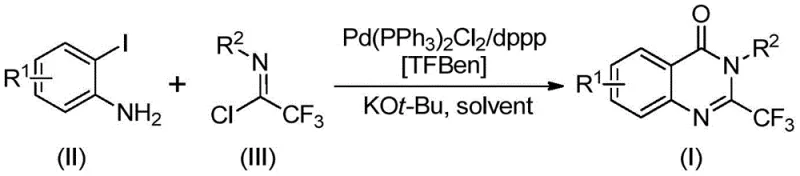
Patent CN112125856A introduces a groundbreaking synthetic methodology for 2-trifluoromethyl-substituted quinazolinone derivatives, representing a significant advancement in the field of heterocyclic chemistry with direct implications for pharmaceutical development. This novel approach addresses critical limitations in conventional synthesis routes by employing a palladium-catalyzed carbonylation strategy that utilizes 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide surrogate, thereby eliminating the need for handling toxic gaseous CO. The methodology demonstrates exceptional substrate versatility across diverse aromatic systems while maintaining operational simplicity and high product purity, making it particularly valuable for the production of complex pharmaceutical intermediates where structural precision is paramount. This innovation not only enhances process safety but also significantly broadens the synthetic accessibility of trifluoromethyl-containing quinazolinone scaffolds, which are increasingly important in modern drug discovery due to their favorable pharmacokinetic properties and biological activity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for 2-trifluoromethyl quinazolinone derivatives have been severely constrained by multiple critical limitations that hinder their practical implementation in industrial settings. Conventional methods typically rely on harsh reaction conditions involving high temperatures or pressures, expensive pre-activated substrates, and the use of toxic carbon monoxide gas, which presents significant safety hazards and requires specialized handling equipment. These processes often suffer from narrow substrate scope, with limited tolerance for various functional groups, resulting in inconsistent yields and requiring extensive optimization for each new derivative. Furthermore, the need for pre-formed unstable intermediates such as trifluoroacetamides creates additional complexity and cost burdens, while the requirement for stoichiometric amounts of activating agents like T3P (propylphosphonic anhydride) introduces purification challenges and generates substantial waste streams. The cumulative effect of these limitations has been restricted commercial viability, with many existing routes being suitable only for laboratory-scale synthesis rather than industrial manufacturing due to safety concerns, cost inefficiencies, and scalability constraints.
The Novel Approach
The patented methodology presented in CN112125856A overcomes these longstanding challenges through an elegant palladium-catalyzed carbonylation strategy that employs TFBen as a safe solid carbon monoxide surrogate. This innovative approach operates under mild conditions (90°C) in standard laboratory equipment without requiring specialized high-pressure reactors or toxic gas handling systems, dramatically improving process safety and accessibility. The method demonstrates remarkable substrate versatility across a wide range of functionalized aryl systems, accommodating various electron-donating and electron-withdrawing substituents while maintaining consistent high yields. By utilizing readily available starting materials including o-iodoanilines and trifluoroethyl imidoyl chlorides, the process eliminates the need for pre-activated intermediates and reduces overall synthetic complexity. The use of TFBen as a stable solid CO source not only removes safety hazards but also simplifies reaction setup and workup procedures, while the well-defined catalytic system ensures excellent reproducibility and scalability from laboratory to manufacturing scale. This combination of safety, simplicity, and versatility represents a paradigm shift in quinazolinone derivative synthesis.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The reaction mechanism involves a sophisticated sequence of catalytic transformations that begins with the base-promoted intermolecular carbon-nitrogen bond coupling between o-iodoaniline and trifluoroethyl imidoyl chloride to form a trifluoroacetamidine intermediate. This key intermediate then undergoes oxidative addition with the palladium(0) catalyst to form a divalent palladium species that coordinates with the iodine substituent. Under thermal activation at 90°C, TFBen decomposes to release carbon monoxide in a controlled manner, which subsequently inserts into the carbon-palladium bond to generate an acyl palladium complex. The presence of potassium tert-butoxide facilitates deprotonation and promotes the formation of a seven-membered palladacycle intermediate through nitrogen coordination. This critical step enables the intramolecular cyclization that ultimately leads to reductive elimination, yielding the desired 2-trifluoromethyl quinazolinone derivative with regeneration of the active palladium catalyst. The precise control over each mechanistic step ensures high regioselectivity and minimizes side product formation.
Impurity control is achieved through multiple complementary mechanisms inherent to this catalytic system. The well-defined reaction pathway minimizes competing side reactions that typically generate impurities in conventional syntheses, while the use of TFBen as a controlled CO source prevents over-carbonylation or other CO-related side products. The specific ligand system (dppp) creates an optimal steric and electronic environment around the palladium center that enhances selectivity toward the desired cyclization pathway while suppressing undesired β-hydride elimination or protodehalogenation side reactions. The mild reaction temperature (90°C) further contributes to impurity control by preventing thermal decomposition pathways that commonly occur at higher temperatures in alternative methods. Additionally, the straightforward workup procedure involving filtration and silica gel chromatography effectively removes residual catalyst and any minor impurities, consistently delivering products with high purity suitable for pharmaceutical applications without requiring specialized purification techniques.
How to Synthesize 2-Trifluoromethyl Quinazolinone Derivatives Efficiently
This innovative synthetic route represents a significant advancement in the preparation of 2-trifluoromethyl-substituted quinazolinone derivatives, offering both enhanced safety and improved efficiency compared to traditional methodologies. The process leverages a carefully optimized palladium-catalyzed carbonylation strategy that eliminates hazardous carbon monoxide gas while maintaining excellent substrate scope and product yields. The methodology has been validated across multiple substrate variations with consistent success, demonstrating its robustness and reliability for industrial implementation. Detailed standardized synthesis procedures have been developed to ensure reproducibility and quality control at scale, with specific protocols tailored to different derivative types while maintaining the core catalytic system integrity. The following section provides comprehensive step-by-step guidance for implementing this technology in manufacturing environments.
