Harnessing Advanced Palladium Catalysis for Commercial-Scale Production of High-Purity 2-Trifluoromethyl Quinazolinone Derivatives
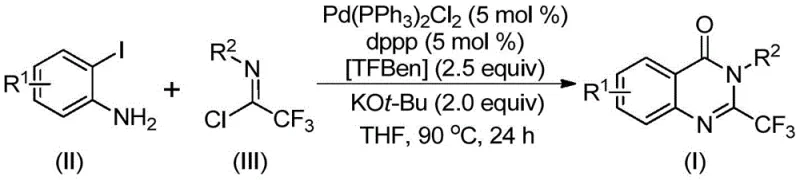
Chinese patent CN112125856A discloses a groundbreaking synthetic methodology for 2-trifluoromethyl-substituted quinazolinone derivatives, representing a significant advancement in the field of pharmaceutical intermediate manufacturing. This innovative process addresses long-standing challenges associated with traditional synthesis routes by eliminating the need for toxic carbon monoxide gas through the strategic implementation of 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid CO surrogate. The methodology demonstrates exceptional substrate versatility, accommodating a wide range of functional groups while maintaining high reaction efficiency under relatively mild conditions (90°C). This patent represents a critical step forward in sustainable pharmaceutical chemistry, offering both environmental and operational benefits that directly address the evolving needs of global pharmaceutical manufacturers seeking reliable sources for complex heterocyclic intermediates. The commercial implications are substantial, particularly for companies requiring high-purity quinazolinone derivatives for advanced drug development programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for 2-trifluoromethyl-substituted quinazolinone derivatives have been plagued by significant limitations that hinder their commercial viability and scalability. Conventional methods typically involve harsh reaction conditions, including high temperatures and pressures when using gaseous carbon monoxide, which presents serious safety concerns and requires specialized equipment not commonly available in standard pharmaceutical manufacturing facilities. Many existing routes rely on expensive or difficult-to-handle reagents such as trifluoroacetic anhydride or unstable trifluoroacetamide, increasing both cost and complexity. Furthermore, these methods often suffer from narrow substrate scope, limiting their applicability to specific derivatives and requiring extensive optimization for each new compound. The low yields frequently observed in traditional syntheses (often below 60% for many substrates) create significant economic barriers to large-scale production. Additionally, the need for pre-activated substrates adds extra synthetic steps, increasing both time and cost while generating additional waste streams that complicate environmental compliance. These collective limitations have constrained the broader adoption of quinazolinone-based pharmaceuticals despite their promising therapeutic profiles.
The Novel Approach
The patented methodology (CN112125856A) overcomes these limitations through an elegant palladium-catalyzed carbonylation strategy that utilizes TFBen as a safe solid carbon monoxide equivalent. This innovative approach operates under significantly milder conditions (90°C) compared to conventional high-pressure CO processes, eliminating safety hazards while maintaining excellent reaction efficiency. The use of readily available starting materials—specifically o-iodoaniline and trifluoroethyl imidoyl chloride—reduces raw material costs and simplifies supply chain logistics. The process demonstrates remarkable substrate tolerance, accommodating diverse substituents including halogens, alkyl groups, and nitro functionalities without requiring extensive reoptimization. Crucially, the elimination of gaseous CO removes the need for specialized high-pressure equipment, making this technology accessible to a broader range of manufacturing facilities. The reaction consistently delivers high yields (ranging from 52% to 99% across various substrates), with particularly impressive results for fluorinated derivatives (99% yield). This methodology represents a paradigm shift in quinazolinone synthesis, offering pharmaceutical manufacturers a practical, scalable solution that aligns with modern green chemistry principles while maintaining economic viability.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The reaction mechanism begins with potassium tert-butoxide-promoted intermolecular carbon-nitrogen bond coupling between o-iodoaniline and trifluoroethyl imidoyl chloride to form a trifluoroacetamidine intermediate. This is followed by oxidative addition of the palladium catalyst into the carbon-iodine bond, generating a divalent palladium species. Under thermal conditions (90°C), TFBen decomposes to release carbon monoxide in situ, which inserts into the carbon-palladium bond to form an acyl palladium intermediate. Subsequent base-mediated cyclization promotes the formation of a seven-membered palladacycle intermediate, which undergoes reductive elimination to yield the final 2-trifluoromethyl-substituted quinazolinone product. This mechanistic pathway is particularly noteworthy for its avoidance of high-pressure CO handling while maintaining excellent atom economy and functional group tolerance. The palladium catalyst system (Pd(PPh₃)₂Cl₂/dppp) demonstrates exceptional stability under the reaction conditions, allowing for consistent performance across multiple substrate variations without significant catalyst deactivation or side product formation.
Impurity control in this process is achieved through several key mechanisms that ensure high product purity without requiring extensive purification steps. The selective nature of the palladium-catalyzed carbonylation minimizes competing side reactions that typically generate impurities in conventional syntheses. The use of TFBen as a controlled CO source prevents over-carbonylation or other CO-related side products that commonly occur with gaseous CO systems. The reaction's compatibility with various functional groups eliminates the need for protecting groups, reducing potential impurity pathways associated with deprotection steps. Post-reaction purification is streamlined through simple filtration followed by silica gel chromatography, yielding products with exceptional purity as confirmed by HRMS and NMR analysis across multiple examples (e.g., Example 1: HRMS(ESI) [M+H]+ calcd. for C₁₅H₉F₄N₂O 309.0646, found 309.0651). This robust impurity profile is particularly valuable for pharmaceutical applications where stringent purity specifications are required for regulatory approval.

