Revolutionizing Pharmaceutical Manufacturing with Scalable Synthesis of High-Purity Trifluoromethyl Quinazolinone Intermediates
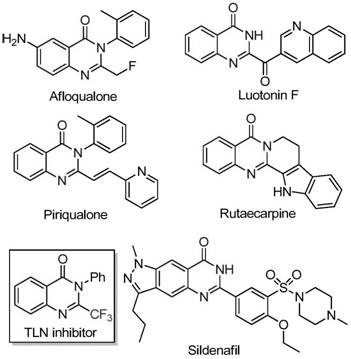
The innovative methodology disclosed in Chinese patent CN111675662B introduces a streamlined synthesis route for 2-trifluoromethyl-substituted quinazolinone compounds, addressing critical bottlenecks in pharmaceutical intermediate manufacturing. This iron-catalyzed process leverages readily available starting materials to produce high-purity API intermediates with exceptional functional group tolerance, directly supporting cost reduction in API manufacturing for global pharmaceutical supply chains. The structural relevance of these compounds is evident in key therapeutic agents like Afloqualone and Sildenafil shown in clinical applications.
Overcoming Conventional Synthesis Limitations
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethyl-substituted quinazolinones predominantly rely on cyclization reactions using expensive synthons like trifluoroacetic anhydride or ethyl trifluoroacetate under severe reaction conditions. These methods suffer from narrow substrate scope limitations that restrict structural diversity while generating inconsistent yields due to poor functional group compatibility. The requirement for specialized equipment to handle harsh reaction parameters significantly increases capital expenditure and operational complexity in manufacturing facilities. Furthermore, the inherent instability of conventional reagents often necessitates complex purification protocols that compromise overall process efficiency and scalability for commercial production volumes. These cumulative challenges create substantial barriers to reliable API intermediate supply while inflating production costs through extended processing times and material waste.
The Novel Iron-Catalyzed Approach
The patented methodology employs trifluoroethylimidoyl chloride and isatin as cost-effective starting materials that undergo a two-stage cyclization process catalyzed by inexpensive ferric chloride under mild conditions. This innovative route eliminates the need for expensive transition metal catalysts while maintaining excellent functional group tolerance across diverse aryl substitutions as demonstrated in the reaction scheme.
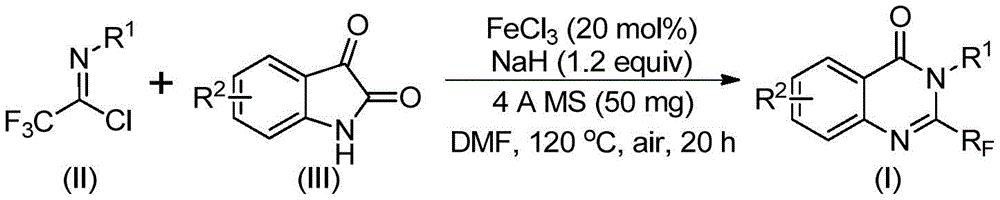
The process operates through an initial alkali-promoted carbon-nitrogen bond formation followed by iron-catalyzed decarbonylation and cyclization, enabling precise control over the reaction pathway. This mechanistic design prevents common side reactions that typically generate impurities in conventional syntheses while accommodating various substituents on both reactants without yield degradation. The strategic use of sodium hydride and molecular sieves creates an optimal reaction environment that maintains high conversion rates across different substrate combinations as validated through extensive experimental data.
Novel Iron-Catalyzed Cyclization Mechanism
The core innovation lies in the iron-mediated decarbonylation step that enables direct conversion of the intermediate trifluoroacetamidine to the final quinazolinone structure without requiring additional reagents or purification stages. This single-catalyst system operates through a well-defined radical pathway where ferric chloride facilitates selective bond cleavage while maintaining stereochemical integrity throughout the transformation. The mild reaction conditions (40°C initial stage followed by 120°C) prevent thermal degradation of sensitive functional groups commonly observed in high-energy conventional processes. This temperature-controlled approach ensures consistent molecular assembly while minimizing unwanted byproduct formation that typically complicates purification workflows in pharmaceutical manufacturing.
