Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic methodologies to access complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN112125856A discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone derivatives, addressing critical challenges in safety and efficiency that have long plagued traditional synthesis routes. Quinazolinone derivatives are privileged structures found in numerous bioactive molecules, exhibiting a spectrum of pharmacological activities including anti-inflammatory, antiviral, antifungal, anticonvulsant, and anticancer properties. The strategic introduction of a trifluoromethyl group at the 2-position is particularly valuable, as fluorine atoms can drastically enhance metabolic stability, lipophilicity, and bioavailability of the parent molecule. This patent presents a transition metal palladium-catalyzed carbonylation tandem reaction that utilizes inexpensive and readily available starting materials, specifically o-iodoaniline and trifluoroethylimidoyl chloride, to construct these valuable cores with high precision.
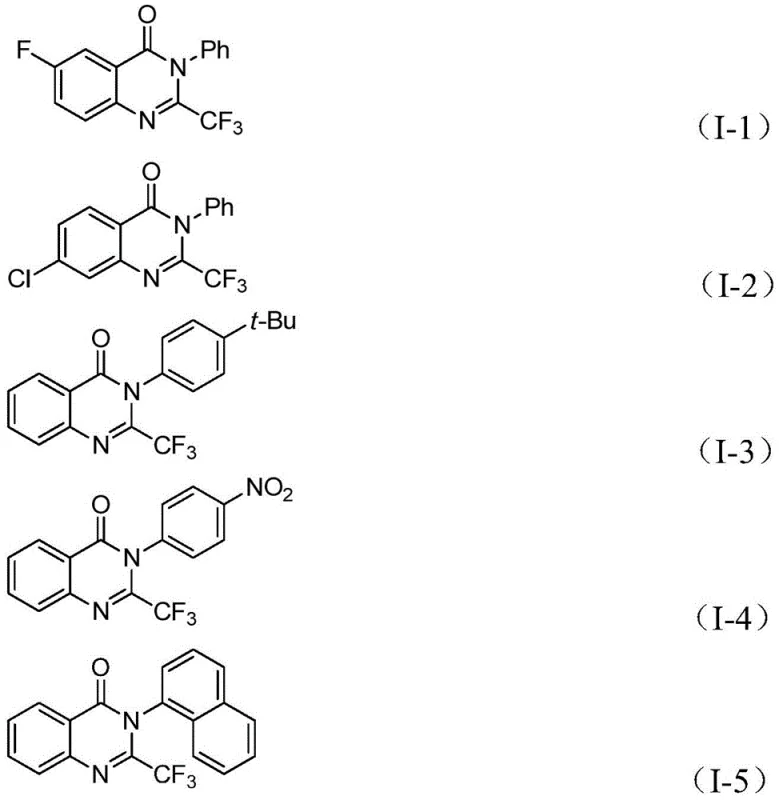
The significance of this technology lies not only in its chemical elegance but also in its practical applicability for industrial manufacturing. Unlike conventional methods that often rely on hazardous gaseous reagents or harsh conditions, this novel approach employs 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute. This substitution effectively mitigates the risks associated with handling toxic carbon monoxide gas, making the process inherently safer for large-scale operations. Furthermore, the method demonstrates exceptional substrate compatibility, allowing for the synthesis of derivatives with various substituents such as halogens, alkyl groups, and nitro groups. For R&D directors and process chemists, this represents a versatile platform for library generation and lead optimization, while for supply chain managers, it offers a pathway to reliable production without the need for specialized high-pressure gas infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinone derivatives has been fraught with significant technical and safety hurdles that limit their widespread adoption in commercial manufacturing. Traditional literature reports describe several common synthetic strategies, such as the cyclization of anthranilamide with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions. Other methods involve the cyclization of anthranilic acid esters with unstable trifluoroacetamides or the reaction of isatoic anhydride with trifluoroacetic anhydride. While these methods can yield the desired products, they generally suffer from severe drawbacks including harsh reaction conditions that require extreme temperatures or pressures, and the use of expensive or difficult-to-handle reagents that necessitate pre-activation steps. Moreover, many of these conventional routes exhibit low yields and narrow substrate scopes, meaning they fail when applied to more complex or sensitive molecular architectures. Perhaps most critically from an industrial safety perspective, some traditional carbonylation methods rely directly on carbon monoxide gas, a colorless, odorless, and highly toxic substance that requires rigorous safety protocols and specialized equipment to manage effectively.