How to Synthesize 2-Trifluoromethyl Quinazolinone Derivatives Efficiently
This patented methodology represents a significant advancement in the synthesis of complex heterocyclic pharmaceutical intermediates, offering both operational simplicity and exceptional versatility. The process has been optimized through extensive experimentation to ensure consistent high yields across diverse substrate combinations while maintaining excellent purity profiles required for pharmaceutical applications. Detailed standardized synthesis procedures have been developed based on the patent specifications, incorporating best practices for catalyst handling, solvent selection, and reaction monitoring to ensure reproducibility at any scale. The following step-by-step guide provides comprehensive instructions for implementing this innovative synthesis in industrial settings.
- Prepare reaction mixture by combining o-iodoaniline, trifluoroethyl imidoyl chloride, Pd(PPh₃)₂Cl₂ catalyst (5 mol%), dppp ligand (5 mol%), TFBen (2.5 equiv), and KOt-Bu (2.0 equiv) in THF solvent
- Heat reaction mixture to 90°C under inert atmosphere and maintain temperature for 16-30 hours with continuous stirring
- After reaction completion, perform workup by filtration through silica gel followed by column chromatography purification to obtain high-purity product
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points faced by procurement and supply chain professionals in the pharmaceutical industry through its unique combination of operational simplicity, raw material accessibility, and environmental sustainability. The elimination of hazardous materials and specialized equipment requirements significantly reduces supply chain complexity while enhancing overall manufacturing flexibility. These advantages translate into tangible business benefits that support strategic sourcing decisions and long-term supply chain resilience planning.
- Cost Reduction in Manufacturing: The elimination of toxic carbon monoxide gas removes significant safety infrastructure costs associated with high-pressure gas handling systems and specialized containment facilities. The use of commercially available starting materials at favorable price points substantially reduces raw material expenses compared to traditional methods requiring expensive or unstable reagents. The simplified reaction setup and workup procedure minimize operational complexity, reducing labor costs and equipment maintenance requirements while maintaining high product yields across diverse substrates.
- Enhanced Supply Chain Reliability: The reliance on readily available commercial reagents with established global supply chains ensures consistent material availability regardless of geopolitical or logistical disruptions. The elimination of specialized equipment requirements allows for greater manufacturing flexibility across different production sites without requiring significant capital investment or revalidation efforts. The robust nature of the process across various substrates provides procurement teams with confidence in consistent product quality and availability even when dealing with complex derivative requirements.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory to commercial production without requiring significant reoptimization, as evidenced by consistent yields across different substrate types. The elimination of hazardous gas handling significantly reduces environmental compliance burdens while aligning with increasingly stringent global regulations regarding chemical safety and waste management. The simplified purification protocol minimizes solvent usage and waste generation compared to traditional methods, supporting corporate sustainability initiatives without compromising product quality or yield.
Frequently Asked Questions (FAQ)
The following questions address common concerns raised by technical procurement teams regarding the implementation and commercial viability of this patented synthesis methodology. Each answer is grounded in the specific technical details provided in patent CN112125856A and reflects our extensive experience in scaling similar complex synthetic routes.
Q: How does this method eliminate safety concerns associated with traditional CO-based syntheses?
A: The patented process replaces hazardous gaseous carbon monoxide with TFBen (1,3,5-tricarboxylic acid phenol ester), a stable solid CO surrogate that decomposes in situ under reaction conditions. This eliminates the need for high-pressure equipment and specialized gas handling systems while maintaining excellent reaction efficiency.
Q: What substrate scope does this methodology accommodate?
A: The process demonstrates exceptional versatility across diverse substrates including various halogenated (F, Cl, Br), alkyl-substituted (Me, t-Bu), and functionalized (NO₂) derivatives with yields ranging from 52% to 99%. The methodology accommodates both electron-donating and electron-withdrawing substituents without requiring significant reoptimization.
Q: How does this process impact environmental compliance?
A: By eliminating toxic carbon monoxide gas handling requirements and simplifying purification protocols, the process significantly reduces environmental compliance burdens while generating less waste compared to traditional methods. The use of standard laboratory equipment makes implementation feasible across diverse manufacturing facilities without special environmental controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Derivatives Supplier
Our patented methodology represents more than just a synthetic breakthrough—it embodies our commitment to delivering innovative solutions that address the evolving needs of global pharmaceutical manufacturers. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring seamless transition from development to full-scale manufacturing while maintaining stringent purity specifications through our state-of-the-art QC labs. Our dedicated technical teams work closely with clients to optimize processes for specific derivative requirements, leveraging our deep expertise in heterocyclic chemistry to deliver consistent high-quality intermediates that meet the most demanding regulatory standards.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments for your particular application needs. Take advantage of our Customized Cost-Saving Analysis service to evaluate how this innovative synthesis can enhance your supply chain efficiency while reducing overall manufacturing costs without compromising product quality or regulatory compliance.