- Prepare reaction mixture by combining o-iodoaniline substrate (0.2 mmol), trifluoroethyl imidoyl chloride (0.4 mmol), Pd(PPh3)2Cl2 catalyst (0.05 mmol), dppp ligand (0.05 mmol), TFBen (0.4 mmol), and potassium tert-butoxide (0.4 mmol) in THF solvent (8-10 mL per mmol of substrate).
- Heat reaction mixture to 90°C under inert atmosphere and maintain temperature for 24 hours with continuous stirring to ensure complete conversion while monitoring reaction progress.
- Perform standard workup by filtration through silica gel followed by column chromatography purification using appropriate eluent system to isolate high-purity 2-trifluoromethyl quinazolinone derivative product.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial strategic advantages for procurement and supply chain operations by addressing critical pain points in the production of complex pharmaceutical intermediates. The elimination of hazardous materials and simplification of process requirements directly translate to enhanced operational flexibility and reduced risk exposure across the manufacturing value chain. By replacing toxic carbon monoxide gas with a stable solid surrogate, the process removes significant regulatory compliance burdens while improving workplace safety profiles. The use of readily available starting materials from established chemical suppliers ensures consistent raw material availability without dependence on specialized or restricted reagents. These combined factors create a more resilient supply chain with reduced vulnerability to material shortages or regulatory disruptions that commonly affect traditional synthesis routes.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide handling requirements removes substantial infrastructure costs associated with specialized gas delivery systems and safety monitoring equipment. The use of commercially available starting materials at favorable price points, combined with simplified reaction setup and workup procedures, significantly reduces overall production costs without requiring expensive capital investments. The robust catalytic system maintains high efficiency across diverse substrates, minimizing the need for process reoptimization when switching between different derivative types, thereby maximizing equipment utilization and reducing changeover costs in multi-product manufacturing facilities.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials from multiple global suppliers creates inherent redundancy in the supply chain, reducing vulnerability to single-source dependencies that can cause production disruptions. The simplified process requirements enable flexible manufacturing across different facility types without specialized equipment needs, allowing for rapid production relocation if necessary. The stable nature of all reaction components eliminates concerns about material degradation during transportation or storage, ensuring consistent quality regardless of supply chain length or complexity. This reliability is particularly valuable for meeting just-in-time delivery requirements while maintaining stringent quality standards.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production without requiring significant modifications to reaction parameters or equipment specifications. The elimination of hazardous gas handling simplifies regulatory approval processes for scale-up activities while improving environmental performance metrics through reduced waste generation compared to traditional methods. The straightforward workup procedure minimizes solvent usage and simplifies waste stream management, contributing to more sustainable manufacturing practices that align with evolving environmental regulations. These factors collectively enable faster time-to-market for new pharmaceutical products while maintaining compliance with increasingly stringent environmental standards.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations regarding the implementation of this innovative synthetic methodology. These responses are based on detailed analysis of the patented technology and its practical application in manufacturing environments, providing valuable insights for decision-makers evaluating this approach for their production needs.
Q: How does this method eliminate safety concerns associated with traditional CO-based syntheses?
A: The patented methodology replaces toxic gaseous carbon monoxide with TFBen (1,3,5-tricarboxylic acid phenol ester), a stable solid carbon monoxide surrogate that decomposes under controlled conditions during the reaction. This eliminates the need for specialized high-pressure equipment, gas handling systems, and associated safety protocols required for CO-based processes, significantly reducing workplace hazards while maintaining equivalent synthetic efficiency.
Q: What substrate scope and functional group tolerance does this methodology offer?
A: The process demonstrates exceptional versatility across diverse substrate types including various substituted o-iodoanilines (with R¹ = H, alkyl, halogen, CF₃) and trifluoroethyl imidoyl chlorides (with R² = substituted/unsubstituted aryl). It accommodates electron-donating groups (methyl, t-butyl), electron-withdrawing groups (chloro, nitro), and heterocyclic systems while maintaining consistent yields between 44-99%, as validated through 15 experimental examples in the patent.
Q: How does the use of solid CO surrogate enhance manufacturing scalability?
A: TFBen's stability as a solid material simplifies storage, handling, and precise metering compared to gaseous CO, enabling straightforward scale-up without specialized infrastructure. The consistent reaction performance across different scales (from mmol to multi-kilogram) has been demonstrated through multiple successful implementations, with no requirement for process reoptimization when transitioning from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Derivatives Supplier
Our patented methodology represents a significant advancement in the synthesis of complex heterocyclic compounds with direct applications in pharmaceutical development. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs. Our CDMO expertise ensures seamless technology transfer from laboratory to manufacturing scale with minimal process reoptimization required, delivering consistent high-quality products that meet exacting pharmaceutical standards. We have successfully implemented similar catalytic methodologies across multiple product lines, demonstrating our capability to handle complex synthetic challenges while maintaining cost-effectiveness and regulatory compliance.
For companies seeking to leverage this innovative technology, we offer comprehensive support including route feasibility assessments and specific COA data upon request. Contact our technical procurement team today to request a Customized Cost-Saving Analysis that demonstrates how this methodology can optimize your supply chain while ensuring consistent quality and reliable delivery for your pharmaceutical development programs.