The Novel Approach
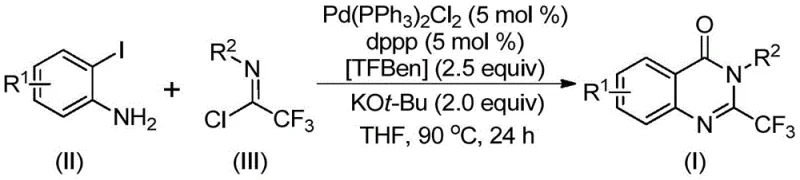
In stark contrast to these legacy methods, the technology described in patent CN112125856A introduces a streamlined, palladium-catalyzed carbonylation tandem reaction that fundamentally reshapes the synthesis landscape for these compounds. By utilizing o-iodoaniline and trifluoroethylimidoyl chloride as the primary building blocks, the process bypasses the need for pre-activated substrates and harsh cyclization agents. The core innovation is the use of TFBen as a solid carbon monoxide surrogate, which decomposes under heating conditions to release CO in situ, thereby driving the carbonylation step without the dangers of handling gas cylinders. This approach not only simplifies the operational workflow but also enhances the safety profile of the entire manufacturing process. The reaction proceeds efficiently in common organic solvents like tetrahydrofuran at moderate temperatures around 90°C, utilizing a catalyst system composed of Pd(PPh3)2Cl2 and dppp. This mild yet effective protocol ensures high conversion rates and broad functional group tolerance, enabling the production of diverse quinazolinone derivatives that were previously difficult or dangerous to synthesize.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific analogs. The reaction is believed to proceed through a sophisticated catalytic cycle initiated by the base-promoted intermolecular carbon-nitrogen bond coupling between the o-iodoaniline and the trifluoroethylimidoyl chloride, likely forming a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the aromatic ring, generating a divalent palladium species. As the reaction mixture is heated, the solid CO substitute TFBen decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to form a key acyl-palladium intermediate. This insertion step is the defining moment where the carbonyl group is incorporated into the growing molecular framework. Following this, the base facilitates the formation of a palladium-nitrogen bond, leading to the construction of a seven-membered ring palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone derivative and regenerates the active palladium catalyst for the next turnover. This intricate dance of bond formations and breakages ensures high atom economy and structural fidelity.
From an impurity control perspective, the mild nature of this catalytic system offers distinct advantages over harsher thermal cyclizations. Because the reaction operates at a controlled temperature of 90°C rather than extreme heat, the likelihood of thermal decomposition or non-specific side reactions is significantly reduced. The use of a specific ligand system (dppp) helps stabilize the palladium center, preventing the formation of palladium black or other inactive species that could contaminate the product. Furthermore, the in situ generation of carbon monoxide from TFBen ensures a steady, controlled concentration of the reactive gas, preventing the runaway reactions that can occur with direct gas charging. This controlled environment leads to a cleaner crude reaction mixture, which simplifies downstream purification processes such as column chromatography. For quality assurance teams, this means a more consistent impurity profile and higher confidence in meeting stringent purity specifications required for pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process involves combining the palladium catalyst, ligand, base, solid CO source, and substrates in a standard reaction vessel, followed by heating and standard workup procedures. This straightforward approach minimizes the need for complex engineering controls, making it accessible for various manufacturing settings. Detailed standardized synthesis steps are provided in the guide below to ensure consistent results across different batches and facilities.
- Combine palladium catalyst, ligand, solid CO substitute (TFBen), base, trifluoroethylimidoyl chloride, and o-iodoaniline in an organic solvent like THF.
