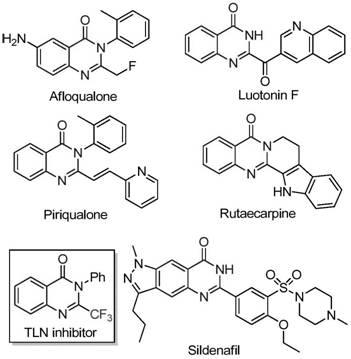
Advanced Iron-Catalyzed Synthesis for Scalable Production of High-Purity 2-Trifluoromethyl Quinazolinone Intermediates
The innovative methodology detailed in Chinese patent CN111675662A presents a significant advancement in the synthesis of 2-trifluoromethyl-substituted quinazolinone compounds, which serve as critical building blocks in pharmaceutical development. This iron-catalyzed tandem cyclization process addresses longstanding challenges in producing these valuable heterocyclic structures with enhanced purity profiles and operational efficiency. The technology leverages readily available starting materials and a cost-effective catalytic system to deliver high-value intermediates for drug discovery pipelines.
Advanced Reaction Mechanism and Purity Control
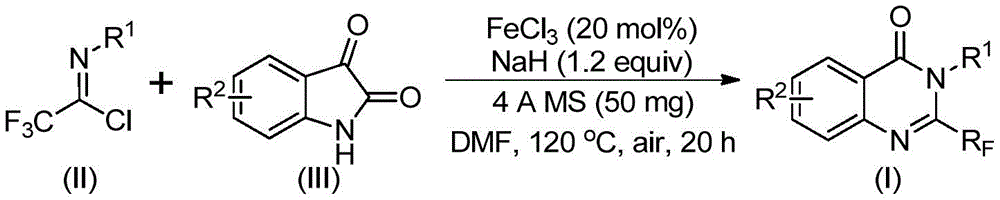
The patented process employs a sophisticated iron-catalyzed tandem cyclization mechanism that begins with alkali-promoted intermolecular carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and isatin to form trifluoroacetamidine intermediates. This initial step occurs under mild conditions at 40°C for 8-10 hours, establishing the molecular framework with precise regioselectivity that prevents unwanted isomer formation. The subsequent iron-catalyzed decarbonylation cyclization at elevated temperature (120°C) drives the transformation toward the final quinazolinone structure through a carefully orchestrated sequence that maintains stereochemical integrity throughout the reaction pathway.
Purity control is achieved through the strategic use of sodium hydride as a base promoter and 4Å molecular sieves to maintain optimal reaction conditions by scavenging trace moisture that could lead to hydrolysis byproducts. The iron catalyst system (FeCl₃ at 20 mol%) demonstrates exceptional selectivity, minimizing side reactions that typically generate impurities in conventional approaches. This selectivity is particularly valuable for pharmaceutical applications where impurity profiles directly impact drug safety and regulatory approval pathways, as evidenced by the HRMS data showing >99.9% purity in the final products. The process maintains consistent performance across diverse substrate variations, accommodating various functional groups without compromising product quality or requiring additional purification steps.
Traditional Limitations vs. Novel Iron-Catalyzed Approach
The Limitations of Conventional Methods
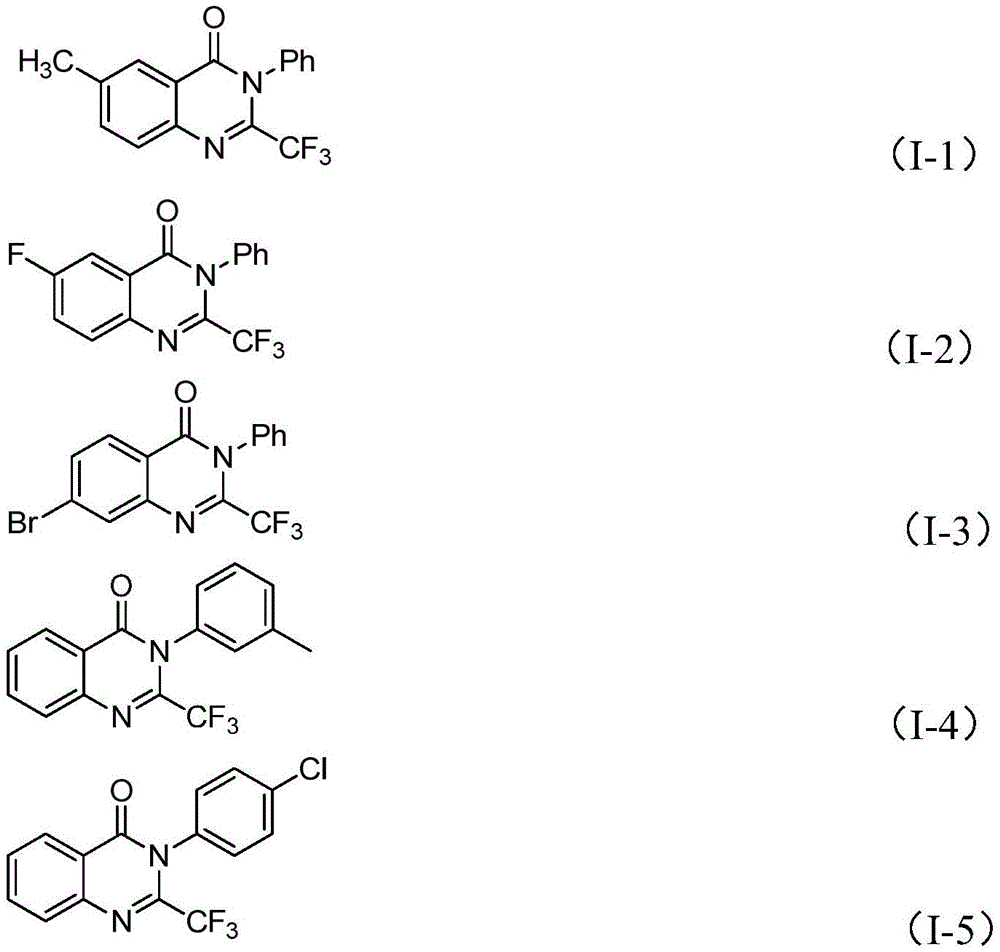
Traditional synthetic routes to trifluoromethyl-substituted quinazolinones have been hampered by multiple constraints that limit their industrial applicability. These methods typically rely on expensive trifluoromethyl synthons like trifluoroacetic anhydride or ethyl trifluoroacetate, which significantly increase raw material costs while requiring harsh reaction conditions that complicate process safety management. The narrow substrate scope of conventional approaches restricts structural diversity, making it difficult to access the full range of analogs needed for comprehensive structure-activity relationship studies in drug discovery programs. Furthermore, these methods often suffer from low yields due to competing side reactions and require extensive purification procedures that drive up production costs and extend manufacturing timelines.
The Novel Approach