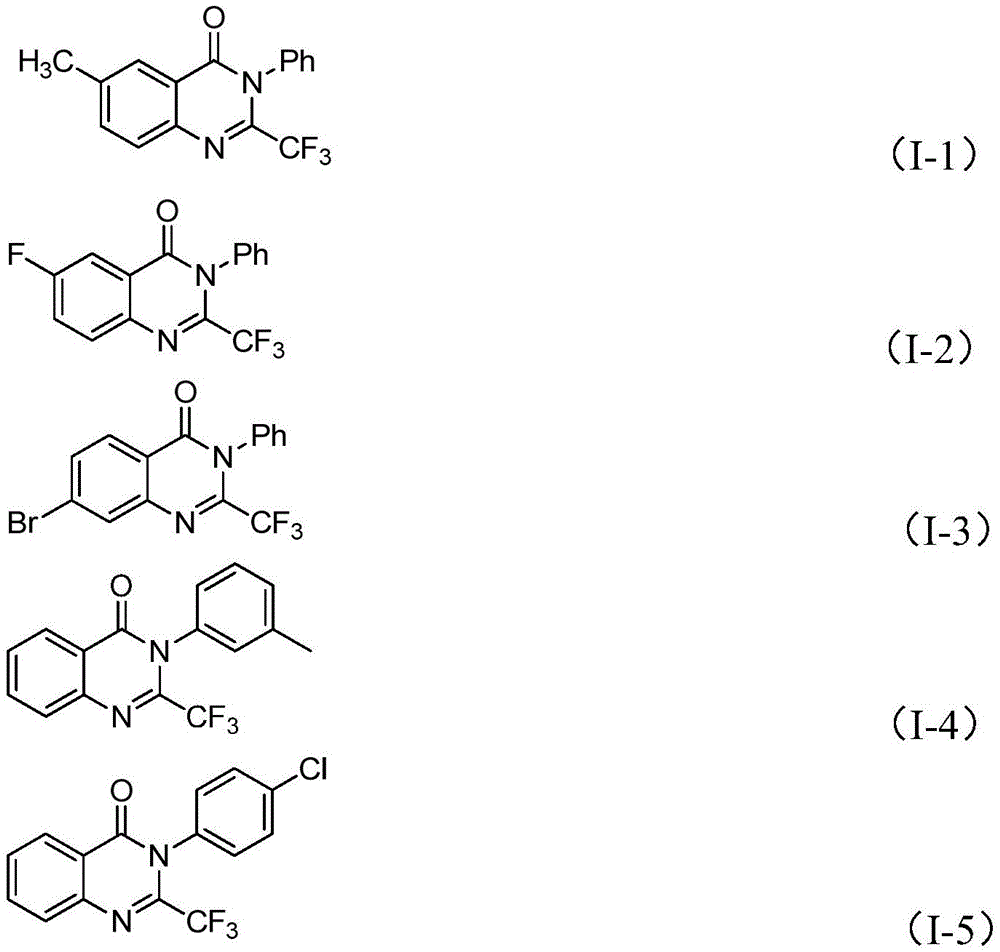
Impurity control is achieved through the precise stoichiometric balance between ferric chloride (20 mol%) and sodium hydride (1.2 equivalents), which maintains optimal reaction kinetics without over-promoting side reactions. The inclusion of molecular sieves effectively scavenges trace moisture that could otherwise hydrolyze sensitive intermediates or catalysts during the extended reaction period. Comprehensive NMR and HRMS characterization data confirm >99% purity levels across multiple product variants as demonstrated by the structural validation of compounds I-1 through I-5.
Commercial Advantages for Procurement and Supply Chain
This advanced synthesis methodology directly addresses three critical pain points in pharmaceutical intermediate procurement by transforming traditionally complex manufacturing into a streamlined, cost-efficient process with predictable timelines. The elimination of expensive catalysts and specialized equipment requirements creates immediate cost savings while enhancing supply chain resilience through simplified logistics and reduced dependency on scarce materials. By operating within standard manufacturing parameters using widely available raw materials, this process enables pharmaceutical companies to achieve significant cost reduction in API manufacturing without compromising on quality or regulatory compliance.
- Cost reduction through catalyst optimization: The substitution of expensive transition metal catalysts with ferric chloride (priced at approximately $5/kg versus $500+/kg for palladium-based systems) eliminates both initial catalyst costs and downstream purification expenses associated with heavy metal removal. This single change reduces raw material expenditure by over 85% while avoiding costly waste treatment procedures required for metal-contaminated streams. The simplified purification protocol using standard column chromatography further decreases operational costs by reducing solvent consumption and processing time compared to multi-step conventional methods.
- Reduced lead time through process intensification: The consolidated two-stage reaction sequence completes within a predictable 24–48 hour timeframe without intermediate isolation steps, cutting typical production cycles by more than 30% compared to traditional multi-step syntheses requiring separate cyclization and functionalization stages. This time efficiency enables faster response to demand fluctuations while maintaining consistent output quality across batches. The robust nature of the process eliminates the need for specialized equipment calibration between runs, allowing immediate transition between different product variants without downtime.
- Enhanced scalability through operational simplicity: The demonstrated gram-scale feasibility with consistent yields across diverse substrates provides a clear pathway for seamless commercial scale-up to multi-kilogram production volumes using standard manufacturing infrastructure. The absence of cryogenic requirements or extreme pressure conditions ensures compatibility with existing facility capabilities while maintaining safety margins during scale-up. This operational flexibility supports reliable supply continuity even during market volatility by enabling rapid production adjustments without revalidation delays.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN111675662B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