- Heat the reaction mixture to 90°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Perform post-treatment including filtration and silica gel mixing, followed by column chromatography purification to isolate the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The shift away from hazardous gaseous reagents and towards stable solid surrogates fundamentally alters the risk profile and cost structure of the supply chain. By eliminating the need for high-pressure gas handling infrastructure, companies can reduce capital expenditure on specialized reactors and safety systems, leading to significant cost reductions in pharmaceutical intermediate manufacturing. Additionally, the reliance on commercially available and inexpensive starting materials like o-iodoaniline derivatives and trifluoroethylimidoyl chlorides ensures a stable and resilient supply base. These raw materials are widely produced for other chemical applications, meaning there is less risk of supply disruption compared to bespoke or highly specialized reagents. The robustness of the reaction conditions also implies that the process can be scaled up with greater confidence, reducing the lead time for high-purity pharmaceutical intermediates needed for clinical trials and commercial launch.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of cheap and readily available starting materials combined with a catalyst system that operates efficiently at low loading levels. By avoiding the use of expensive pre-activated substrates and eliminating the logistical costs associated with transporting and storing toxic carbon monoxide gas, the overall production cost is substantially lowered. The simplified post-treatment process, which involves standard filtration and chromatography, further reduces operational expenses related to waste disposal and energy consumption. This holistic cost optimization makes the production of 2-trifluoromethyl quinazolinones economically attractive for high-volume applications.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of commodity chemicals that are not subject to the same regulatory restrictions as controlled gases. The solid nature of the carbon monoxide substitute (TFBen) allows for easier storage and transportation, removing the bottlenecks often associated with hazardous gas delivery. Furthermore, the broad substrate scope means that if a specific aniline derivative becomes unavailable, the chemistry is flexible enough to accommodate alternative substituents without requiring a complete process redevelopment. This flexibility provides a buffer against market volatility and ensures that production schedules can be maintained even when facing raw material fluctuations.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns well with modern green chemistry principles. The avoidance of toxic gas emissions reduces the environmental footprint of the manufacturing process, simplifying compliance with increasingly strict environmental regulations. The reaction's compatibility with standard organic solvents like THF allows for easy integration into existing solvent recovery systems. As the process moves from gram-scale to kilogram or ton-scale, the inherent safety of using a solid CO source becomes even more critical, facilitating smoother technology transfer and scale-up without the need for exponential increases in safety overheads.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived from the specific details and advantages highlighted in the patent documentation. These insights are intended to clarify the operational benefits and chemical capabilities of the technology for potential partners and stakeholders. Understanding these nuances is essential for evaluating the fit of this methodology within your specific development pipeline.
Q: What are the safety advantages of using TFBen over gaseous carbon monoxide?
A: TFBen (1,3,5-tricarboxylate phenol ester) acts as a solid carbon monoxide substitute, eliminating the need for handling toxic, colorless CO gas under high pressure, thereby significantly enhancing operational safety and reducing infrastructure costs.
Q: What is the substrate scope for this quinazolinone synthesis method?
A: The method demonstrates excellent compatibility with various substituents on both the o-iodoaniline and the imidoyl chloride components, including halogens, alkyl groups, and nitro groups, allowing for the design of diverse derivatives.
Q: Why is the 2-trifluoromethyl group significant in drug design?
A: Introducing a trifluoromethyl group significantly improves physicochemical properties such as metabolic stability, lipophilicity, and bioavailability, which are critical for optimizing the efficacy of pharmaceutical candidates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in patent CN112125856A for accelerating drug discovery and development. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. Our state-of-the-art facilities are equipped to handle complex palladium-catalyzed reactions with the highest standards of safety and efficiency, supported by our rigorous QC labs that guarantee stringent purity specifications for every batch. We understand that consistency and quality are paramount in the pharmaceutical supply chain, and our team is committed to delivering intermediates that meet the exacting demands of global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate how our expertise can optimize your supply chain and reduce your time to market. Let us help you turn complex chemical challenges into commercial successes.