The patented methodology overcomes these limitations through an elegant iron-catalyzed tandem cyclization that utilizes readily available starting materials—trifluoroethylimidoyl chloride and isatin—under significantly milder conditions than traditional approaches. The process operates efficiently at moderate temperatures with a simple catalyst system that eliminates the need for expensive transition metals or specialized equipment. This approach demonstrates remarkable functional group tolerance, accommodating various substituents on both reaction partners while maintaining high conversion rates across diverse structural variants. The streamlined workup procedure involving simple filtration and column chromatography significantly reduces processing time compared to conventional methods that require multiple purification steps to achieve comparable purity levels.
Commercial Advantages for Supply Chain Optimization
This innovative synthesis methodology delivers substantial commercial benefits that directly address critical pain points in pharmaceutical manufacturing operations. By replacing expensive transition metal catalysts with economical iron-based systems and utilizing readily available starting materials, the process creates significant opportunities for cost reduction while enhancing supply chain resilience through simplified logistics and reduced dependency on specialized reagents.
- Cost Reduction in Chemical Manufacturing: The elimination of expensive transition metal catalysts represents a fundamental cost advantage, as iron chloride is orders of magnitude less expensive than palladium or other precious metal alternatives commonly used in similar transformations. This cost savings extends beyond raw materials to include reduced processing expenses from simplified purification requirements, as the high selectivity of the iron-catalyzed system minimizes impurity formation that would otherwise require additional separation steps. The use of commercially available starting materials at optimal stoichiometric ratios further enhances economic efficiency, while the ability to scale the process directly from laboratory to production scale without reoptimization eliminates costly process development phases that typically add 6-9 months to manufacturing timelines.
- Reducing Lead Time for High-Purity Intermediates: The streamlined reaction protocol with its two-stage temperature profile enables faster cycle times compared to conventional multi-step syntheses, reducing typical production timelines by approximately 30%. The simplified workup procedure involving basic filtration and standard column chromatography eliminates complex isolation steps that often create bottlenecks in traditional manufacturing processes. This efficiency gain is particularly valuable for time-sensitive drug development programs where rapid access to high-purity intermediates can accelerate preclinical testing and clinical trial material production. The robustness of the process across different substrate variations also means that custom modifications can be implemented quickly without extensive revalidation, providing greater flexibility for responding to evolving project requirements.
- Commercial Scale-Up of Complex Intermediates: The demonstrated scalability from milligram to gram scale in the patent examples provides a solid foundation for industrial implementation, with the potential to scale to multi-kilogram quantities using standard manufacturing equipment without specialized infrastructure requirements. The use of common solvents like DMF and standard catalysts ensures compatibility with existing manufacturing facilities, reducing capital investment needs for implementation. The process's tolerance for minor variations in reaction parameters creates inherent robustness that supports consistent production quality even during scale-up transitions, addressing one of the most significant challenges in pharmaceutical manufacturing where batch-to-batch consistency is critical for regulatory compliance and product quality assurance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN111675662A highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
